Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 513-522.doi: 10.12122/j.issn.1673-4254.2026.03.05
Caixia LI1( ), Lihua CUI1, Jie CAO2, Yuxing FAN2, Xueying ZHOU3, Shukun ZHANG1, Yanjie ZUO3(
), Lihua CUI1, Jie CAO2, Yuxing FAN2, Xueying ZHOU3, Shukun ZHANG1, Yanjie ZUO3( )
)
Received:2025-09-11
Online:2026-03-20
Published:2026-03-26
Contact:
Yanjie ZUO
E-mail:licaixia2013 @163.com;zyjcz9886@163.com
Caixia LI, Lihua CUI, Jie CAO, Yuxing FAN, Xueying ZHOU, Shukun ZHANG, Yanjie ZUO. Huoxue Qingjie Ling alleviates bile duct ligation-induced hepatic fibrosis in rats by regulating the Sirt1-autophagy signaling pathway[J]. Journal of Southern Medical University, 2026, 46(3): 513-522.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.05
| Gene | Primer sequences (5'-3') (Forward) | Primer sequences (5'-3') (Reverse) |
|---|---|---|
| FN | GATTCTTCTGGCGTCTGCAC | GATTCTTCTGGCGTCTGCAC |
| COL I | GGATAGGGACTTGTGTGA | GCTGGAAGAGTGAAGAGG |
| α-SMA | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Beclin-1 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Atg5 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| LC3B | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| P62 | ACGACTGGACGCATTTGTCT | TGGTGGGAGATGTGGGATCA |
| Sirt 1 | ACCGAGACAACCTCCTGTTG | ATGGACCTATCCGTGGCCTT |
| GAPDH | AGATGGGTGAAGGTCGGTGTG | CTGGAAGATGGTGATGGGTT |
Tab.1 Primer sequences for qRT-PCR
| Gene | Primer sequences (5'-3') (Forward) | Primer sequences (5'-3') (Reverse) |
|---|---|---|
| FN | GATTCTTCTGGCGTCTGCAC | GATTCTTCTGGCGTCTGCAC |
| COL I | GGATAGGGACTTGTGTGA | GCTGGAAGAGTGAAGAGG |
| α-SMA | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Beclin-1 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Atg5 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| LC3B | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| P62 | ACGACTGGACGCATTTGTCT | TGGTGGGAGATGTGGGATCA |
| Sirt 1 | ACCGAGACAACCTCCTGTTG | ATGGACCTATCCGTGGCCTT |
| GAPDH | AGATGGGTGAAGGTCGGTGTG | CTGGAAGATGGTGATGGGTT |
| Group | Dose (g/kg) | Body mass (g) | Liver mass (g) | Liver index (%) |
|---|---|---|---|---|
| Sham | 296.0±6.6 | 11.4±1.0 | 3.8±0.3 | |
| BDL | 198.1±11.81) | 17.9±1.81) | 9.0±0.81) | |
| HXQJL-L | 7.74 | 235.6±11.22) | 14.9±0.82) | 6.3±0.42) |
| HXQJL-H | 15.48 | 258.2±10.92)3) | 12.4±1.32)3) | 4.8±0.62) |
| EX527 | 0.005 | 163.6±8.82) | 21.4±1.02) | 13.1±1.02) |
| EX527+HXQJL | 0.005+15.48 | 204.0±13.04) | 17.5±2.14) | 8.6±0.94) |
Tab.2 Effect of Huoxue Qingjie Ling (HXQJL) on body weight, liver mass and liver index in rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Body mass (g) | Liver mass (g) | Liver index (%) |
|---|---|---|---|---|
| Sham | 296.0±6.6 | 11.4±1.0 | 3.8±0.3 | |
| BDL | 198.1±11.81) | 17.9±1.81) | 9.0±0.81) | |
| HXQJL-L | 7.74 | 235.6±11.22) | 14.9±0.82) | 6.3±0.42) |
