Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (2): 247-258.doi: 10.12122/j.issn.1673-4254.2026.02.02
Jiayao YANG1,2( ), Yulian He1,2, Yanlei GUO1, Fanghong SHANG1,2,3, Lei HUA1,2, Yong YANG1,2,4, Xiaomei ZHANG1,2,3,4(
), Yulian He1,2, Yanlei GUO1, Fanghong SHANG1,2,3, Lei HUA1,2, Yong YANG1,2,4, Xiaomei ZHANG1,2,3,4( ), Jiangping WEI1,2,3(
), Jiangping WEI1,2,3( )
)
Received:2025-08-10
Online:2026-02-20
Published:2026-03-10
Contact:
Xiaomei ZHANG, Jiangping WEI
E-mail:yangjiayao959@163.com;ZXM761@163.com;sichuanwjp@163.com
Supported by:Jiayao YANG, Yulian He, Yanlei GUO, Fanghong SHANG, Lei HUA, Yong YANG, Xiaomei ZHANG, Jiangping WEI. Radix codonopsis combined with Poria improves cognitive impairment in rats with unilateral common carotid artery ligation by regulating the ERα/PI3K/Akt signaling pathway[J]. Journal of Southern Medical University, 2026, 46(2): 247-258.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.02.02
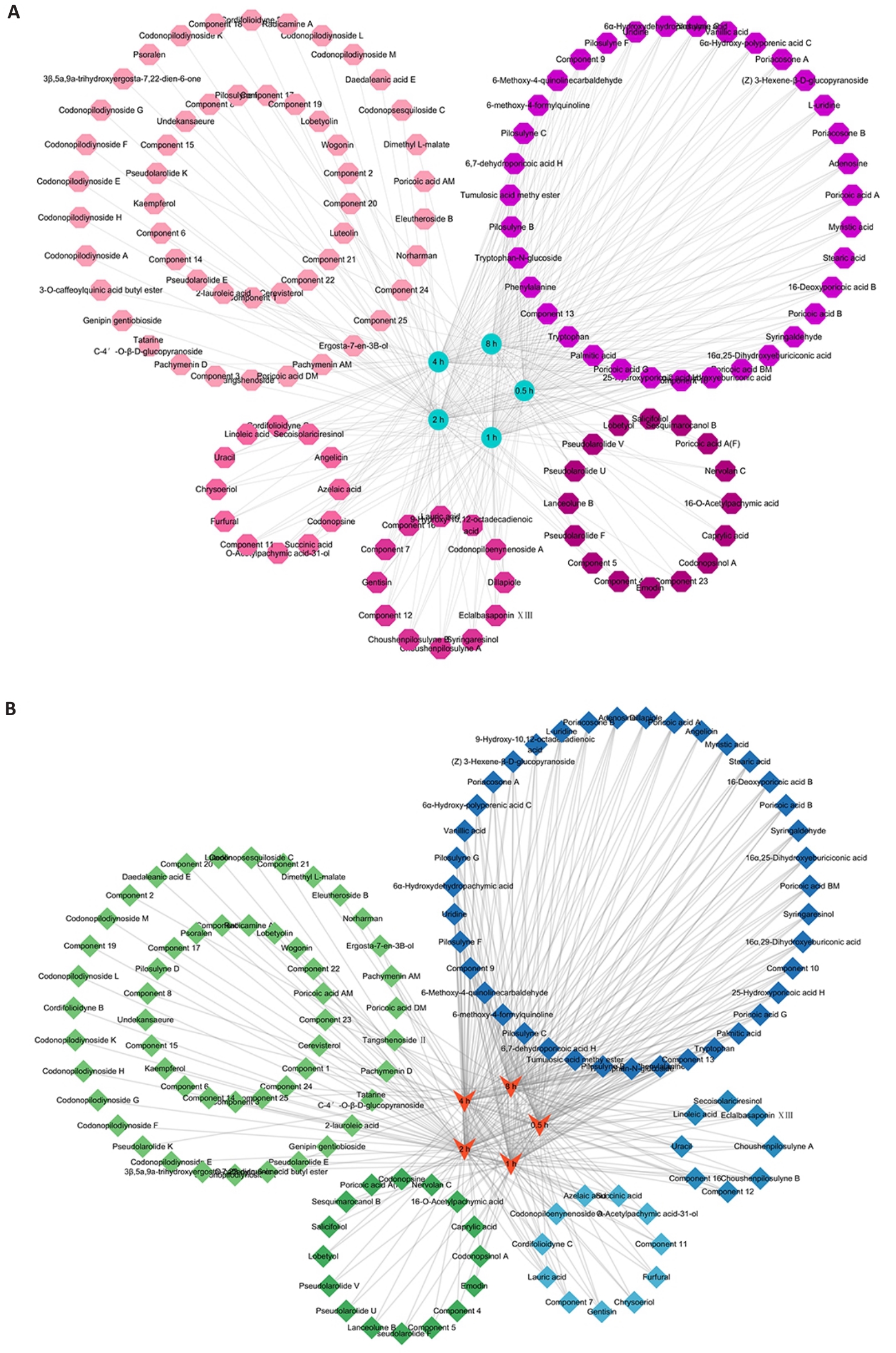
Fig.1 UPLC-Q-TOF-MS/MS for analyzing the components of Radix codonopsis combined with Poria (CRP) in blood and brain of medicated rats at different time points. A: Network diagram depicting the distribution relationships of CRP components in the blood at different time points. B: Network diagram of the distribution relationships of CRP components in the brain at different time points. The circles of the same color denote components detected at the same time point.
| NO. | Retain time (min) | Ion peak assignment | Theoretical value mass (m/z) | Measured value mass (m/z) | Deviation (ppm) | Chemical formula | Name |
|---|---|---|---|---|---|---|---|
| 1 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 6,7-dehydroporicoic acid H |
| 2 | 6.96 | [M-H]- | 466.30831 | 465.30019 | -1.8 | C30H42O4 | daedaleanic acid E |
| 3 | 7.98 | [M+H]+ | 444.32396 | 445.33071 | -1.2 | C28H44O4 | 3β,5a,9a-trihydroxyergosta-7,22-dien-6-one |
| 4 | 1.87 | [M+H]+ | 162.05282 | 163.06008 | -0.1 | C6H10O5 | Dimethyl L-malate |
| 5 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | L-uridine |
| 6 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid A |
| 7 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poricoic acid B |
| 8 | 9.91 | [M-H]- | 200.17763 | 199.1705 | 0.7 | C12H24O2 | lauric acid |
| 9 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | palmitic acid |
| 10 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Poricoic acid AM |
