南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 325-334.doi: 10.12122/j.issn.1673-4254.2026.02.10
• • 上一篇
邹晓松1,2( ), 张幸1, 李平1, 田瑞雪1, 路晓淼1,2(
), 张幸1, 李平1, 田瑞雪1, 路晓淼1,2( )
)
收稿日期:2025-07-11
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
路晓淼
E-mail:17755243117@163.com;luxmiao@sina.com
作者简介:邹晓松,在读硕士研究生,E-mail: 17755243117@163.com
基金资助:
Xiaosong ZOU1,2( ), Xing ZHANG1, Ping LI1, Ruixue TIAN1, Xiaomiao LU1,2(
), Xing ZHANG1, Ping LI1, Ruixue TIAN1, Xiaomiao LU1,2( )
)
Received:2025-07-11
Online:2026-02-20
Published:2026-03-10
Contact:
Xiaomiao LU
E-mail:17755243117@163.com;luxmiao@sina.com
摘要:
目的 探讨姜黄素通过影响Ran结合蛋白3样(RANBP3L)的表达对2型糖尿病(T2DM)引起的骨质疏松的治疗作用。 方法 取MC3T3-E1细胞,成骨诱导培养后随机分为以下4组:对照组(Control),高糖处理组(HG),姜黄素组(Cur),高糖处理+姜黄素组(HG+Cur),药物预先干预24 h。CCK-8法筛选姜黄素最佳干预浓度;碱性磷酸酶(ALP)和茜素红S(ARS)染色检测成骨分化和矿化相关标志物;采用RNA-seq测序分析姜黄素干预后高糖下培养MC3T3-E1成骨细胞转录特征。体内实验中,通过高脂饮食(HFD)和链脲霉素(STZ)注射建立T2DM小鼠模型,20只8周龄雄性C57BL/6小鼠随机分为4组: 正常+溶剂组(Ctrl+Veh)、T2DM模型+溶剂组(T2DM+Veh)、T2DM模型+ RANBP3L敲低+溶剂组(T2DM+KD+Veh)、T2DM模型+ RANBP3L敲低+姜黄素组(T2DM+KD+Cur),5只/组。T2DM+KD+Cur组每日给予58 mg/kg的姜黄素灌胃处理,其余各组给予等量溶剂灌胃。ELISA检测小鼠血脂水平;HE染色观察骨微结构特征;茜素红S染色检测钙盐沉积情况;免疫组化染色检测ALP、RANBP3L表达;Q-PCR和Western blotting检测小鼠骨组织中RANBP3L、OCN、RANKL表达,及TNF-α/NF-κB通路相关蛋白表达情况。 结果 姜黄素可促进高糖环境下MC3T3-E1细胞的增殖和成骨分化,下调RANBP3L蛋白的表达(P<0.01)。体内研究表明,当RANBP3L基因敲减后,模型小鼠的骨微结构增强,成骨标志物水平提高,血糖浓度降低,血脂平衡改善,且TNF-α/NF-κB信号通路相关蛋白受抑制(P<0.05),联用姜黄素后可进一步降低小鼠骨组织中RANBP3L的表达并加强治疗效果(P<0.05)。 结论 姜黄素可通过降低RANBP3L的表达,促进成骨分化能力,缓解糖脂代谢紊乱,减轻二型糖尿病骨质疏松症,可能通过抑制TNF-α/NF-κB信号通路发挥作用。
邹晓松, 张幸, 李平, 田瑞雪, 路晓淼. 姜黄素通过抑制RANBP3L基因表达减轻2型糖尿病小鼠的骨质疏松症[J]. 南方医科大学学报, 2026, 46(2): 325-334.