| HXQJL-H | 15.48 | 258.2±10.92)3) | 12.4±1.32)3) | 4.8±0.62) |
| EX527 | 0.005 | 163.6±8.82) | 21.4±1.02) | 13.1±1.02) |
| EX527+HXQJL | 0.005+15.48 | 204.0±13.04) | 17.5±2.14) | 8.6±0.94) |
| Group | Dose (g/kg) | ALT (U/L) | AST (U/L) | ALP (U/L) | GGT (U/L) | |
|---|---|---|---|---|---|---|
| Sham | 52.0±15.8 | 136.8±13.3 | 155±26.9 | 1.8±0.7 | ||
| BDL | 128.2±13.11) | 383.3±711) | 389.2±53.51) | 47.7±19.61) | ||
| HXQJL-L | 7.74 | 59.3±9.62) | 182.8±20.52) | 214.7±31.42) | 15.8±4.22) | |
| HXQJL-H | 15.48 | 53.6±7.42) | 141.9±28.12) | 179.4±57.62) | 10.1±4.92) | |
| EX527 | 0.005 | 169.7±16.42) | 490±107.82) | 577.5±70.92) | 78.2±7.72) | |
| EX527+HXQJL | 0.005+15.48 | 105.7±31.64) 289.7±101.34) | 361.2±54.24) | 41.3±14.44) | ||
Tab.3 Effect of HXQJL on serum levels of ALT, AST, ALP and GGT in rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | ALT (U/L) | AST (U/L) | ALP (U/L) | GGT (U/L) | |
|---|---|---|---|---|---|---|
| Sham | 52.0±15.8 | 136.8±13.3 | 155±26.9 | 1.8±0.7 | ||
| BDL | 128.2±13.11) | 383.3±711) | 389.2±53.51) | 47.7±19.61) | ||
| HXQJL-L | 7.74 | 59.3±9.62) | 182.8±20.52) | 214.7±31.42) | 15.8±4.22) | |
| HXQJL-H | 15.48 | 53.6±7.42) | 141.9±28.12) | 179.4±57.62) | 10.1±4.92) | |
| EX527 | 0.005 | 169.7±16.42) | 490±107.82) | 577.5±70.92) | 78.2±7.72) | |
| EX527+HXQJL | 0.005+15.48 | 105.7±31.64) 289.7±101.34) | 361.2±54.24) | 41.3±14.44) | ||
| Group | Dose (g/kg) | Pathology score | Sirius red positive area (%) |
|---|---|---|---|
| Sham | 1.2±0.1 | 3.2±0.6 | |
| BDL | 3.9±0.31) | 47.5±7.81) | |
| HXQJL-L | 7.74 | 1.9±0.32) | 28.9±5.52) |
| HXQJL-H | 15.48 | 1.6±0.12) | 16.0±2.22) 3) |
| EX527 | 0.005 | 5.3±0.52) | 64.9±5.42) |
| EX527+HXQJL | 0.005+15.48 | 3.1±0.64) | 47.3±3.22) |
Tab.4 Effect of HXQJL on liver histopathological damage in rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Pathology score | Sirius red positive area (%) |
|---|---|---|---|
| Sham | 1.2±0.1 | 3.2±0.6 | |
| BDL | 3.9±0.31) | 47.5±7.81) | |
| HXQJL-L | 7.74 | 1.9±0.32) | 28.9±5.52) |
| HXQJL-H | 15.48 | 1.6±0.12) | 16.0±2.22) 3) |
| EX527 | 0.005 | 5.3±0.52) | 64.9±5.42) |
| EX527+HXQJL | 0.005+15.48 | 3.1±0.64) | 47.3±3.22) |
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 1.00±0.19 | 1.00±0.08 | 1.03±0.15 | |
| BDL | 2.84±0.621) | 11.87±3.151) | 4.62±1.121) | |
| HXQJL-L | 7.74 | 1.62±0.262) | 6.07±1.192) | 2.67±0.622) |
| HXQJL-H | 15.48 | 1.36±0.442) | 3.46±0.692) | 1.79±0.472) |
| EX527 | 0.005 | 5.36±1.062) | 18.96±3.362) | 8.56±1.392) |
| EX527+HXQJL | 0.005+15.48 | 3.08±1.404) | 13.79±4.924) | 3.91±1.54) |
Tab.5 Effect of HXQJL on mRNA level of FN, COL I and α-SMAin the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 1.00±0.19 | 1.00±0.08 | 1.03±0.15 | |
| BDL | 2.84±0.621) | 11.87±3.151) | 4.62±1.121) | |