| 11 | 6.68 | [M-H]- | 584.40769 | 583.40041 | 0 | C36H56O6 | Tumulosic acid methy ester |
| 12 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | Poricoic acid A(F) |
| 13 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid BM |
| 14 | 6.4 | [M-H]- | 486.33453 | 485.32735 | 0.2 | C30H46O5 | Poricoic acid G |
| 15 | 5.34 | [M-H]- | 470.33961 | 469.33272 | 0.8 | C30H46O4 | 3β,16α-Dihydroxy-lanosta-7,9(11),24-trien-21-oic acid |
| 16 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 25-Hydroxyporicoic acid H |
| 17 | 6.62 | [M-H]- | 468.32396 | 467.31682 | 0.3 | C30H44O4 | 16-Deoxyporicoic acid B |
| 18 | 11.03 | [M+H]+ | 528.34509 | 529.3529 | 1 | C32H48O6 | Poricoic acid DM |
| 19 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone A |
| 20 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone B |
| 21 | 6.11 | [M-H]- | 496.31887 | 495.31138 | -0.4 | C31H44O5 | 6α-Hydroxy-polyporenic acid C |
| 22 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,25-Dihydroxyeburiciconic acid |
| 23 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,29-Dihydroxyeburiconic acid |
| 24 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Pachymenin D |
| 25 | 8.09 | [M+H]+ | 540.34509 | 541.35241 | 0.1 | C33H48O6 | 6α-Hydroxydehydropachymic acid |
| 26 | 5.28 | [M-H]-/[M+H]+ | 204.08988 | 203.08279 | 1 | C11H12N2O2 | tryptophan |
| 27 | 2.73 | [M+H]+ | 112.02728 | 113.03426 | -2.6 | C4H4N2O2 | uracil |
| 28 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | uridine |
| 29 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-Methoxy-4-quinolinecarbaldehyde |
| 30 | 4.32 | [M+H]+ | 165.07898 | 166.08625 | 0 | C9H11NO2 | phenylalanine(L-Phenylalanine) |
| 31 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | tangshenoside Ⅱ |
| 32 | 8.14 | [M-H]- | 262.12051 | 261.11355 | 1.2 | C15H18O4 | nervolan C |
| 33 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | Eleutheroside B(syringin) |
| 34 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | dillapiole |
| 35 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | 1-allyl-2,6-dimethoxy-3,4-methylenedioxybenzene |
| 36 | 11.43 | [M+H]+ | 418.16277 | 419.16949 | -1.3 | C22H26O8 | syringaresinol |
| 37 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | secoisolariciresinol |
| 38 | 5.88 | [M-H]- | 498.29814 | 497.29218 | 2.6 | C30H42O6 | pseudolarolide E |
| 39 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | apigenin |