Xiaosong ZOU, Xing ZHANG, Ping LI, Ruixue TIAN, Xiaomiao LU. Curcumin improves osteoporosis in type 2 diabetic mice by inhibiting RANBP3L expression[J]. Journal of Southern Medical University, 2026, 46(2): 325-334.
| Gene | Primer sequences (5'to3') | Size (bp) |
|---|---|---|
| GAPDH-m | F:AGGTCGGTGTGAACGGATTTG | 95 |
| R:GGGGTCGTTGATGGCAACA | ||
| RANBP3L | F:GAGAAAGCGTGTAAGGTCTTCA | 82 |
| R:CGTTTGAGACAAAGTTGCCAC | ||
| OCN | F:AGTGTGAGCTTAACCCTGCT | 156 |
| R:GAGGATCAAGTCCCGGAGAG | ||
| RANKL | F:CGCTCTGTTCCTGTACTTTCG | 114 |
| R:GAGTCCTGCAAATCTGCGTT |
表1 实时聚合酶链反应引物序列
Tab.1 Primer sequences for RT-qPCR
| Gene | Primer sequences (5'to3') | Size (bp) |
|---|---|---|
| GAPDH-m | F:AGGTCGGTGTGAACGGATTTG | 95 |
| R:GGGGTCGTTGATGGCAACA | ||
| RANBP3L | F:GAGAAAGCGTGTAAGGTCTTCA | 82 |
| R:CGTTTGAGACAAAGTTGCCAC | ||
| OCN | F:AGTGTGAGCTTAACCCTGCT | 156 |
| R:GAGGATCAAGTCCCGGAGAG | ||
| RANKL | F:CGCTCTGTTCCTGTACTTTCG | 114 |
| R:GAGTCCTGCAAATCTGCGTT |
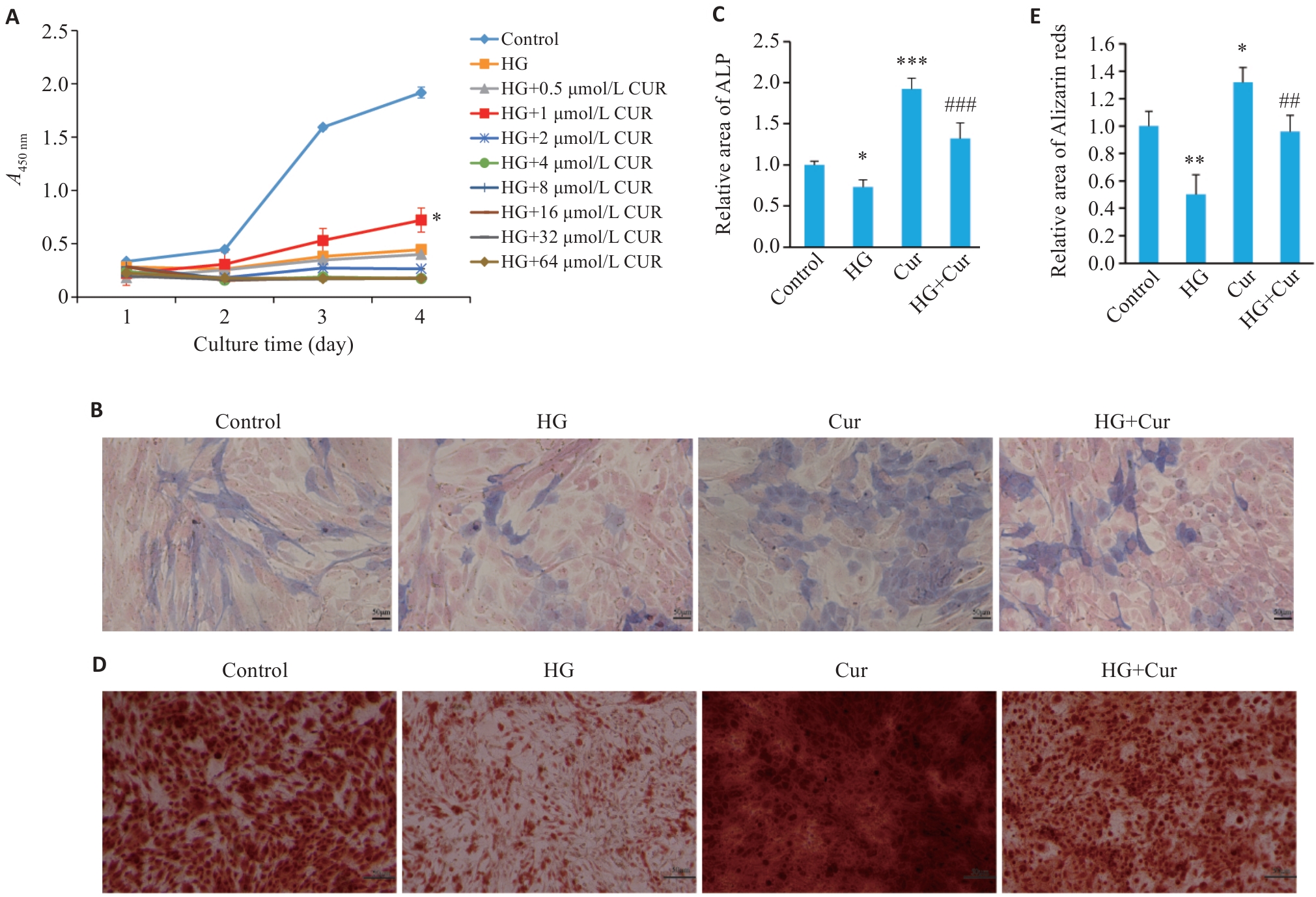
图1 姜黄素促进高糖条件下MC3T3-E1细胞的增殖及成骨分化
Fig.1 Curcumin promotes proliferation and osteogenic differentiation of MC3T3-E1 cells in high-glucose (HG) culture. A: CCK-8 assay for assessing cell viability following HG culture and treatment with different concentrations of curcumin. B: ALP staining of the cells after curcumin treatment for 7 days (Scale bar=50 μm). C: Alizarin red staining of the cells after curcumin treatment for 14 days. D: Relative area of ALP staining (Scale bar=50 μm). E: Relative area of Alizarin red staining. n=3. *P<0.05 vs HG group, **P<0.01, ***P<0.001 vs Control; ##P<0.01, ###P<0.001 vs HG.
