Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 582-591.doi: 10.12122/j.issn.1673-4254.2026.03.12
Yizhen CHEN( ), Weili WANG, Meng CHENG, Wei ZHANG, Yilin GAO, Xin HONG, Lei ZHANG, Rong DAI(
), Weili WANG, Meng CHENG, Wei ZHANG, Yilin GAO, Xin HONG, Lei ZHANG, Rong DAI( ), Yiping WANG(
), Yiping WANG( )
)
Received:2025-08-04
Online:2026-03-20
Published:2026-03-26
Contact:
Rong DAI, Yiping WANG
E-mail:chenyizhen1996@126.com;azydairong@163.com;wypwyp54@aliyun.com
Supported by:Yizhen CHEN, Weili WANG, Meng CHENG, Wei ZHANG, Yilin GAO, Xin HONG, Lei ZHANG, Rong DAI, Yiping WANG. Qingshen Granules inhibits renal fibrosis in mice by regulating glycolytic reprogramming and H3K18 lactylation[J]. Journal of Southern Medical University, 2026, 46(3): 582-591.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.12
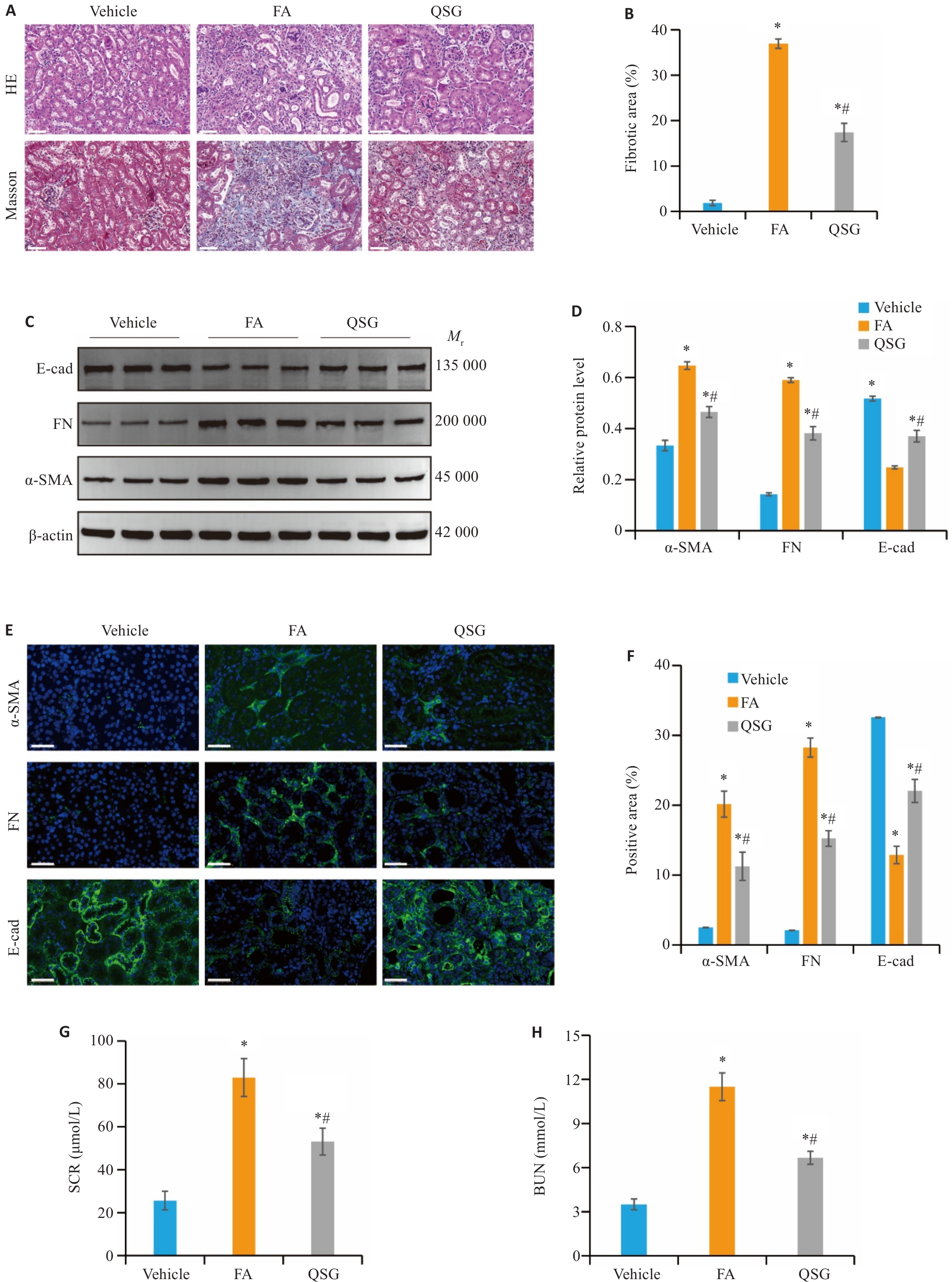
Fig.1 QSG inhibits FA-induced renal fibrosis in mice. A, B: HE and Masson staining (Scale bar=50 µm) and the quantitative data (n=6). C, D: Western blotting and quantitative analysis of expression levels of α-SMA, FN and E-cad in the renal tissues in each group (n=3). E, F: Immunofluorescence staining of α-SMA, FN and E-cad (Scale bar=50 µm) and the quantitative data (n=6). G, H: Serum creatinine (SCR) and blood urea nitrogen (BUN) levels of the mice (n=6). *P<0.05 vs Vehicle group; #P<0.05 vs FA group.