| HXQJL-L | 7.74 | 1.62±0.262) | 6.07±1.192) | 2.67±0.622) |
| HXQJL-H | 15.48 | 1.36±0.442) | 3.46±0.692) | 1.79±0.472) |
| EX527 | 0.005 | 5.36±1.062) | 18.96±3.362) | 8.56±1.392) |
| EX527+HXQJL | 0.005+15.48 | 3.08±1.404) | 13.79±4.924) | 3.91±1.54) |
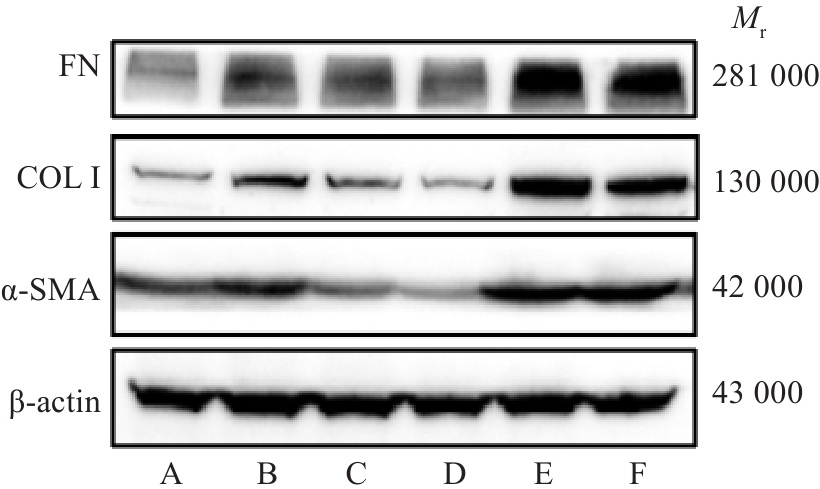
Fig.2 Effect of HXQJL on expression of FN, COL I and α‑SMA protein in liver tissues in rats with BDL-induced liver fibrosis. A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 0.11±0.02 | 0.14±0.03 | 0.36±0.06 | |
| BDL | 0.36±0.021) | 0.38±0.011) | 0.75±0.071) | |
| HXQJL-L | 7.74 | 0.14±0.01 | 0.20±0.032) | 0.40±0.012) |
| HXQJL-H | 15.48 | 0.12±0.012) | 0.17±0.032) | 0.33±0.042) |
| EX527 | 0.005 | 2.33±0.172) | 1.12±0.112) | 1.22±0.102) |
| EX527+HXQJL | 0.005+15.48 | 1.44±0.134) | 0.70±0.064) | 1.00±0.024) |
Tab.6 Effect of HXQJL on protein expression levels of FN, COL I and α-SMAin the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 0.11±0.02 | 0.14±0.03 | 0.36±0.06 | |
| BDL | 0.36±0.021) | 0.38±0.011) | 0.75±0.071) | |
| HXQJL-L | 7.74 | 0.14±0.01 | 0.20±0.032) | 0.40±0.012) |
| HXQJL-H | 15.48 | 0.12±0.012) | 0.17±0.032) | 0.33±0.042) |
| EX527 | 0.005 | 2.33±0.172) | 1.12±0.112) | 1.22±0.102) |
| EX527+HXQJL | 0.005+15.48 | 1.44±0.134) | 0.70±0.064) | 1.00±0.024) |
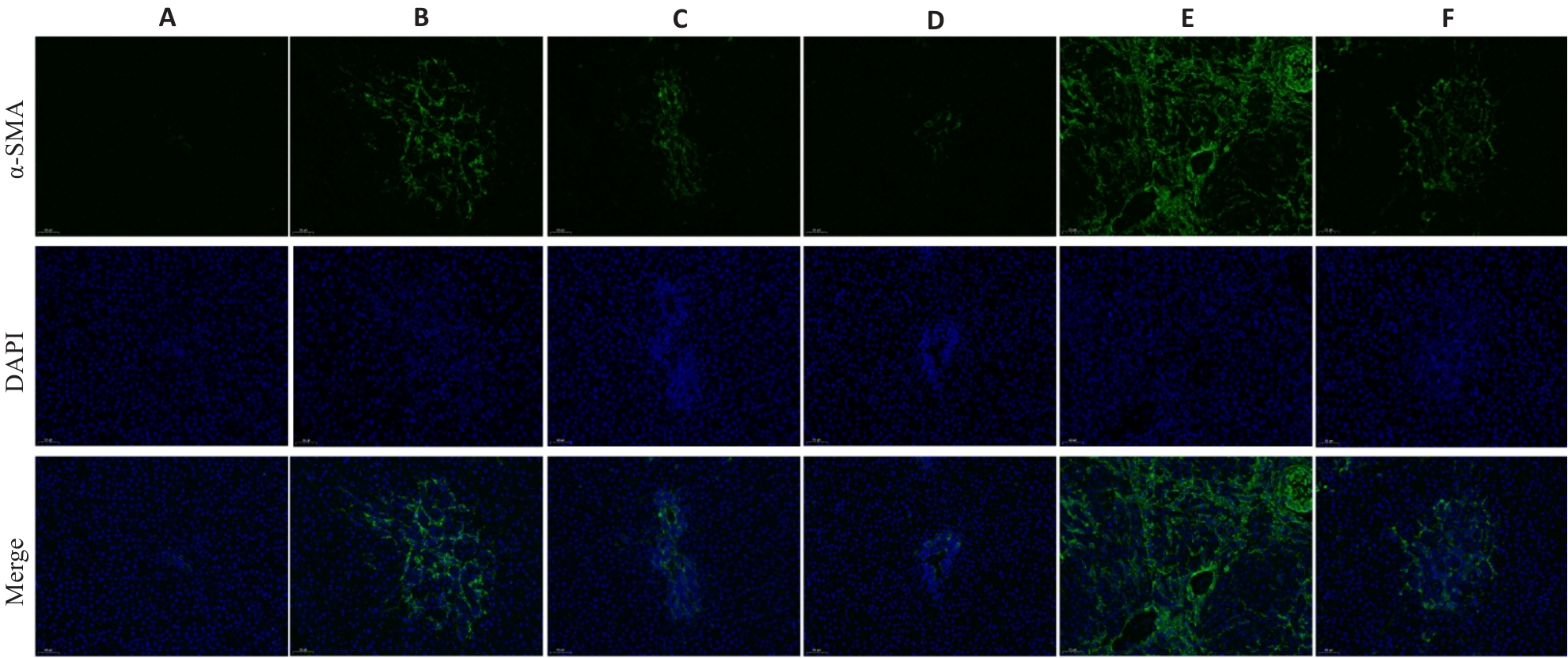
Fig.3 Effect of HXQJL onα-SMA expression in the liver tissues of rats with BDL-induced liver fibrosis(×200). A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Groups | Dose (g/kg) | Beclin-1 | Atg5 | LC3B | p62 |
|---|---|---|---|---|---|
| Sham | 1.01±0.08 | 1.00±0.20 | 1.00±0.17 | 1.00±0.14 | |
| BDL | 2.13±0.491) | 1.61±0.121) | 1.48±0.141) | 0.63±0.101) | |
| HXQJL-L | 7.74 | 1.11±0.082) | 1.09±0.182) | 0.92±0.162) | 0.83±0.182) |
| HXQJL-H | 15.48 | 1.03±0.182) | 0.85±0.202) | 0.83±0.252) | 0.98±0.142) |
| EX527 | 0.005 | 2.99±0.252) | 2.01±1.382) | 1.92±0.172) | 0.24±0.082) |
| EX527+HXQJL | 0.005+15.48 | 1.91±0.434) | 1.5±0.304) | 1.47±0.274) | 0.68±0.164) |
Tab.7 Effect of HXQJL on mRNA levels of Beclin-1, Atg5, LC3B and p62in the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Groups | Dose (g/kg) | Beclin-1 | Atg5 | LC3B | p62 |
|---|---|---|---|---|---|
| Sham | 1.01±0.08 | 1.00±0.20 | 1.00±0.17 | 1.00±0.14 | |
| BDL | 2.13±0.491) | 1.61±0.121) | 1.48±0.141) | 0.63±0.101) | |
| HXQJL-L | 7.74 | 1.11±0.082) | 1.09±0.182) | 0.92±0.162) | 0.83±0.182) |
| HXQJL-H | 15.48 | 1.03±0.182) | 0.85±0.202) | 0.83±0.252) | 0.98±0.142) |
| EX527 | 0.005 | 2.99±0.252) | 2.01±1.382) | 1.92±0.172) | 0.24±0.082) |
| EX527+HXQJL | 0.005+15.48 | 1.91±0.434) | 1.5±0.304) | 1.47±0.274) | 0.68±0.164) |
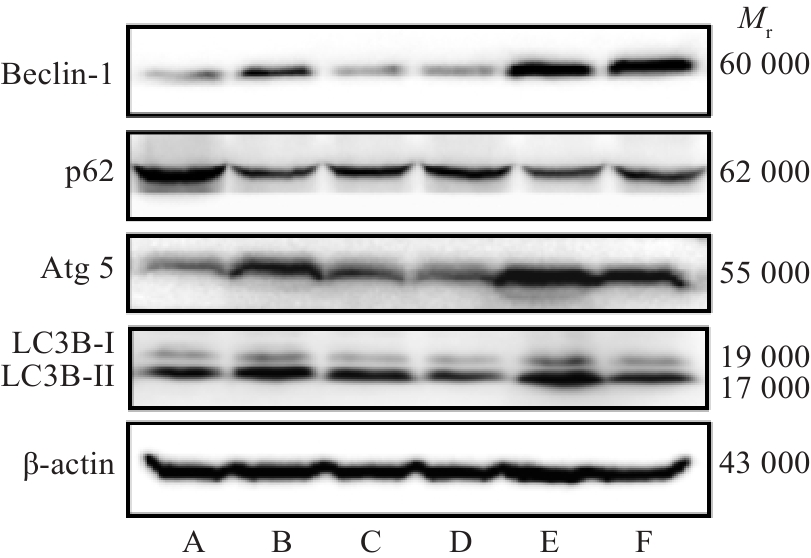
Fig. 4 Effect of HXQJL on expressions of Beclin-1, Atg5, LC3B and p62 in the liver tissues of rats with BDL-induced liver fibrosis. A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | Beclin-1 | Atg5 | p62 | LC3B-II/LC3B-I |