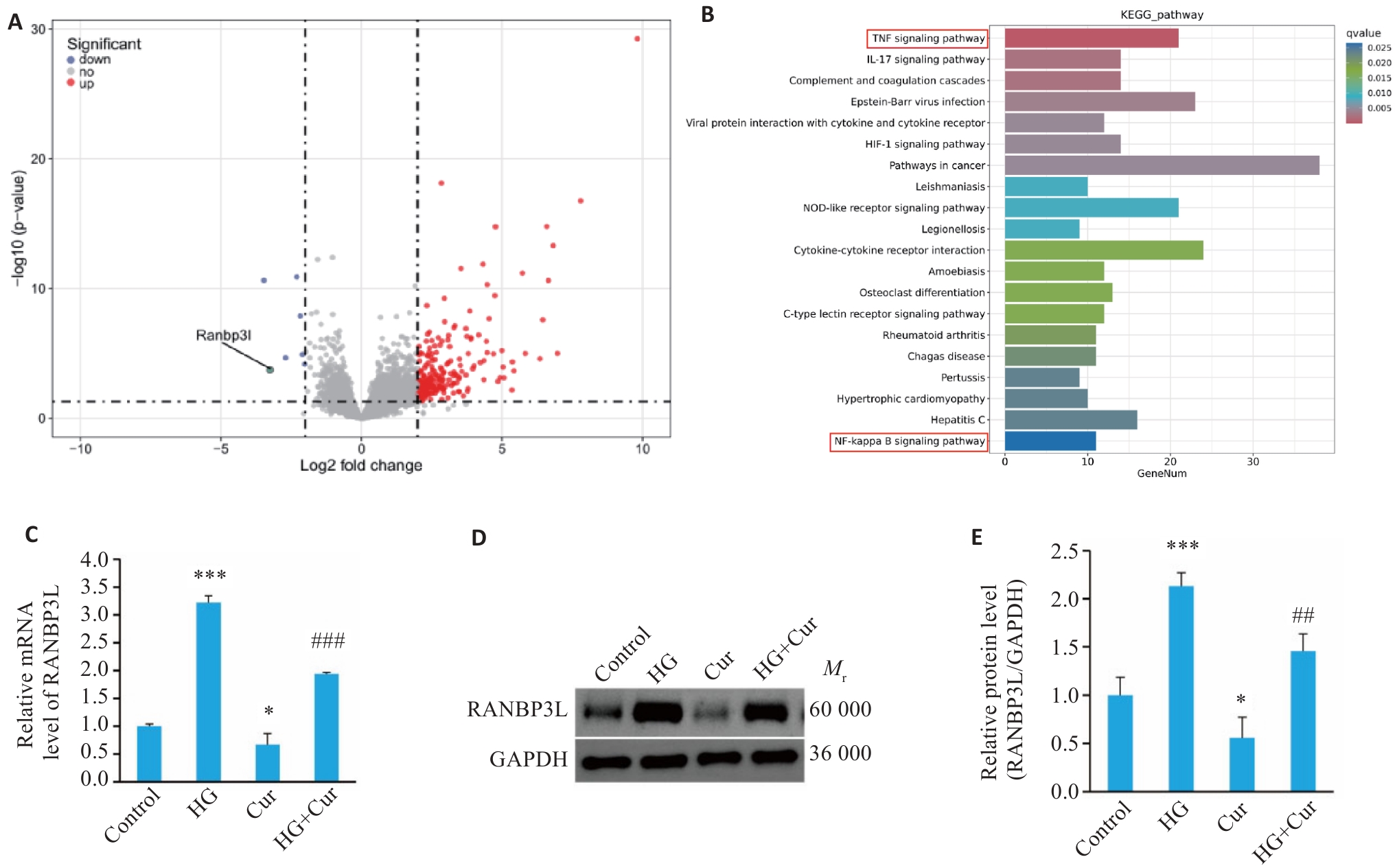
图2 姜黄素可降低高糖条件下MC3T3-E1细胞RANBP3L的表达
Fig.2 Curcumin reduces RANBP3L expression in MC3T3-E1 cells cultured in high glucose. A: Volcano plot showing the distribution of differentially expressed mRNAs. B: KEGG pathway analysis of the pivotal signaling pathways within the differentially expressed genes. C: mRNA expression of RANBP3L detected by qRT-PCR. D, E: Protein expression of RANBP3L detected by Western blotting. n=3. *P<0.05, ***P<0.001 vs Control; ##P<0.01, ###P<0.001 vs HG.
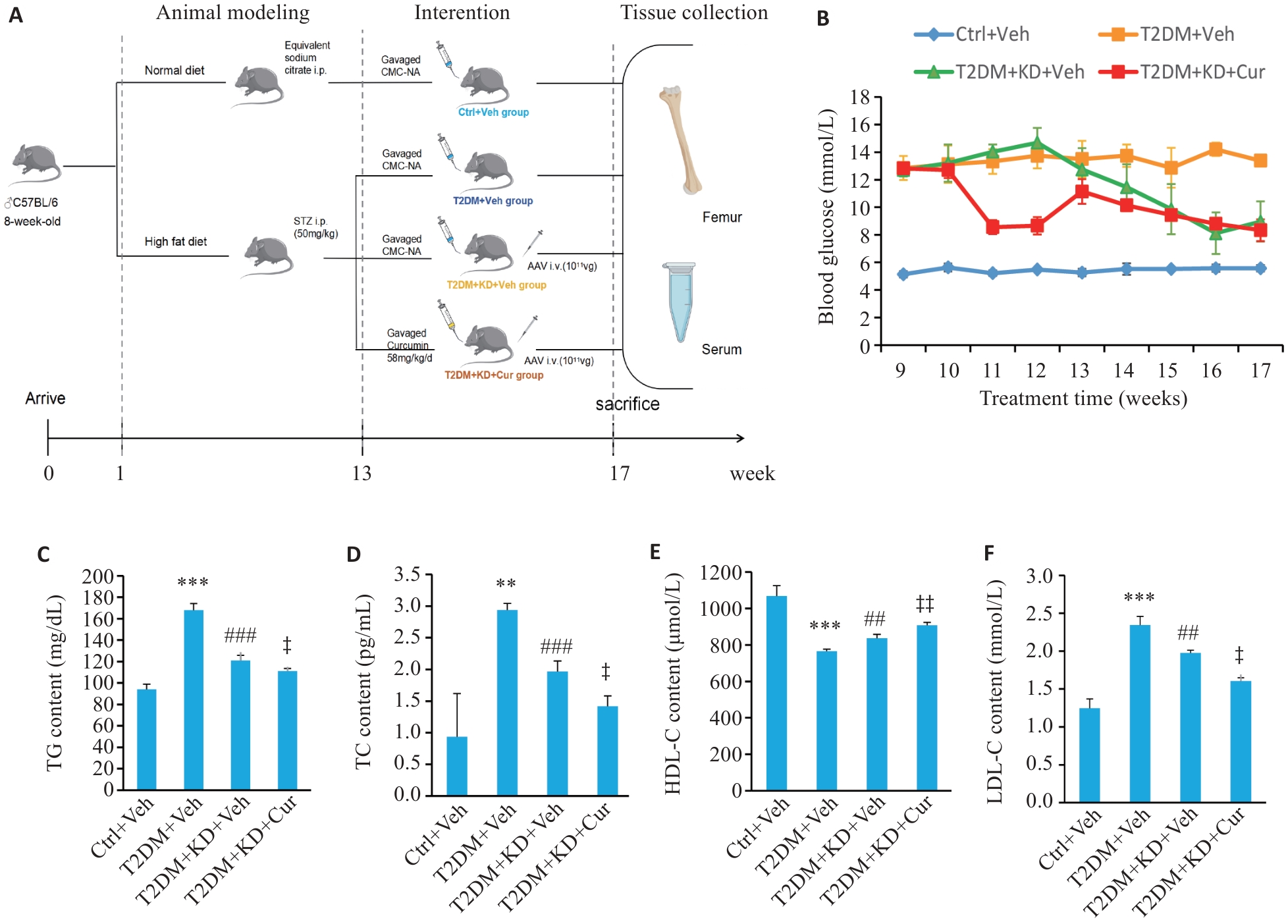
图3 各组小鼠的血糖和血脂水平
Fig.3 Blood glucose and lipid profiles of the mice in each group. A: Schematic diagram of the experimental process. B: Fasting blood glucose of the mice after STZ injection. C-F: TC, TG, LDL-C and HDL-C at the 17rd week. n=5. **P<0.01, ***P<0.001 vs Ctrl+Veh; ##P<0.01, ###P<0.001 vs T2DM+Veh; ‡P<0.05, ‡‡P<0.01 vs T2DM+KD+Veh.