| 40 | 11.9 | [M-H]-/[M+H]+ | 280.24023 | 279.23241 | -1.9 | C18H32O2 | linoleic acid |
| 41 | 11.34 | [M-H]- | 228.20893 | 227.20169 | 0.2 | C14H28O2 | myristic acid |
| 42 | 8.33 | [M-H]- | 284.27153 | 283.26384 | -1.5 | C18H36O2 | stearic acid |
| 43 | 6.52 | [M-H]- | 188.10486 | 187.09787 | 1.5 | C9H16O4 | azelaic acid |
| 44 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | Palmitic Acid (n-hexadecanoic acid) |
| 45 | 5.8 | [M-H]- | 182.05791 | 181.05037 | -1.5 | C9H10O4 | syringaldehyde |
| 46 | 1.9 | [M+H]+ | 96.02113 | 97.02847 | 0.7 | C5H4O2 | Furfural(2-furaldehyde) |
| 47 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | emodin |
| 48 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside F |
| 49 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside G |
| 50 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside K |
| 51 | 7.75 | [M-H]-/[M+H]+ | 250.12051 | 249.11329 | 0.2 | C14H18O4 | pilosulyne B |
| 52 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne F |
| 53 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne G |
| 54 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | pseudolarolide U |
| 55 | 1.97 | [M+H]+ | 255.11067 | 256.11797 | 0.1 | C12H17NO5 | radicamine A |
| 56 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-methoxy-4-formylquinoline |
| 57 | 5.91 | [M-H]- | 486.18496 | 485.1766 | -2.2 | C21H30N2O11 | tatarine C-4′-O-β-D-glucopyranoside |
| 58 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | 1,6-Hexanediol-3,4-di (4-hydroxy-3-methoxyphenyl) |
| 59 | 5.9 | [M-H]- | 540.23593 | 539.22752 | -2.1 | C30H36O9 | sesquimarocanol B |
Tab.1 Components of CRP in the blood and brain in CRP-treated UCCA rats
| NO. | Retain time (min) | Ion peak assignment | Theoretical value mass (m/z) | Measured value mass (m/z) | Deviation (ppm) | Chemical formula | Name |
|---|---|---|---|---|---|---|---|
| 1 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 6,7-dehydroporicoic acid H |
| 2 | 6.96 | [M-H]- | 466.30831 | 465.30019 | -1.8 | C30H42O4 | daedaleanic acid E |
| 3 | 7.98 | [M+H]+ | 444.32396 | 445.33071 | -1.2 | C28H44O4 | 3β,5a,9a-trihydroxyergosta-7,22-dien-6-one |
| 4 | 1.87 | [M+H]+ | 162.05282 | 163.06008 | -0.1 | C6H10O5 | Dimethyl L-malate |
| 5 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | L-uridine |
| 6 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid A |