|---|---|---|---|---|---|
| Sham | 0.48±0.01 | 0.60±0.04 | 1.25±0.11 | 1.81±0.01 | |
| BDL | 0.77±0.021) | 0.99±0.071) | 0.43±0.031) | 2.35±0.121) | |
| HXQJL-L | 7.74 | 0.52±0.032) | 0.73±0.032) | 0.57±0.03 | 2.14±0.022) |
| HXQJL-H | 15.48 | 0.49±0.022) | 0.67±0.042) | 0.83±0.072) | 1.83±0.022) |
| EX527 | 0.005 | 1.15±0.112) | 1.27±0.082) | 0.25±0.052) | 2.66±0.032) |
| EX527+HXQJL | 0.005+15.48 | 0.78±0.104) | 0.86±0.034) | 0.47±0.034) | 2.56±0.094) |
Tab.8 Effect of HXQJL on protein level of Beclin-1, Atg5, LC3B and p62in the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Beclin-1 | Atg5 | p62 | LC3B-II/LC3B-I |
|---|---|---|---|---|---|
| Sham | 0.48±0.01 | 0.60±0.04 | 1.25±0.11 | 1.81±0.01 | |
| BDL | 0.77±0.021) | 0.99±0.071) | 0.43±0.031) | 2.35±0.121) | |
| HXQJL-L | 7.74 | 0.52±0.032) | 0.73±0.032) | 0.57±0.03 | 2.14±0.022) |
| HXQJL-H | 15.48 | 0.49±0.022) | 0.67±0.042) | 0.83±0.072) | 1.83±0.022) |
| EX527 | 0.005 | 1.15±0.112) | 1.27±0.082) | 0.25±0.052) | 2.66±0.032) |
| EX527+HXQJL | 0.005+15.48 | 0.78±0.104) | 0.86±0.034) | 0.47±0.034) | 2.56±0.094) |
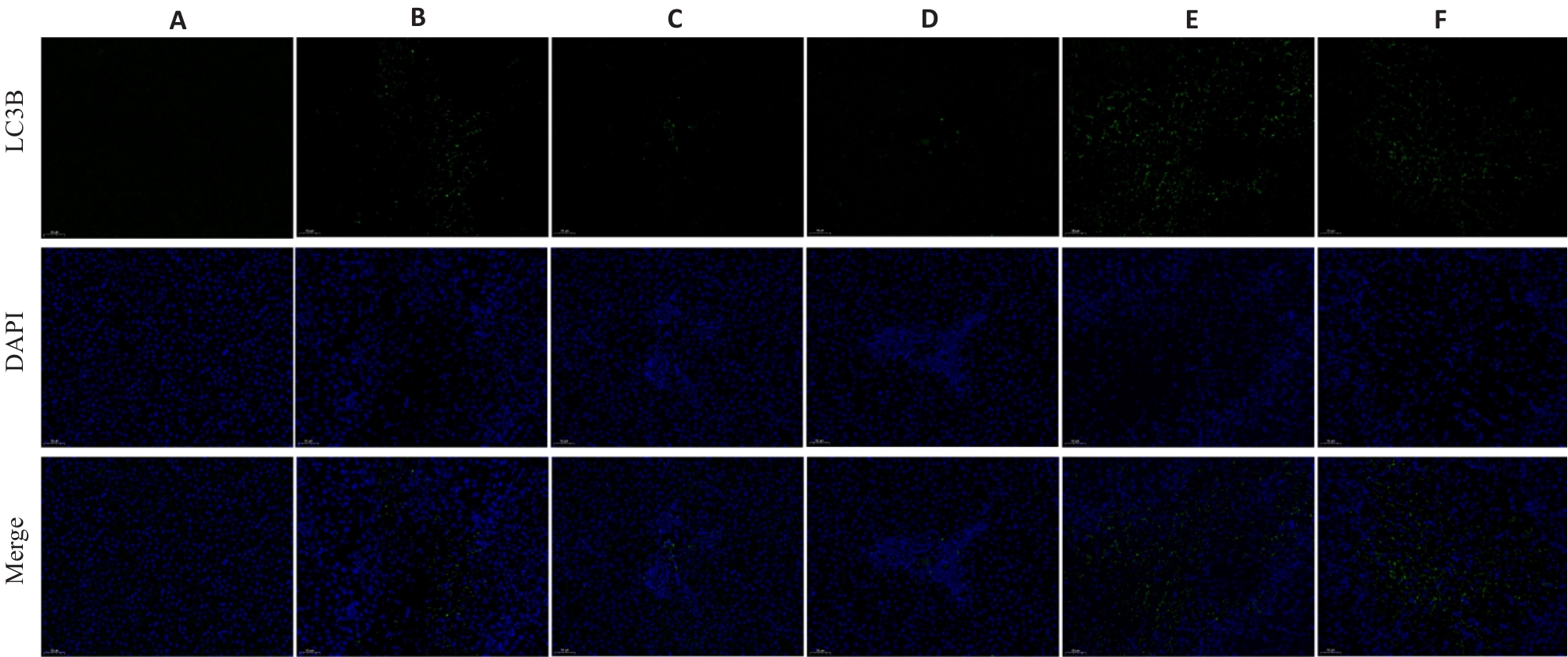
Fig.5 Effect of HXQJL on expressions of LC3B in the liver tissues of rats with BDL-induced liver fibrosis (×200). A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.00±0.14 | |
| BDL | 0.60±0.041) | |
| HXQJL-L | 7.74 | 0.99±0.112) |
| HXQJL-H | 15.48 | 0.99±0.082) |
| EX527 | 0.005 | 0.28±0.082) |
| EX527+HXQJL | 0.005+15.48 | 0.51±0.154) |
Tab.9 Effect of HXQJL on mRNA level ofSirt1in the liver tissues of the rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.00±0.14 | |
| BDL | 0.60±0.041) | |
| HXQJL-L | 7.74 | 0.99±0.112) |
| HXQJL-H | 15.48 | 0.99±0.082) |
| EX527 | 0.005 | 0.28±0.082) |
| EX527+HXQJL | 0.005+15.48 | 0.51±0.154) |
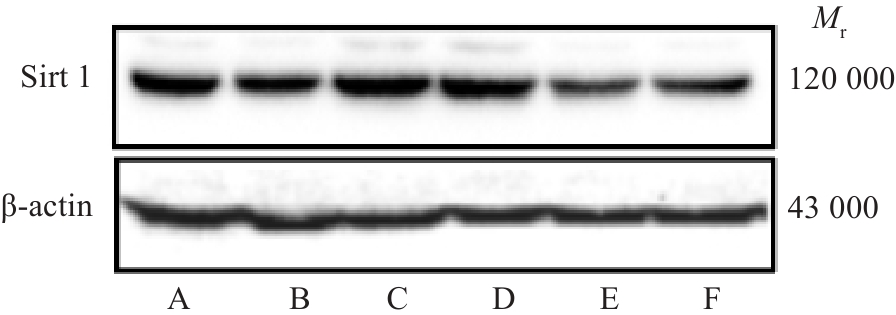
Fig.6 Effect of HXQJL on expression of Sirt 1 in liver tissues in rats with BDL-induced liver fibrosis. A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.14±0.10 | |
| BDL | 0.43±0.041) | |
| HXQJL-L | 7.74 | 1.09±0.042) |
| HXQJL-H | 15.48 | 1.13±0.102) |
| EX527 | 0.005 | 0.21±0.012) |
| EX527+HXQJL | 0.005+15.48 | 0.39±0.034) |
Tab.10 Effect of HXQJL on protein level of Sirt1in the liver tissues of the rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.14±0.10 | |
| BDL | 0.43±0.041) | |
| HXQJL-L | 7.74 | 1.09±0.042) |
| HXQJL-H | 15.48 | 1.13±0.102) |
| EX527 | 0.005 | 0.21±0.012) |
| EX527+HXQJL | 0.005+15.48 | 0.39±0.034) |
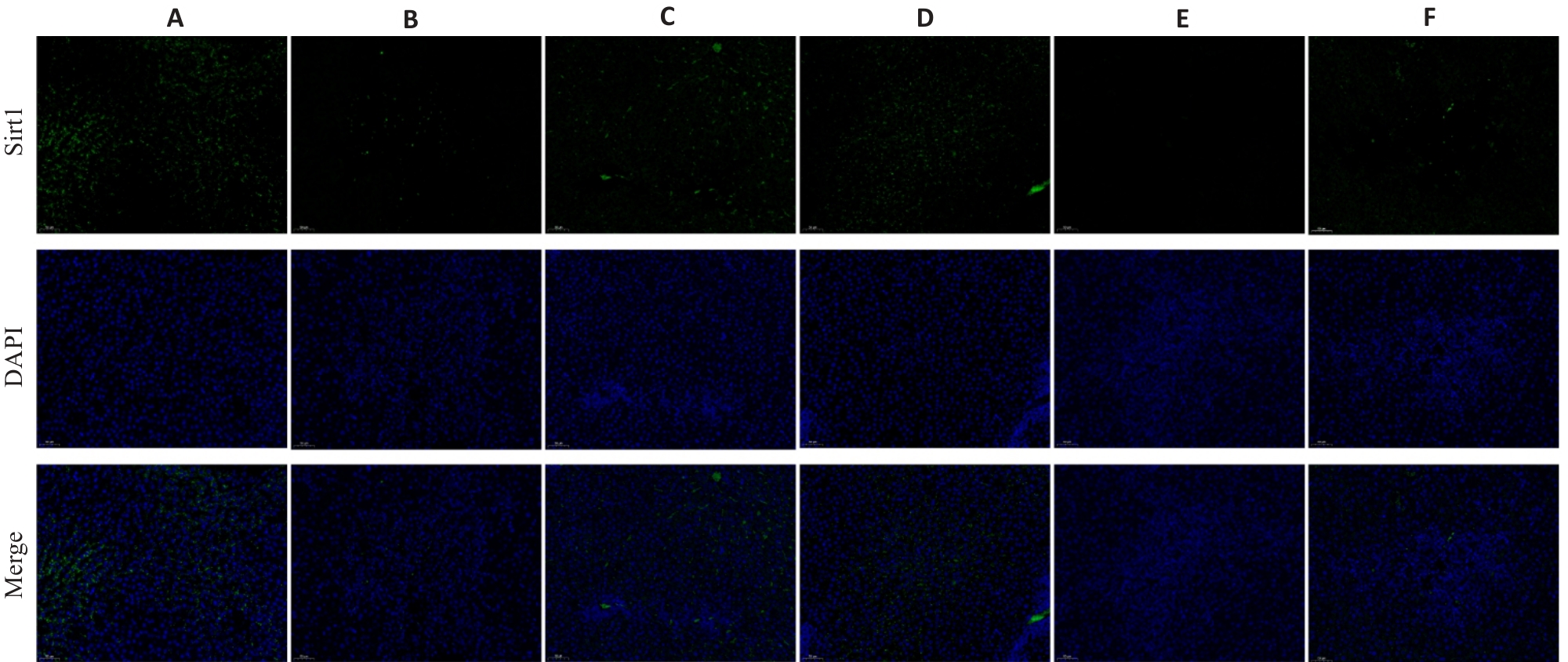
Fig.7 Effect of HXQJL on Sirt 1 expression in the liver tissues of the rats with BDL-induced liver fibrosis(×200). A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| [1] | Horn P, Tacke F. Metabolic reprogramming in liver fibrosis[J]. Cell Metab, 2024, 36(7): 1439-55. doi:10.1016/j.cmet.2024.05.003 |