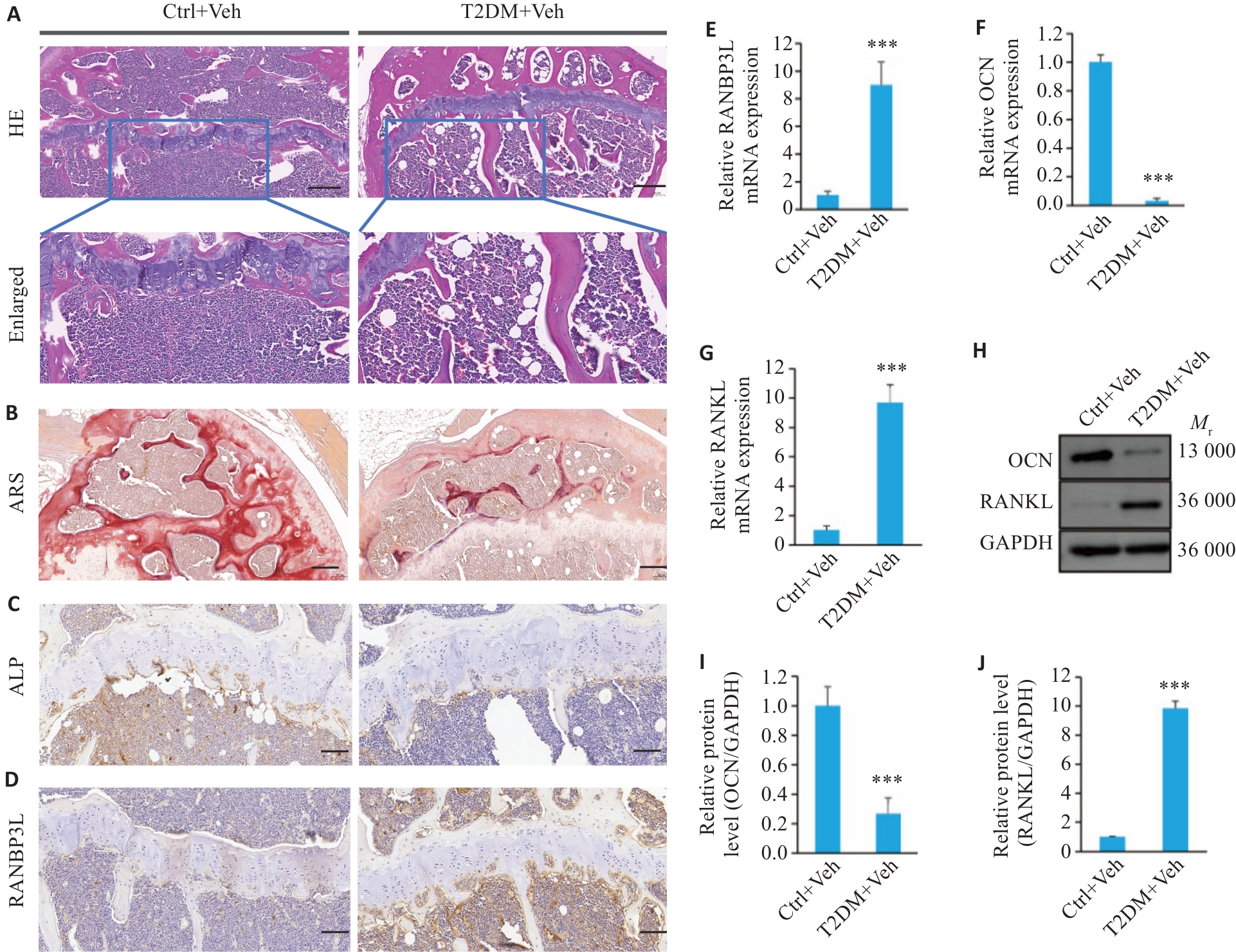
图4 糖尿病模型小鼠表现出骨形成减少
Fig.4 Diabetic mice show reduced bone formation. A: HE staining of the bone tissues (Scale bar=200 μm). B: Alizarin Red Staining of the bone tissues (Scale bar=200 μm). C, D: Immunohistochemistry for ALP and RANBP3L (Scale bar=100 μm). E-G: mRNA expression of RANBP3L, OCN and RANKL detected by qRT-PCR. H-J: Protein expressions of OCN and RANKL detected by Western blotting. n=5. ***P<0.001 vs Ctrl+Veh.