| 7 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poricoic acid B |
| 8 | 9.91 | [M-H]- | 200.17763 | 199.1705 | 0.7 | C12H24O2 | lauric acid |
| 9 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | palmitic acid |
| 10 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Poricoic acid AM |
| 11 | 6.68 | [M-H]- | 584.40769 | 583.40041 | 0 | C36H56O6 | Tumulosic acid methy ester |
| 12 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | Poricoic acid A(F) |
| 13 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid BM |
| 14 | 6.4 | [M-H]- | 486.33453 | 485.32735 | 0.2 | C30H46O5 | Poricoic acid G |
| 15 | 5.34 | [M-H]- | 470.33961 | 469.33272 | 0.8 | C30H46O4 | 3β,16α-Dihydroxy-lanosta-7,9(11),24-trien-21-oic acid |
| 16 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 25-Hydroxyporicoic acid H |
| 17 | 6.62 | [M-H]- | 468.32396 | 467.31682 | 0.3 | C30H44O4 | 16-Deoxyporicoic acid B |
| 18 | 11.03 | [M+H]+ | 528.34509 | 529.3529 | 1 | C32H48O6 | Poricoic acid DM |
| 19 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone A |
| 20 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone B |
| 21 | 6.11 | [M-H]- | 496.31887 | 495.31138 | -0.4 | C31H44O5 | 6α-Hydroxy-polyporenic acid C |
| 22 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,25-Dihydroxyeburiciconic acid |
| 23 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,29-Dihydroxyeburiconic acid |
| 24 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Pachymenin D |
| 25 | 8.09 | [M+H]+ | 540.34509 | 541.35241 | 0.1 | C33H48O6 | 6α-Hydroxydehydropachymic acid |
| 26 | 5.28 | [M-H]-/[M+H]+ | 204.08988 | 203.08279 | 1 | C11H12N2O2 | tryptophan |
| 27 | 2.73 | [M+H]+ | 112.02728 | 113.03426 | -2.6 | C4H4N2O2 | uracil |
| 28 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | uridine |
| 29 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-Methoxy-4-quinolinecarbaldehyde |
| 30 | 4.32 | [M+H]+ | 165.07898 | 166.08625 | 0 | C9H11NO2 | phenylalanine(L-Phenylalanine) |
| 31 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | tangshenoside Ⅱ |
| 32 | 8.14 | [M-H]- | 262.12051 | 261.11355 | 1.2 | C15H18O4 | nervolan C |
| 33 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | Eleutheroside B(syringin) |
| 34 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | dillapiole |
| 35 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | 1-allyl-2,6-dimethoxy-3,4-methylenedioxybenzene |
| 36 | 11.43 | [M+H]+ | 418.16277 | 419.16949 | -1.3 | C22H26O8 | syringaresinol |