| [2] | Filali-Mouncef Y, Hunter C, Roccio F, et al. The ménage à trois of autophagy, lipid droplets and liver disease[J]. Autophagy, 2022, 18(1): 50-72. doi:10.1080/15548627.2021.1895658 |
| [3] | Hernández-Gea V, Ghiassi-Nejad Z, Rozenfeld R, et al. Autophagy releases lipid that promotes fibrogenesis by activated hepatic stellate cells in mice and in human tissues[J]. Gastroenterology, 2012, 142(4): 938-46. doi:10.1053/j.gastro.2011.12.044 |
| [4] | Ma LQ, Fu RG, Duan ZY, et al. Sirt1 is essential for resveratrol enhancement of hypoxia-induced autophagy in the type 2 diabetic nephropathy rat[J]. Pathol Res Pract, 2016, 212(4): 310-8. doi:10.1016/j.prp.2016.02.001 |
| [5] | Gu LN, Tao XF, Xu YW, et al. Dioscin alleviates BDL- and DMN-induced hepatic fibrosis via Sirt1/Nrf2-mediated inhibition of p38 MAPK pathway[J]. Toxicol Appl Pharmacol, 2016, 292: 19-29. doi:10.1016/j.taap.2015.12.024 |
| [6] | 李锡晶, 沈洪昇, 王璐瑶, 等. 基于TGR5/NLRP3信号通路探讨活血清解灵对非酒精性脂肪性肝炎大鼠炎症反应的影响[J]. 中国现代应用药学, 2024, 41(10): 1324-31. doi:10.13748/j.cnki.issn1007-7693.20224076 |
| [7] | 无徐列明, 刘 平, 沈锡中, 等. 肝纤维化中西医结合诊疗指南(2019年版)[J]. 中国中西医结合杂志, 2019, 39(11): 1286-95. |
| [8] | 李婉华, 张桂贤, 沈洪昇, 等. 基于TGR5-NLRP3-Caspase1通路探讨活血清解灵对大鼠胆汁淤积性肝病的治疗作用[C]//第15届中国中西医结合学会基础理论专业委员会学术年会暨第二届广东省中西医结合学会转化医学专业委员会年会论文集. 广州, 2019: 47-8. |
| [9] | Cai YJ, Zheng Q, Sun R, et al. Recent progress in the study of Artemisiae Scopariae Herba (Yin Chen), a promising medicinal herb for liver diseases[J]. Biomed Pharmacother, 2020, 130: 110513. doi:10.1016/j.biopha.2020.110513 |
| [10] | Gijbels E, Pieters A, De Muynck K, et al. Rodent models of cholestatic liver disease: a practical guide for translational research[J]. Liver Int, 2021, 41(4): 656-82. doi:10.1111/liv.14800 |
| [11] | Ibrahim SH, Kamath BM, Loomes KM, et al. Cholestatic liver diseases of genetic etiology: Advances and controversies[J]. Hepatology, 2022, 75(6): 1627-46. doi:10.1002/hep.32437 |
| [12] | 王琳琳, 朱正望, 赵静涵, 等. 茵陈蒿汤对胆汁淤积性肝损伤的干预作用及调控机制[J/OL]. 中国实验方剂学杂志, 2025: 1-18. (2025-04-22). . |
| [13] | 向 杰, 邓敏贞, 李泽森, 等. 茵陈蒿汤方剂挥发油对肝纤维化小鼠的治疗作用及其机制[J]. 暨南大学学报: 自然科学与医学版, 2023, 44(6): 563-75. doi:10.11778/j.jdxb.20230125 |
| [14] | 朱晓东, 邸学士, 杨佳潞, 等. 基于网络药理学探讨茵陈蒿汤治疗肝纤维化的作用机制[J]. 中医药学报, 2021, 49(6): 29-36. doi:10.19664/j.cnki.1002-2392.210134 |
| [15] | 王 晶, 欧阳冰琛. 茵陈蒿汤防治肝脏疾病的药理作用及药动学研究进展[J]. 药物评价研究, 2021, 44(3): 628-37. doi:10.7501/j.issn.1674-6376.2021.03.026 |
| [16] | Cui XY, Zhang RH, Li YF, et al. Bie Jia Jian pill ameliorates BDL-induced cholestatic hepatic fibrosis in rats by regulating intestinal microbial composition and TMAO-mediated PI3K/AKT signaling pathway[J]. J Ethnopharmacol, 2025, 337(Pt 2): 118910. doi:10.1016/j.jep.2024.118910 |