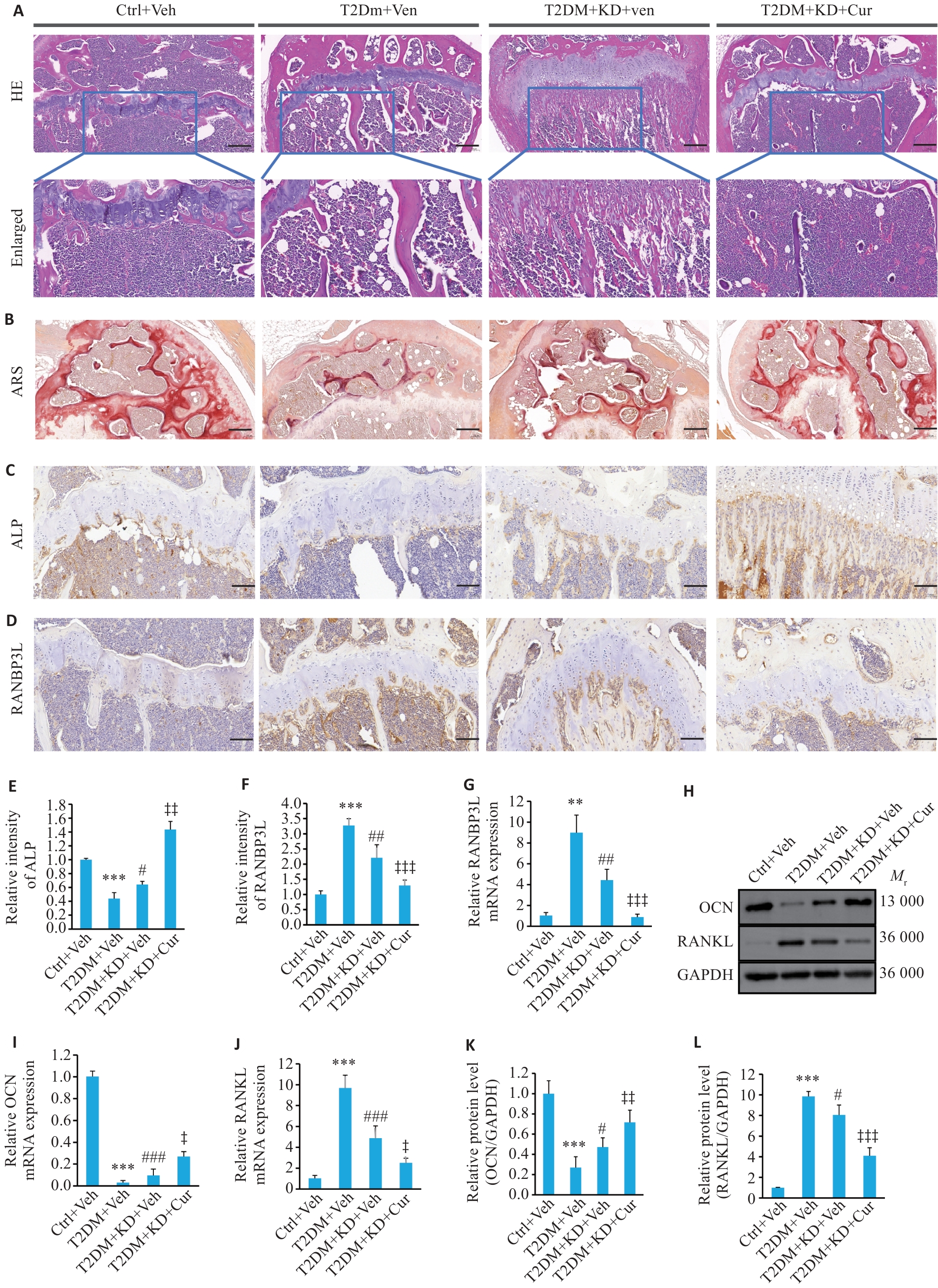
图5 姜黄素下调RANBP3L促进T2DM小鼠骨形成
Fig.5 Curcumin down-regulates RANBP3L expression and promotes bone formation in T2DM mice. A: HE staining of the bone tissues (Scale bar=200 μm). B: Alizarin Red Staining of the bone tissues (Scale bar=200 μm). C-F: Immunohistochemistry for ALP and RANBP3L and quantitative histograms (Scale bar =100 μm). G, I and J: mRNA expressions of RANBP3L, OCN and RANKL detected by qRT-PCR. H, K and L: Protein expressions of OCN and RANKL detected by Western blotting. n=5. **P<0.01, ***P<0.001 vs Ctrl+Veh; #P<0.05, ##P<0.01, ###P<0.001 vs T2DM+Veh; ‡P<0.05, ‡‡P<0.01, ‡‡‡P<0.001 vs T2DM+KD+Veh.