| 37 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | secoisolariciresinol |
| 38 | 5.88 | [M-H]- | 498.29814 | 497.29218 | 2.6 | C30H42O6 | pseudolarolide E |
| 39 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | apigenin |
| 40 | 11.9 | [M-H]-/[M+H]+ | 280.24023 | 279.23241 | -1.9 | C18H32O2 | linoleic acid |
| 41 | 11.34 | [M-H]- | 228.20893 | 227.20169 | 0.2 | C14H28O2 | myristic acid |
| 42 | 8.33 | [M-H]- | 284.27153 | 283.26384 | -1.5 | C18H36O2 | stearic acid |
| 43 | 6.52 | [M-H]- | 188.10486 | 187.09787 | 1.5 | C9H16O4 | azelaic acid |
| 44 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | Palmitic Acid (n-hexadecanoic acid) |
| 45 | 5.8 | [M-H]- | 182.05791 | 181.05037 | -1.5 | C9H10O4 | syringaldehyde |
| 46 | 1.9 | [M+H]+ | 96.02113 | 97.02847 | 0.7 | C5H4O2 | Furfural(2-furaldehyde) |
| 47 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | emodin |
| 48 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside F |
| 49 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside G |
| 50 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside K |
| 51 | 7.75 | [M-H]-/[M+H]+ | 250.12051 | 249.11329 | 0.2 | C14H18O4 | pilosulyne B |
| 52 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne F |
| 53 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne G |
| 54 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | pseudolarolide U |
| 55 | 1.97 | [M+H]+ | 255.11067 | 256.11797 | 0.1 | C12H17NO5 | radicamine A |
| 56 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-methoxy-4-formylquinoline |
| 57 | 5.91 | [M-H]- | 486.18496 | 485.1766 | -2.2 | C21H30N2O11 | tatarine C-4′-O-β-D-glucopyranoside |
| 58 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | 1,6-Hexanediol-3,4-di (4-hydroxy-3-methoxyphenyl) |
| 59 | 5.9 | [M-H]- | 540.23593 | 539.22752 | -2.1 | C30H36O9 | sesquimarocanol B |
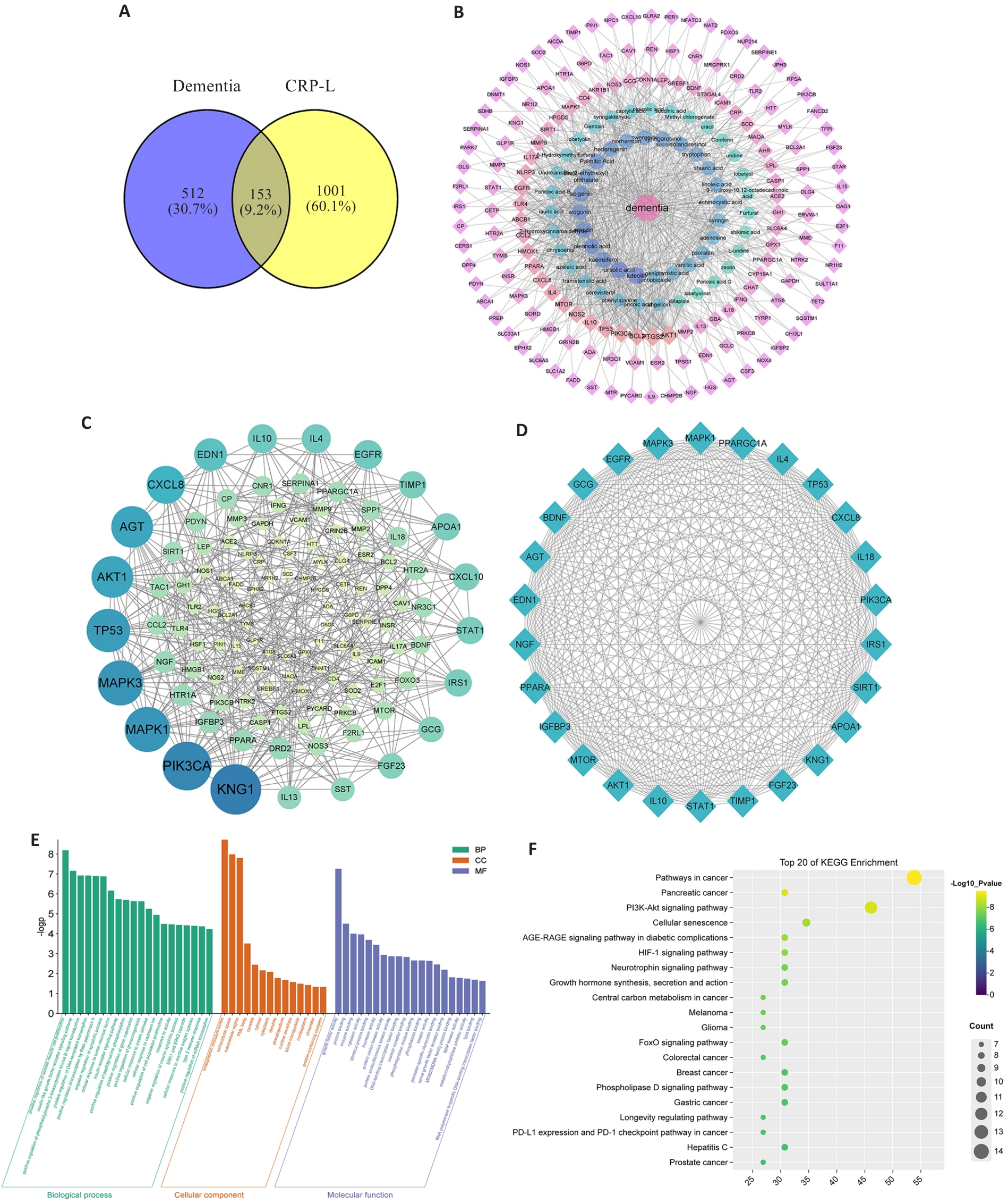
Fig.2 Network pharmacology analysis of the components of CRP in the blood and brain of CRP-treated UCCA rats. A: Venn diagram of the targets of the components of CRP in rat blood and brain and the targets of dementia. B: Disease-compounds-targets network. C: PPI network of the intersection targets for treatment of dementia by CRP components in the blood and brain. D: Network diagram of the key targets. E: GO enrichment analysis of the key targets. F: KEGG enrichment analysis of the key targets.