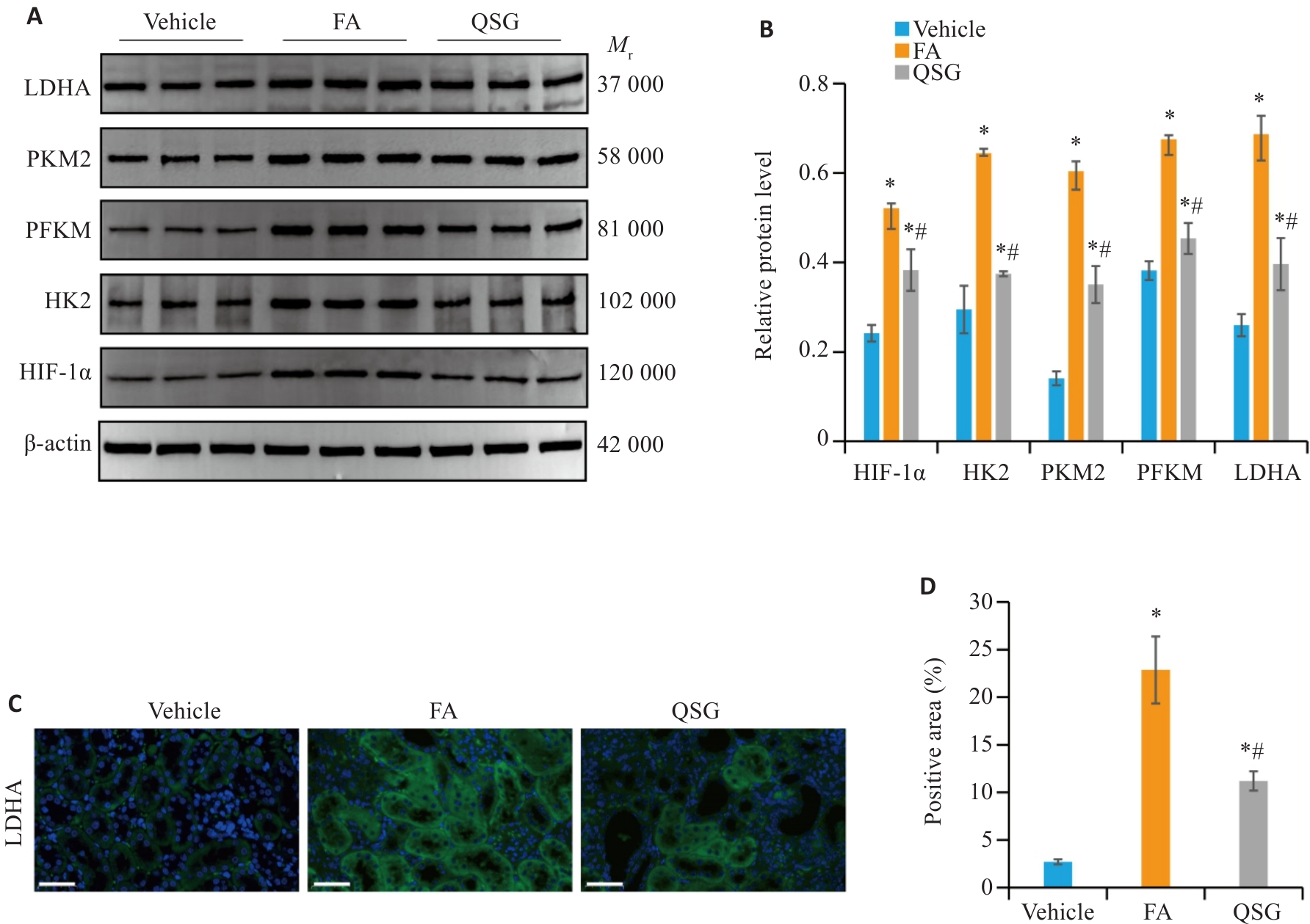
Fig.2 QSG regulates glycolytic reprogramming in renal tissues of the mice with FA-induced renal fibrosis. A, B: Western blotting and quantitative analysis of expression levels of HIF-1α, HK2, PFKM, PKM2 and LDHA in the renal tissues in each group (n=3). C, D: Immunofluorescence analysis of LDHA (Scale bar=50 µm) and the quantitative data (n=6). *P<0.05 vs Vehicle group; #P<0.05 vs FA group.