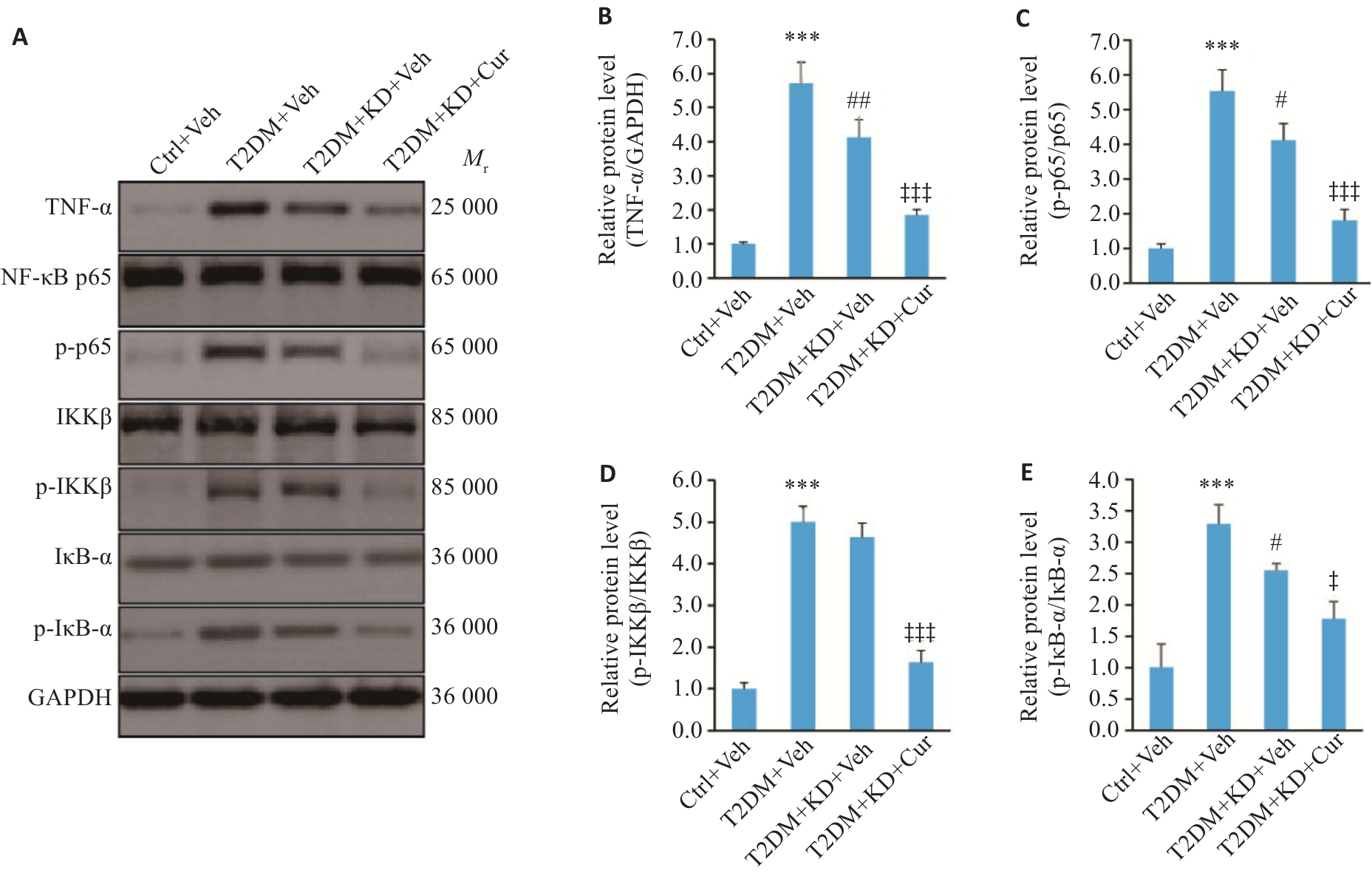
图6 下调RANBP3L抑制TNF-α/NF-κB信号通路
Fig.6 Effects of RANBP3L knockdown and curcumin treatment on the TNF‑α/NF‑κB signaling pathway. A-E: Protein expressions of TNF-α, p-p65, p-IKKβ and p-IκB-α detected by Western blotting. n=5. ***P<0.001 vs Ctrl+Veh; #P<0.05, ##P<0.01 vs T2DM+Veh; ‡P<0.05, ‡‡‡P<0.001 vs T2DM+KD+Veh.
| [1] | Wu B, Fu ZY, Wang XY, et al. A narrative review of diabetic bone disease: Characteristics, pathogenesis, and treatment[J]. Front Endocrinol (Lausanne), 2022, 13: 1052592. doi:10.3389/fendo.2022.1052592 |
| [2] | Armutcu F, McCloskey E. Insulin resistance, bone health, and fracture risk[J]. Osteoporos Int, 2024, 35(11): 1909-17. doi:10.1007/s00198-024-07227-w |
| [3] | de Araújo IM, Moreira MLM, de Paula FJA. Diabetes and bone[J]. Arch Endocrinol Metab, 2022, 66(5): 633-41. doi:10.20945/2359-3997000000552 |
| [4] | Halim M, Halim A. The effects of inflammation, aging and oxidative stress on the pathogenesis of diabetes mellitus (type 2 diabetes)[J]. Diabetes Metab Syndr, 2019, 13(2): 1165-72. doi:10.1016/j.dsx.2019.01.040 |
| [5] | Hofbauer LC, Busse B, Eastell R, et al. Bone fragility in diabetes: novel concepts and clinical implications[J]. Lancet Diabetes Endocrinol, 2022, 10(3): 207-20. doi:10.1016/s2213-8587(21)00347-8 |
| [6] | Lecka-Czernik B. Diabetes, bone and glucose-lowering agents: basic biology[J]. Diabetologia, 2017, 60(7): 1163-9. doi:10.1007/s00125-017-4269-4 |
| [7] | Su JQ, Luo YS, Hu S, et al. Advances in research on type 2 diabetes mellitus targets and therapeutic agents[J]. Int J Mol Sci, 2023, 24(17): 13381. doi:10.3390/ijms241713381 |
| [8] | Padhi S, Nayak AK, Behera A. Type II diabetes mellitus: a review on recent drug based therapeutics[J]. Biomed Pharmacother, 2020, 131: 110708. doi:10.1016/j.biopha.2020.110708 |
| [9] | Akaberi M, Sahebkar A, Emami SA. Turmeric and curcumin: from traditional to modern medicine[J]. Adv Exp Med Biol, 2021, 1291: 15-39. doi:10.1007/978-3-030-56153-6_2 |
| [10] | Kocaadam B, Şanlier N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health[J]. Crit Rev Food Sci Nutr, 2017, 57(13): 2889-95. doi:10.1080/10408398.2015.1077195 |
| [11] | Abu-Taweel GM, Attia MF, Hussein J, et al. Curcumin nanoparticles have potential antioxidant effect and restore tetrahydrobiopterin levels in experimental diabetes[J]. Biomed Pharmacother, 2020, 131: 110688. doi:10.1016/j.biopha.2020.110688 |
| [12] | Pivari F, Mingione A, Brasacchio C, et al. Curcumin and type 2 diabetes mellitus: prevention and treatment[J]. Nutrients, 2019, 11(8): 1837. doi:10.3390/nu11081837 |
| [13] | Bozkurt O, Kocaadam-Bozkurt B, Yildiran H. Effects of curcumin, a bioactive component of turmeric, on type 2 diabetes mellitus and its complications: an updated review[J]. Food Funct, 2022, 13(23): 11999-2010. doi:10.1039/d2fo02625b |
| [14] | Zhu XY, Xu XL, Du CG, et al. An examination of the protective effects and molecular mechanisms of curcumin, a polyphenol curcuminoid in diabetic nephropathy[J]. Biomed Pharmacother, 2022, 153: 113438. doi:10.1016/j.biopha.2022.113438 |
| [15] | Yang J, Miao X, Yang FJ, et al. Therapeutic potential of curcumin in diabetic retinopathy (Review)[J]. Int J Mol Med, 2021, 47(5): 75. doi:10.3892/ijmm.2021.4908 |