| [17] | Xiao Z, Gao SQ, Li SS, et al. Taohong Siwu Decoction modulates glutathione metabolism to suppress hepatocyte ferroptosis and demonstrates anti-fibrotic effects in the liver[J]. J Ethnopharmacol, 2025, 350: 120025. doi:10.1016/j.jep.2025.120025 |
| [18] | Qu JR, Xue XY, Wang ZX, et al. Si-Wu-Tang attenuates liver fibrosis via regulating lncRNA H19-dependent pathways involving cytoskeleton remodeling and ECM deposition[J]. Chin J Nat Med, 2024, 22(1): 31-46. doi:10.1016/s1875-5364(24)60560-1 |
| [19] | Liu HM, Dong F, Li GQ, et al. Liuweiwuling tablets attenuate BDL-induced hepatic fibrosis via modulation of TGF-β/Smad and NF-κB signaling pathways[J]. J Ethnopharmacol, 2018, 210: 232-41. doi:10.1016/j.jep.2017.08.029 |
| [20] | Hou LS, Zhai XP, Zhang YW, et al. Targeted inhibition of autophagy in hepatic stellate cells by hydroxychloroquine: an effective therapeutic approach for the treatment of liver fibrosis[J]. Liver Int, 2024, 44(8): 1937-51. doi:10.1111/liv.15915 |
| [21] | Liu Y, Yao L, Liu YY, et al. Micheliolide alleviates hepatic fibrosis by inhibiting autophagy in hepatic stellate cells via the TrxR1/2-mediated ROS/MEK/ERK pathway[J]. Pharmaceuticals (Basel), 2025, 18(3): 287. doi:10.3390/ph18030287 |
| [22] | Wu LW, Zhang QH, Mo WH, et al. Quercetin prevents hepatic fibrosis by inhibiting hepatic stellate cell activation and reducing autophagy via the TGF-β1/Smads and PI3K/Akt pathways[J]. Sci Rep, 2017, 7: 9289. doi:10.1038/s41598-017-09673-5 |
| [23] | Ji J, Yu Q, Dai WQ, et al. Apigenin alleviates liver fibrosis by inhibiting hepatic stellate cell activation and autophagy via TGF-β 1/Smad3 and p38/PPAR α pathways[J]. PPAR Res, 2021, 2021: 6651839. doi:10.1155/2021/6651839 |
| [24] | Qin TT, Hasnat M, Wang ZW, et al. Geniposide alleviated bile acid-associated NLRP3 inflammasome activation by regulating SIRT1/FXR signaling in bile duct ligation-induced liver fibrosis[J]. Phytomedicine, 2023, 118: 154971. doi:10.1016/j.phymed.2023.154971 |
| [25] | Xia Y, Luo QH, Gao Q, et al. SIRT1 activation ameliorates Rhesus monkey liver fibrosis by inhibiting the TGF‑β/smad signaling pathway[J]. Chem Biol Interact, 2024, 394: 110979. doi:10.1016/j.cbi.2024.110979 |
| [26] | Zheng W, Guan F, Xu G, et al. FAT10 silencing prevents liver fibrosis through regulating SIRT1 expression in hepatic stellate cells[J]. Int J Med Sci, 2023, 20(4): 557-65. doi:10.7150/ijms.77367 |
| [27] | Lu HJ, Zhang RH, Zhang SK, et al. HSC-derived exosomal miR-199a-5p promotes HSC activation and hepatocyte EMT via targeting SIRT1 in hepatic fibrosis[J]. Int Immunopharmacol, 2023, 124(Pt B): 111002. doi:10.1016/j.intimp.2023.111002 |
| [28] | Yang L, Ao QF, Zhong QM, et al. SIRT1/IGFBPrP1/TGF β1 axis involved in cucurbitacin B ameliorating concanavalin A-induced mice liver fibrosis[J]. Basic Clin Pharmacol Toxicol, 2020, 127(5): 371-9. doi:10.1111/bcpt.13446 |