| Group | Dose (g/kg) | First day (s) | Second day (s) | Third day (s) | Fourth day (s) | Fifth day (s) |
|---|---|---|---|---|---|---|
| Sham | - | 106.00±31.81 | 70.36±22.25 | 54.49±17.23^^ | 22.33±7.06^^^## | 16.39±5.18^^^## |
| Model | - | 111.55±35.28 | 73.46±23.23 | 57.63±18.23 | 70.05±22.15** | 57.67±18.24^^** |
| Aricept | 1.17×10-3 | 93.94±29.71 | 78.36±24.80 | 43.53±13.77^^ | 29.67±9.38^^^# | 26.05±8.24^^^# |
| CRP-L | 3.5 | 92.78±29.34 | 85.58±27.02 | 59.17±18.71 | 33.98±10.75^^^# | 23.75±7.51^^^## |
| CRP-H | 7 | 98.54±31.16 | 98.89±31.27 | 69.63±22.02 | 50.43±15.95^^^ | 22.81±7.21^^^## |
Tab.2 Effects of CRP on escape latency of UCCA rats in the hidden platform (Mean±SD)
| Group | Dose (g/kg) | First day (s) | Second day (s) | Third day (s) | Fourth day (s) | Fifth day (s) |
|---|---|---|---|---|---|---|
| Sham | - | 106.00±31.81 | 70.36±22.25 | 54.49±17.23^^ | 22.33±7.06^^^## | 16.39±5.18^^^## |
| Model | - | 111.55±35.28 | 73.46±23.23 | 57.63±18.23 | 70.05±22.15** | 57.67±18.24^^** |
| Aricept | 1.17×10-3 | 93.94±29.71 | 78.36±24.80 | 43.53±13.77^^ | 29.67±9.38^^^# | 26.05±8.24^^^# |
| CRP-L | 3.5 | 92.78±29.34 | 85.58±27.02 | 59.17±18.71 | 33.98±10.75^^^# | 23.75±7.51^^^## |
| CRP-H | 7 | 98.54±31.16 | 98.89±31.27 | 69.63±22.02 | 50.43±15.95^^^ | 22.81±7.21^^^## |
| Group | Dose (g/kg) | Platform quadrant cumulative Time (s) | Times of platform crossing time (time) | Average swimming speed (cm/s) |
|---|---|---|---|---|