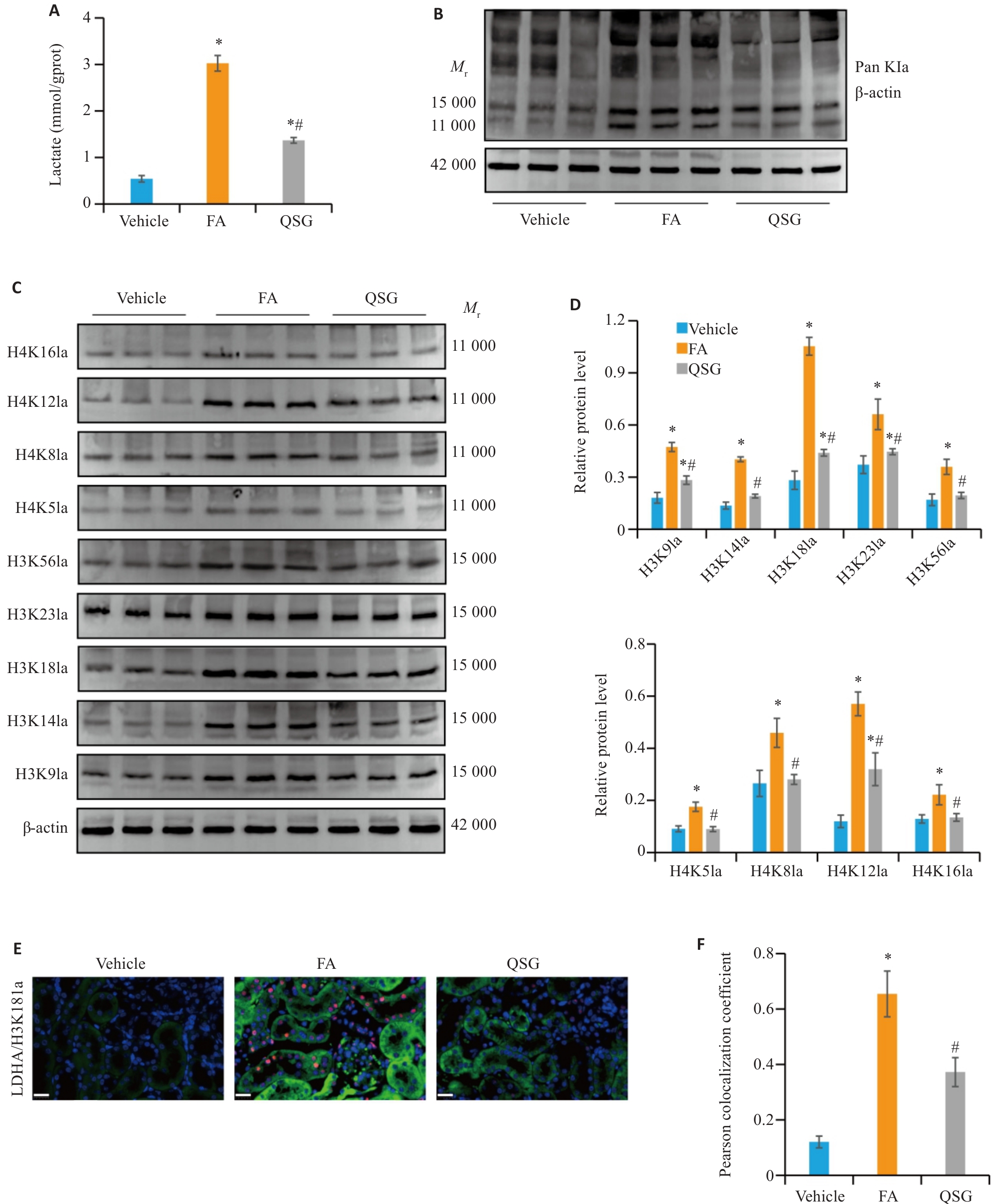
Fig.3 QSG inhibits lactate accumulation and histone lactylation in renal tissues of the mice. A: Renal tissue lactate levels in each group (n=6). B: Western blotting of Pan Kla in the renal tissues in each group (n=3). C, D:Western blotting and quantitative analysis of expression levels of H3K9la, H3K14la, H3K18la, H3K23la, H3K56la, H4K5la, H4K8la, H4K12la and H4K16la in the renal tissues in each group (n=3). E: Immunofluorescence co-staining of LDHA (green) and H3K18la (red) in renal tissues (Scale bar=20 µm). F: Quantitative analysis of Pearson colocalization coefficient for LDHA/H3K18la in the renal tissues (n=6). *P<0.05 vs Vehicle group; #P<0.05 vs FA group.