| [29] | Zhu J, Wang RW, Xu T, et al. Salvianolic acid a attenuates endoplasmic reticulum stress and protects against cholestasis-induced liver fibrosis via the SIRT1/HSF1 pathway[J]. Front Pharmacol, 2018, 9: 1277. doi:10.3389/fphar.2018.01277 |
| [30] | Patra S, Praharaj PP, Singh A, et al. Targeting SIRT1-regulated autophagic cell death as a novel therapeutic avenue for cancer prevention[J]. Drug Discov Today, 2023, 28(9): 103692. doi:10.1016/j.drudis.2023.103692 |
| [31] | Mei ZG, Huang YG, Feng ZT, et al. Electroacupuncture ameliorates cerebral ischemia/reperfusion injury by suppressing autophagy via the SIRT1-FOXO1 signaling pathway[J]. Aging (Albany NY), 2020, 12(13): 13187-205. doi:10.18632/aging.103420 |
| [1] | Tong QIAO, Lin YIN, Keni ZHANG, Minzhu NIU, Ju HUANG, Zhijun Geng, Jing LI, Jianguo HU. Poricoic acid A alleviates dextran sulfate sodium-induced colitis in mice by regulating AMPK/mTOR-mediated autophagy and inhibiting intestinal epithelial cell apoptosis [J]. Journal of Southern Medical University, 2026, 46(1): 131-140. |
| [2] | Weiyi LI, Lu JIANG, Zongxing ZHANG, Dan CHEN, Zhuoma BAO, Li HUANG, Lin YUAN. Qianggu Kangshu Formula attenuates osteoclast differentiation in rheumatoid arthritis by inhibiting the HIF-1α/BNIP3 autophagy signaling pathway [J]. Journal of Southern Medical University, 2025, 45(7): 1389-1396. |
| [3] | Xinheng WANG, Xiaohan SHAO, Tongtong LI, Lu ZHANG, Qinjun YANG, Weidong YE, Jiabing TONG, Zegeng LI, Xiangming FANG. Pingchuanning Formula suppresses airway inflammation in a rat model of asthmatic cold syndrome by regulating the HMGB1/Beclin-1 axis-mediated autophagy [J]. Journal of Southern Medical University, 2025, 45(6): 1153-1162. |
| [4] | Liping GUAN, Yan YAN, Xinyi LU, Zhifeng LI, Hui GAO, Dong CAO, Chenxi HOU, Jingyu ZENG, Xinyi LI, Yang ZHAO, Junjie WANG, Huilong FANG. Compound Centella asiatica formula alleviates Schistosoma japonicum-induced liver fibrosis in mice by inhibiting the inflammation-fibrosis cascade via regulating the TLR4/MyD88 pathway [J]. Journal of Southern Medical University, 2025, 45(6): 1307-1316. |
| [5] | Lu ZHANG, Huanzhang DING, Haoran XU, Ke CHEN, Bowen XU, Qinjun YANG, Di WU, Jiabing TONG, Zegeng LI. Shenqi Buzhong Formula ameliorates mitochondrial dysfunction in a rat model of chronic obstructive pulmonary disease by activating the AMPK/SIRT1/PGC-1α pathway [J]. Journal of Southern Medical University, 2025, 45(5): 969-976. |
| [6] | Yanyan DONG, Kejing ZHANG, Jun CHU, Quangen CHU. Didang Decoction-medicated serum enhances autophagy in high glucose-induced rat glomerular endothelial cells via the PI3K/Akt/mTOR signaling pathway [J]. Journal of Southern Medical University, 2025, 45(3): 461-469. |