| [16] | Zhang W, Kang PF, Zhou J, et al. Curcumin improves diabetic cardiomyopathy by inhibiting pyroptosis through AKT/Nrf2/ARE pathway[J]. Mediators Inflamm, 2023, 2023: 3906043. doi:10.1155/2023/3906043 |
| [17] | Inchingolo AD, Inchingolo AM, Malcangi G, et al. Effects of resveratrol, curcumin and quercetin supplementation on bone metabolism-a systematic review[J]. Nutrients, 2022, 14(17): 3519. doi:10.3390/nu14173519 |
| [18] | Alsulaim AK, Almutaz TH, Albati AA, et al. Therapeutic potential of curcumin, a bioactive compound of turmeric, in prevention of streptozotocin-induced diabetes through the modulation of oxidative stress and inflammation[J]. Molecules, 2023, 29(1): 128. doi:10.3390/molecules29010128 |
| [19] | Liang YL, Zhu BB, Li SH, et al. Curcumin protects bone biomechanical properties and microarchitecture in type 2 diabetic rats with osteoporosis via the TGFβ/Smad2/3 pathway[J]. Exp Ther Med, 2020, 20(3): 2200-8. |
| [20] | Chernyakov D, Groß A, Fischer A, et al. Loss of RANBP3L leads to transformation of renal epithelial cells towards a renal clear cell carc-inoma like phenotype[J]. J Exp Clin Cancer Res, 2021, 40(1): 226. doi:10.1186/s13046-021-01982-y |
| [21] | Mueller L, Cordes VC, Bischoff FR, et al. Human RanBP3, a group of nuclear RanGTP binding proteins[J]. FEBS Lett, 1998, 427(3): 330-6. doi:10.1016/s0014-5793(98)00459-1 |
| [22] | Chen FF, Lin X, Xu PL, et al. Nuclear export of smads by RanBP3L regulates bone morphogenetic protein signaling and mesenchymal stem cell differentiation[J]. Mol Cell Biol, 2015, 35(10): 1700-11. doi:10.1128/mcb.00121-15 |
| [23] | Chen YL, Zhao W, Hu A, et al. Type 2 diabetic mellitus related osteoporosis: focusing on ferroptosis[J]. J Transl Med, 2024, 22(1): 409. doi:10.1186/s12967-024-05191-x |
| [24] | Galicia-Garcia U, Benito-Vicente A, Jebari S, et al. Pathophysiology of type 2 diabetes mellitus[J]. Int J Mol Sci, 2020, 21(17): 6275. doi:10.3390/ijms21176275 |
| [25] | Daryabor G, Atashzar MR, Kabelitz D, et al. The effects of type 2 diabetes mellitus on organ metabolism and the immune system[J]. Front Immunol, 2020, 11: 1582. doi:10.3389/fimmu.2020.01582 |
| [26] | 谢亚佳, 邓 真, 范德生. 姜黄素促进高糖环境下骨髓间充质干细胞成骨分化的作用及机制研究[J]. 上海中医药杂志, 2020, 54(2): 85-90. |
| [27] | Bukhari SNA, Hussain F, Thu HE, et al. Synergistic effects of combined therapy of curcumin and Fructus Ligustri Lucidi for treatment of osteoporosis: cellular and molecular evidence of enhanced bone formation[J]. J Integr Med, 2019, 17(1): 38-45. doi:10.1016/j.joim.2018.08.003 |
| [28] | 胥甜甜, 田昊春, 杨新民, 等. 姜黄素抑制NF-κB信号通路缓解氧化应激对成骨分化的损害发挥抗骨质疏松作用[J]. 中国药理学通报, 2024, 40(1): 46-54. |
| [29] | Cai L, Hou BJ, Hu JP. Tomatidine ameliorates high-fat-diet/streptozocin (HFD/STZ)-induced type 2 diabetes mellitus in mice[J]. Arch Physiol Biochem, 2024, 130(6): 848-53. doi:10.1080/13813455.2023.2298404 |
| [30] | Srinivasan K, Viswanad B, Asrat L, et al. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: a model for type 2 diabetes and pharmacological screening[J]. Pharmacol Res, 2005, 52(4): 313-20. doi:10.1016/j.phrs.2005.05.004 |
| [31] | Athyros VG, Doumas M, Imprialos KP, et al. Diabetes and lipid metabolism[J]. Hormones: Athens, 2018, 17(1): 61-7. doi:10.1007/s42000-018-0014-8 |
| [32] | Pan YY, Zhao DD, Yu N, et al. Curcumin improves glycolipid metabolism through regulating peroxisome proliferator activated receptor γ signalling pathway in high-fat diet-induced obese mice and 3T3-L1 adipocytes[J]. R Soc Open Sci, 2017, 4(11): 170917. doi:10.1098/rsos.170917 |