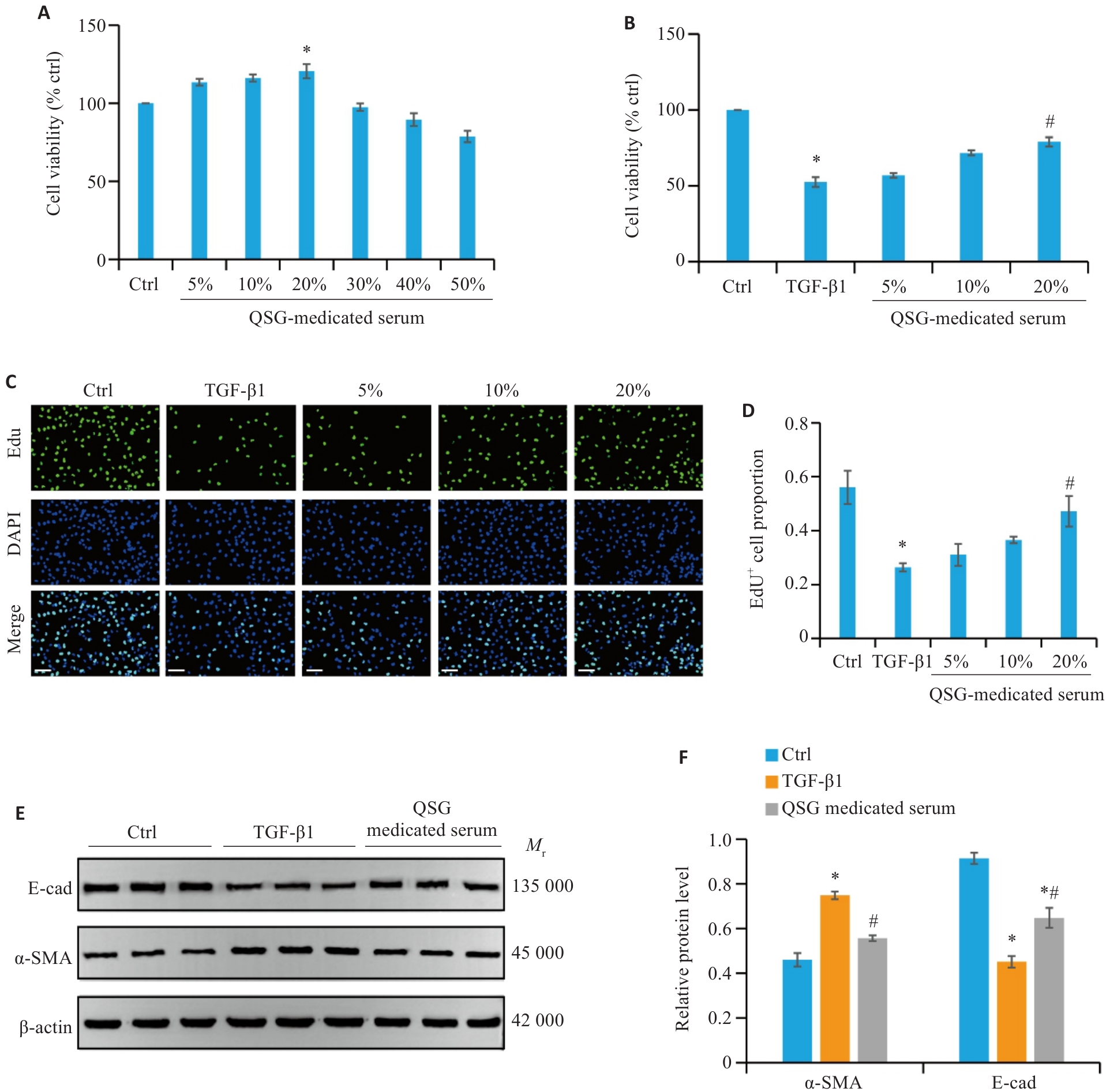
Fig.4 QSG-medicated serum inhibits TGF-β1-induced fibrosis in HK-2 cells. A: Cell viability of HK-2 cells treated with different concentrations of QSG (n=6). B: Cell viability of TGF‑β1-stimulated HK-2 cells treated with different concentrations of QSG (n=6). C, D: EdU cell proliferation assay (Scale bar=100 μm; n=3). E, F: Western blotting and quantitative analysis of expression levels of α-SMA and E-cad in the renal tissues in each group (n=3). *P<0.05 vs Control group; #P<0.05 vs TGF-β1 group.