| [7] | Ming LIAO, Wenhua ZHONG, Ran ZHANG, Juan LIANG, Wentaorui XU, Wenjun WAN, Chao LI Shu WU. Protein C activator derived from snake venom protects human umbilical vein endothelial cells against hypoxia-reoxygenation injury by suppressing ROS via upregulating HIF-1α and BNIP3 [J]. Journal of Southern Medical University, 2025, 45(3): 614-621. |
| [8] | Daiping HUA, Qiaoyu XUAN, Lanting SUN, Qingsheng YU, Qin WANG, Tao WANG, Qiyan MA, Wenming YANG, Han WANG. LncRNA Meg3 expression level is negatively correlated with liver fibrosis severity in patients with Wilson disease [J]. Journal of Southern Medical University, 2025, 45(11): 2365-2374. |
| [9] | Ming YI, Ye LUO, Lu WU, Zeheng WU, Cuiping JIANG, Shiyu CHEN, Xiao KE. Astragaloside IV alleviates D-GAL-induced endothelial cell senescence by promoting mitochondrial autophagy via inhibiting the PINK1/Parkin pathway [J]. Journal of Southern Medical University, 2025, 45(11): 2427-2436. |
| [10] | Simeng LI, Jianning CHEN, Siman SHEN, Wanglong LIU, Lili YU, Liangqing ZHANG. Salvianolic acid B promotes mitochondrial homeostasis and improves cardiac function in mice with ischemia-reperfusion injury by inhibiting Sirt1 protein degradation [J]. Journal of Southern Medical University, 2025, 45(10): 2062-2070. |
| [11] | Kelei GUO, Yingli LI, Chenguang XUAN, Zijun HOU, Songshan YE, Linyun LI, Liping CHEN, Li HAN, Hua BIAN. Yiqi Yangyin Huazhuo Tongluo Formula alleviates diabetic podocyte injury by regulating miR-21a-5p/FoxO1/PINK1-mediated mitochondrial autophagy [J]. Journal of Southern Medical University, 2025, 45(1): 27-34. |
| [12] | Junping ZHAN, Shuo HUANG, Qingliang MENG, Wei FAN, Huimin GU, Jiakang CUI, Huilian WANG. Buyang Huanwu Decoction reduces mitochondrial autophagy in rheumatoid arthritis synovial fibroblasts in hypoxic culture by inhibiting the BNIP3-PI3K/Akt pathway [J]. Journal of Southern Medical University, 2025, 45(1): 35-42. |
| [13] | Zhiliang CHEN, Yonggang YANG, Xia HUANG, Yan CHENG, Yuan QU, Qiqi HENG, Yujia FU, Kewei LI, Ning GU. Differential expressions of exosomal miRNAs in patients with chronic heart failure and hyperuricemia: diagnostic values of miR-27a-5p and miR-139-3p [J]. Journal of Southern Medical University, 2025, 45(1): 43-51. |
| [14] | Xingmei CHEN, Qinwen LIU, Yi LI, Xiaoyu ZHONG, Qiling FAN, Ke MA, Liuting LUO, Daogang GUAN, Zhibo ZHU. Analysis of core functional components in Yinchenhao Decoction and their pathways for treating liver fibrosis [J]. Journal of Southern Medical University, 2024, 44(8): 1508-1517. |
| [15] | Yao CHENG, Yuanying WANG, Feiyang YAO, Pan HU, Mingxian CHEN, Ning WU. Baicalin suppresses type 2 dengue virus-induced autophagy of human umbilical vein endothelial cells by inhibiting the PI3K/AKT pathway [J]. Journal of Southern Medical University, 2024, 44(7): 1272-1283. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||