| [33] | Su LQ, Wang YD, Chi HY. Effect of curcumin on glucose and lipid metabolism, FFAs and TNF-α in serum of type 2 diabetes mellitus rat models[J]. Saudi J Biol Sci, 2017, 24(8): 1776-80. doi:10.1016/j.sjbs.2017.11.011 |
| [34] | Patel S, Santani D. Role of NF-kappa B in the pathogenesis of diabetes and its associated complications[J]. Pharmacol Rep, 2009, 61(4): 595-603. doi:10.1016/s1734-1140(09)70111-2 |
| [35] | Zamanian MY, Alsaab HO, Golmohammadi M, et al. NF‑κB pathway as a molecular target for curcumin in diabetes mellitus treatment: Focusing on oxidative stress and inflammation[J]. Cell Biochem Funct, 2024, 42(4): e4030. doi:10.1002/cbf.4030 |
| [36] | Li YF, Qiao YQ, Wang HH, et al. Intraperitoneal injection of PDTC on the NF-kB signaling pathway and osteogenesis indexes of young adult rats with anterior palatal suture expansion model[J]. PLoS One, 2021, 16(7): e0243108. doi:10.1371/journal.pone.0243108 |
| [37] | Yin Y, Weng YM, Ma ZY, et al. Tectochrysin alleviates periodontitis by modulating M2/M1 macrophage ratio and oxidative stress via nuclear factor kappa B/heme oxygenase-1/nuclear factor erythroid 2-related factor 2 pathway[J]. Immunol Invest, 2025, 54(1): 97-111. doi:10.1080/08820139.2024.2418938 |
| [38] | Fan DS, Lu JQ, Yu NJ, et al. Curcumin prevents diabetic osteoporosis through promoting osteogenesis and angiogenesis coupling via NF-κB signaling[J]. Evid Based Complement Alternat Med, 2022, 2022: 4974343. doi:10.1155/2022/4974343 |
| [39] | Wang LM, Zhao N, Zhang J, et al. Tumor necrosis factor-alpha inhibits osteogenic differentiation of pre-osteoblasts by down-regulation of EphB4 signaling via activated nuclear factor-kappaB signaling pathway[J]. J Periodontal Res, 2018, 53(1): 66-72. doi:10.1111/jre.12488 |
| [40] | Martiniakova M, Babikova M, Omelka R. Pharmacological agents and natural compounds: available treatments for osteoporosis[J]. J Physiol Pharmacol, 2020, 71(3). DOI: 10.26402/jpp.2020.3.01 . |
| [1] | 李丹丹, 楚佳鑫, 闫妍, 徐文隽, 朱行春, 孙韵, 丁浩峰, 任丽, 朱博. 姜黄素通过下调HIF-1α通路抑制非小细胞肺癌脂质代谢[J]. 南方医科大学学报, 2025, 45(5): 1039-1046. |
| [2] | 陈凯, 孟兆菲, 闵静婷, 王佳慧, 李正红, 高琴, 胡俊锋. 姜黄素通过抑制TXNIP/TRX-1/GPX4通路介导的铁死亡减轻脓毒症小鼠肺损伤[J]. 南方医科大学学报, 2024, 44(9): 1805-1813. |
| [3] | 任 丽, 邹明远, 朱行春, 徐文隽, 刘 刚, 孙俊杰, 范方田, 张从利. 姜黄素通过Keap1-Nrf2通路抑制甲状腺乳头状癌B-CPAP细胞的增殖、迁移及侵袭[J]. 南方医科大学学报, 2023, 43(8): 1356-1362. |
| [4] | 朱梦云, 崔双慧, 郝泽宇, 王文锐, 杨清玲, 陈昌杰, 王剑锋, 周 琦. 姜黄素通过 Wnt/β-catenin 信号通路诱导人晶状体上皮细胞的凋亡和细胞周期阻滞[J]. 南方医科大学学报, 2021, 41(5): 722-728. |
| [5] | 罗起胜,罗宏成,符黄德,黄海能,黄华东,罗琨祥,李传玉,胡仁统,郑传华,蓝川琉,唐乾利. 姜黄素通过抑制HDGF/β-catenin复合物降低胶质瘤细胞的侵袭性[J]. 南方医科大学学报, 2019, 39(08): 911-. |
| [6] | 朱国华,张琦,戴海萍,沈群. 姜黄素诱导Jurkat细胞凋亡时MAPKs及MMPs家族的表达[J]. 南方医科大学学报, 2013, 33(12): 1792-. |
| [7] | 李牧,王丽,刘海莉,苏宝山,刘变利,林温静,李昭荣,常黎华. 姜黄素通过降低一氧化氮合酶水平抑制宫颈癌HeLa细胞的侵袭和转移[J]. 南方医科大学学报, 2013, 33(12): 1752-. |
| [8] | 罗烨,欧阳平,赖文岩,许顶立. 姜黄素对肿瘤坏死因子-α诱导的大鼠平滑肌细胞Syndecan-4蛋白及磷酸化p44/42丝裂原活化蛋白激酶表达的影响[J]. 南方医科大学学报, 2012, 32(05): 722-. |
| [9] | 邱实1, 谭升顺1, 张江安1, 刘安1, 袁景奕1, 饶国洲2, 王文勇3. 姜黄素诱导人黑色素瘤A375细胞凋亡以及对c-myc、caspase-3表达的影响[J]. 南方医科大学学报, 2005, 25(12): 1517-1521. |
| [10] | 许刚, 黄文, 张卫民, 赖卓胜, 何美蓉, 王亚东, 张亚历. 姜黄素和儿茶素联用对二甲基肼诱导的大鼠大肠癌变过程中环氧合酶2表达的影响[J]. 南方医科大学学报, 2005, 25(01): 48-52. |
| [11] | 简燕婷1, 王继德1, 麦国丰2, 张亚历1, 赖卓胜1. 姜黄素对模型大鼠肠粘膜炎症因子的调控[J]. 南方医科大学学报, 2004, 24(12): 1353-1358. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||