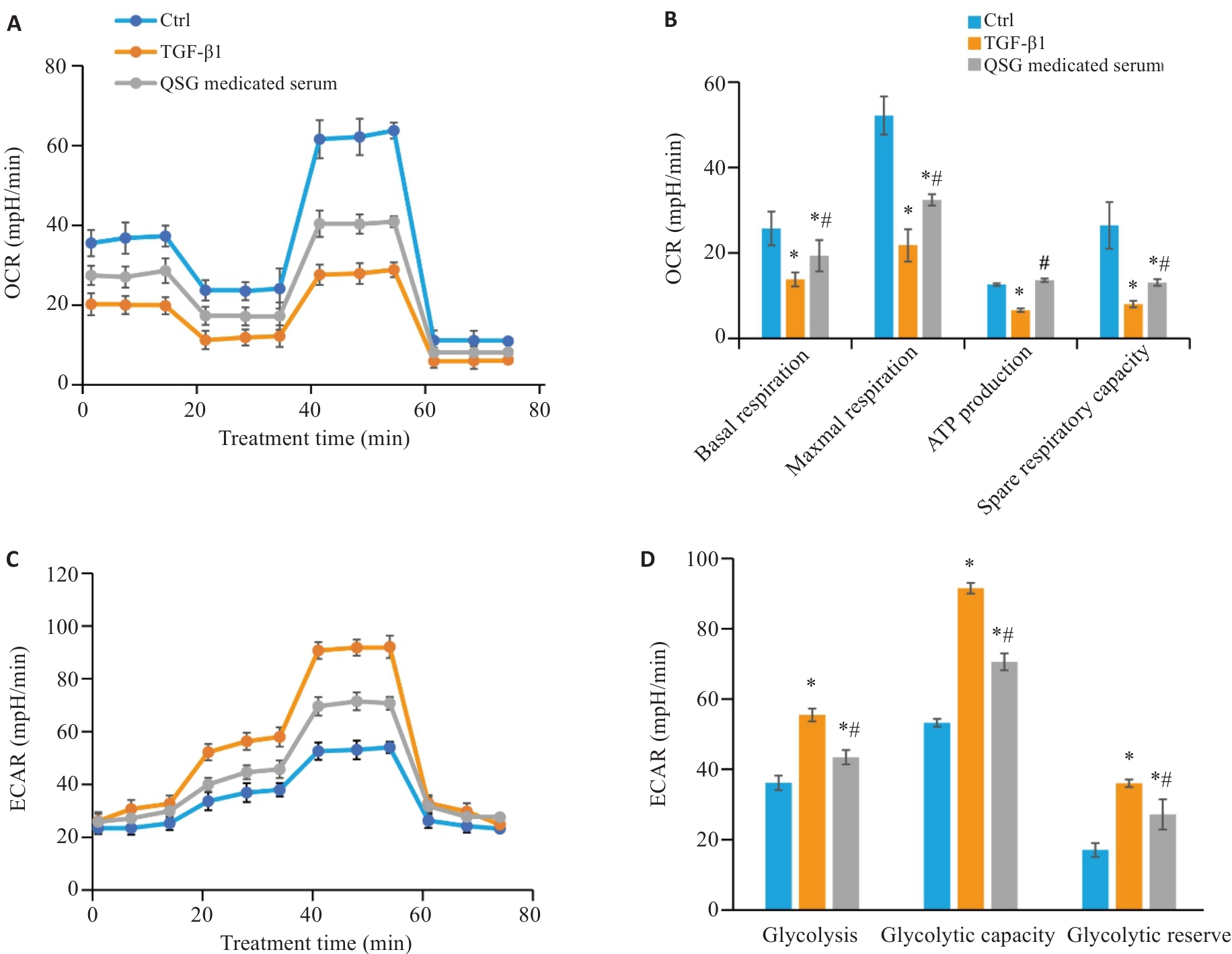
Fig.5 QSG-medicated serum regulates TGF-β1-induced glycolytic reprogramming in HK-2 cells. A, B: Changes in extracellular oxygen consumption rate (OCR) in HK-2 cells in each group (n=3). C, D: Changes in extracellular acidification rate (ECAR) in HK-2 cells in each group (n=3). *P<0.05 vs Control group; #P<0.05 vs TGF-β1 group.
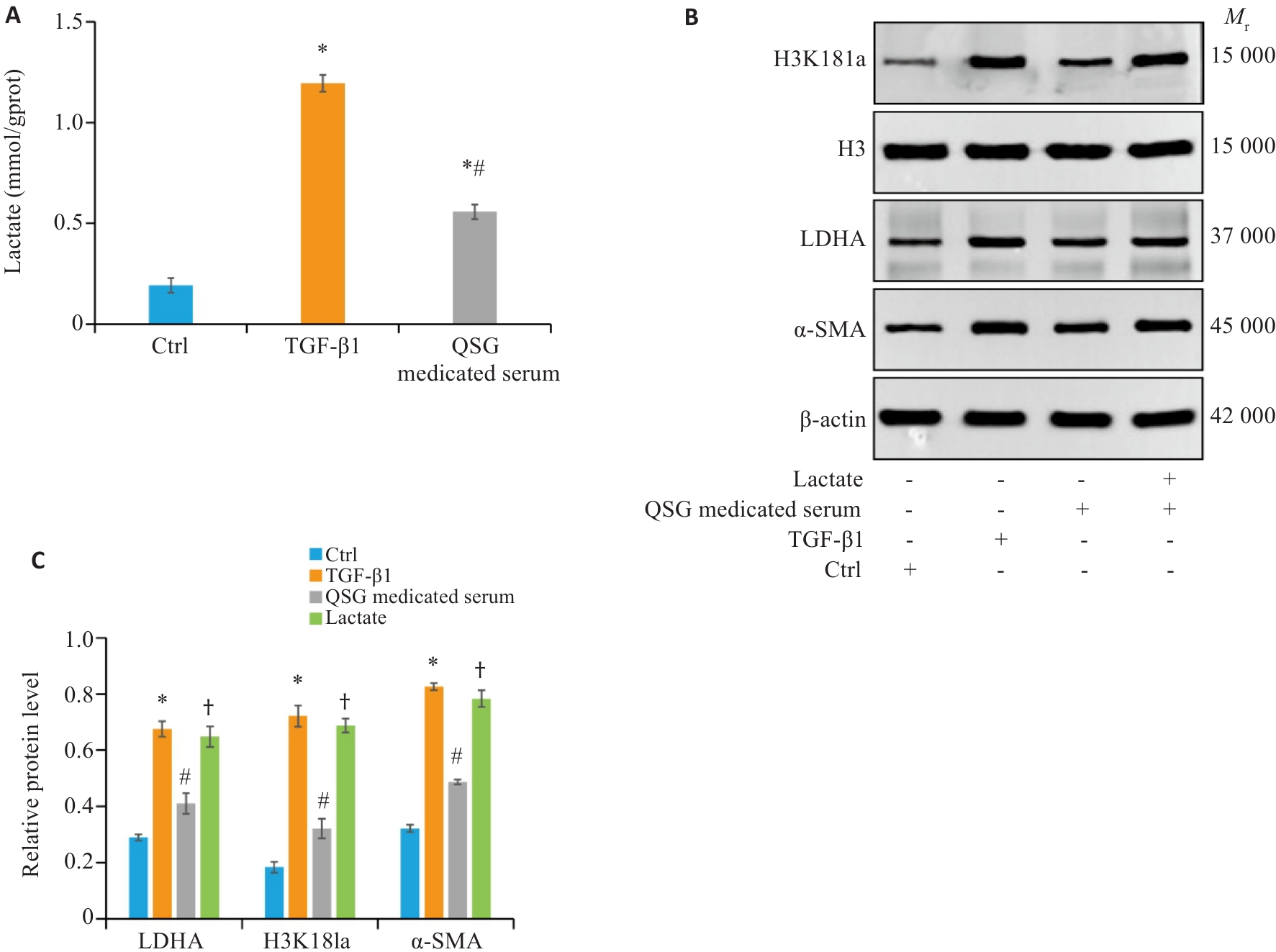
Fig.6 QSG-medicated serum inhibits TGF-β1-induced lactate accumulation and histone lactylation in HK-2 cells. A: Renal tissue lactate levels in each group (n=6). B, C: Western blotting and quantitative analysis of expression levels of LDHA, H3K18la and α‑SMA in the HK-2 cell in each group (n=3). *P<0.05 vs Control group; #P<0.05 vs TGF‑β1 group; †P<0.05 vs QSG-medicated serum group.
| [1] | Stevens PE, Ahmed SB, Carrero JJ, et al. KDIGO 2024 clinical practice guideline for the evaluation and management of chronic kidney disease[J]. Kidney Int, 2024, 105(4): S117-314. doi:10.1016/j.kint.2023.10.018 |
| [2] | Wang LM, Xu X, Zhang M, et al. Prevalence of chronic kidney disease in China: results from the sixth China chronic disease and risk factor surveillance[J]. JAMA Intern Med, 2023, 183(4): 298-310. doi:10.1001/jamainternmed.2022.6817 |
| [3] | Huang RS, Fu P, Ma L. Kidney fibrosis: from mechanisms to therapeutic medicines[J]. Signal Transduct Target Ther, 2023, 8(1): 129. doi:10.1038/s41392-023-01379-7 |
| [4] | Kang HM, Ahn SH, Choi P, et al. Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development[J]. Nat Med, 2015, 21(1): 37-46. doi:10.1038/nm.3762 |
| [5] | Zhu ZJ, Hu JJ, Chen ZW, et al. Transition of acute kidney injury to chronic kidney disease: role of metabolic reprogramming[J]. Metabolism, 2022, 131: 155194. doi:10.1016/j.metabol.2022.155194 |
| [6] | Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation[J]. Science, 2009, 324(5930): 1029-33. doi:10.1126/science.1160809 |
| [7] | Zhang L, Tian MQ, Zhang M, et al. Forkhead box protein K1 promotes chronic kidney disease by driving glycolysis in tubular epithelial cells[J]. Adv Sci (Weinh), 2024, 11(36): e2405325. doi:10.1002/advs.202405325 |
| [8] | Izzo LT, Wellen KE. Histone lactylation links metabolism and gene regulation[J]. Nature, 2019, 574(7779): 492-3. doi:10.1038/d41586-019-03122-1 |
| [9] | Hu XF, Chen W, Yang M, et al. IGFBP5 promotes EndoMT and renal fibrosis through H3K18 lactylation in diabetic nephropathy[J]. Cell Mol Life Sci, 2025, 82(1): 215. doi:10.1007/s00018-025-05718-5 |
| [10] | Wang D, Wang YP, Li CP, et al. Effects of Qingshen Granules on immune function in patients with comorbid chronic renal failure and damp-heat syndrome: a multicenter, randomized, controlled trial[J]. Evid Based Complement Alternat Med, 2020, 2020: 5057894. doi:10.1155/2020/5057894 |
| [11] | Wang YP, Zhang L, Jin H, et al. Based on HIF-1 α/wnt/β-catenin pathway to explore the effect of Qingshen Granules on chronic renal failure patients: a randomized controlled trial[J]. Evid Based Complement Alternat Med, 2019, 2019: 7656105. doi:10.1155/2019/7656105 |
| [12] | Wang YP, Wang D, Jin H, et al. Effects of Qingshen granules on Janus Kinase/signal transducer and activator of transcription signaling pathway in rats with unilateral ureteral obstruction[J]. J Tradit Chin Med, 2018, 38(2): 182-9. doi:10.1016/j.jtcm.2017.04.001 |
| [13] | Zhou WJ, Liang W, Hu MX, et al. Qingshen granules inhibits dendritic cell glycolipid metabolism to alleviate renal fibrosis via PI3K-AKT-mTOR pathway[J]. Phytomedicine, 2024, 135: 156148. doi:10.1016/j.phymed.2024.156148 |
| [14] | 戴 荣, 曹泽平, 刘传娇, 等. 清肾颗粒通过调控外泌体、miR-330-3p以及CREBBP表达抑制小鼠肾纤维化[J]. 南方医科大学学报, 2024, 44(8): 1431-40. doi:10.12122/j.issn.1673-4254.2024.08.01 |
| [15] | 刘 敏, 金 华, 呼 琴, 等. 清肾颗粒含药血清通过miR-23b-5p靶向Nrf2通路减轻NRK-52E细胞转分化[J]. 南方医科大学学报, 2023, 43(12): 2078-85. |
| [16] | Wang WL, Dai R, Cheng M, et al. Metabolic reprogramming and renal fibrosis: what role might Chinese medicine play[J]? Chin Med, 2024, 19(1): 148. doi:10.1186/s13020-024-01004-x |
| [17] | Miguel V, Shaw IW, Kramann R. Metabolism at the crossroads of inflammation and fibrosis in chronic kidney disease[J]. Nat Rev Nephrol, 2025, 21(1): 39-56. doi:10.1038/s41581-024-00889-z |
| [18] | Yang S, Wu H, Li YC, et al. Inhibition of PFKP in renal tubular epithelial cell restrains TGF-β induced glycolysis and renal fibrosis[J]. Cell Death Dis, 2023, 14(12): 816. doi:10.1038/s41419-023-06347-1 |
| [19] | Cao HD, Luo J, Zhang Y, et al. Tuberous sclerosis 1 (Tsc1) mediated mTORC1 activation promotes glycolysis in tubular epithelial cells in kidney fibrosis[J]. Kidney Int, 2020, 98(3): 686-98. doi:10.1016/j.kint.2020.03.035 |
| [20] | Wang ML, Zeng F, Ning FL, et al. Ceria nanoparticles ameliorate renal fibrosis by modulating the balance between oxidative phosphorylation and aerobic glycolysis[J]. J Nanobiotechnology, 2022, 20(1): 3. doi:10.1186/s12951-021-01122-w |
| [21] | Zhang XX, Chen JC, Lin RH, et al. Lactate drives epithelial-mesenchymal transition in diabetic kidney disease via the H3K14la/KLF5 pathway[J]. Redox Biol, 2024, 75: 103246. doi:10.1016/j.redox.2024.103246 |
| [22] | Wang YT, Li HY, Jiang SM, et al. The glycolytic enzyme PFKFB3 drives kidney fibrosis through promoting histone lactylation-mediated NF‑κB family activation[J]. Kidney Int, 2024, 106(2): 226-40. doi:10.1016/j.kint.2024.04.016 |
| [23] | Xiang TY, Wang XJ, Huang SJ, et al. Inhibition of PKM2 by shikonin impedes TGF‑β1 expression by repressing histone lactylation to alleviate renal fibrosis[J]. Phytomedicine, 2025, 136: 156324. doi:10.1016/j.phymed.2024.156324 |
| [24] | Liu Y, Zhou R, Guo YF, et al. Muscle-derived small extracellular vesicles induce liver fibrosis during overtraining[J]. Cell Metab, 2025, 37(4): 824-41.e8. doi:10.1016/j.cmet.2024.12.005 |
| [25] | Shen Y, Jiang L, Wen P, et al. Tubule-derived lactate is required for fibroblast activation in acute kidney injury[J]. Am J Physiol Renal Physiol, 2020, 318(3): F689-701. doi:10.1152/ajprenal.00229.2019 |
| [26] | Hou Y, Liu DW, Guo ZS, et al. Lactate and lactylation in AKI-to-CKD: epigenetic regulation and therapeutic opportunities[J]. Cell Prolif, 2025, 58(9): e70034. doi:10.1111/cpr.70034 |
| [27] | Wang PW, Xie DX, Xiao T, et al. H3K18 lactylation promotes the progression of arsenite-related idiopathic pulmonary fibrosis via YTHDF1/m6A/NREP[J]. J Hazard Mater, 2024, 461: 132582. doi:10.1016/j.jhazmat.2023.132582 |
| [28] | Wang NX, Wang WW, Wang XQ, et al. Histone lactylation boosts reparative gene activation post-myocardial infarction[J]. Circ Res, 2022, 131(11): 893-908. doi:10.1161/circresaha.122.320488 |
| [29] | Zhou YQ, Yan JX, Huang H, et al. The m6A reader IGF2BP2 regulates glycolytic metabolism and mediates histone lactylation to enhance hepatic stellate cell activation and liver fibrosis[J]. Cell Death Dis, 2024, 15(3): 189. doi:10.1038/s41419-024-06509-9 |
| [30] | Li WH, Zhou C, Yu L, et al. Tumor-derived lactate promotes resistance to bevacizumab treatment by facilitating autophagy enhancer protein RUBCNL expression through histone H3 lysine 18 lactylation (H3K18la) in colorectal cancer[J]. Autophagy, 2024, 20(1): 114-30. doi:10.1080/15548627.2023.2249762 |
| [31] | Jin Z, Yun L, Cheng P. Tanshinone I reprograms glycolysis metabolism to regulate histone H3 lysine 18 lactylation (H3K18la) and inhibits cancer cell growth in ovarian cancer[J]. Int J Biol Macromol, 2025, 291: 139072. doi:10.1016/j.ijbiomac.2024.139072 |
| [32] | Qiao J, Tan Y, Liu HC, et al. Histone H3K18 and ezrin lactylation promote renal dysfunction in sepsis-associated acute kidney injury[J]. Adv Sci (Weinh), 2024, 11(28): e2307216. doi:10.1002/advs.202307216 |
| [33] | Ye ZH, Sun YS, Yang SY, et al. Lgals3 promotes calcium oxalate crystal formation and kidney injury through histone lactylation-mediated FGFR4 activation[J]. Adv Sci (Weinh), 2025, 12(12): e2413937. doi:10.1002/advs.202413937 |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||