| Sham | - | 32.37±10.24# | 4.22±1.34 | 14.74±4.66 |
| Model | - | 21.33±6.75* | 2.50±0.79 | 12.44±3.94 |
| Aricept | 1.17×10-3 | 34.68±10.97## | 4.20±1.33 | 13.69±4.33 |
| CRP-L | 3.5 | 33.94±10.73# | 5.80±1.83## | 13.58±4.29 |
| CRP-H | 7 | 29.22±9.24 | 3.20±1.01 | 13.87±4.39 |
Tab.3 Effects of CRP on spatial exploration of UCCA rats (Mean±SD)
| Group | Dose (g/kg) | Platform quadrant cumulative Time (s) | Times of platform crossing time (time) | Average swimming speed (cm/s) |
|---|---|---|---|---|
| Sham | - | 32.37±10.24# | 4.22±1.34 | 14.74±4.66 |
| Model | - | 21.33±6.75* | 2.50±0.79 | 12.44±3.94 |
| Aricept | 1.17×10-3 | 34.68±10.97## | 4.20±1.33 | 13.69±4.33 |
| CRP-L | 3.5 | 33.94±10.73# | 5.80±1.83## | 13.58±4.29 |
| CRP-H | 7 | 29.22±9.24 | 3.20±1.01 | 13.87±4.39 |
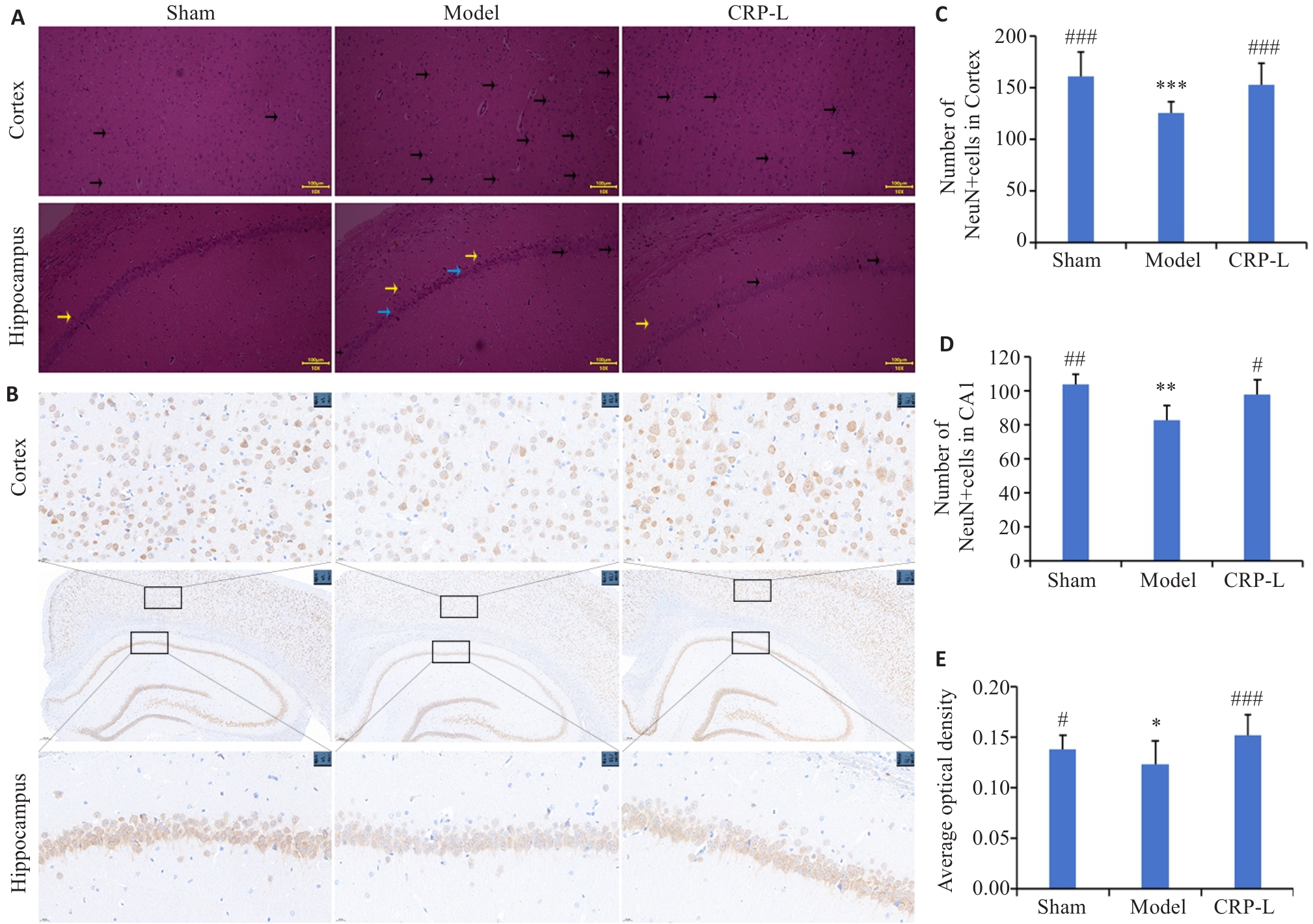
Fig.3 Effects of CRP on pathological morphology and number of neurons in the hippocampus and cortex of UCCA rats. A: HE staining (Black arrow indicate the cells undergoing degeneration or death, yellow arrows indicate the nerve cells in disordered alignment, and blue arrows indicate enlargement of intercellular spaces and decreased cell layers; original magnification: ×10). B: NeuN staining (×5; ×40). C: Number of NeuN+ cells in the cortex. D: Number of NeuN+ cells in the CA1 area. E: Average optical density. Data are presented as Mean±SD (n=6). *P<0.05, **P<0.01, ***P<0.001 vs Sham; #P<0.05, ##P<0.01, ###P<0.001 vs Model.
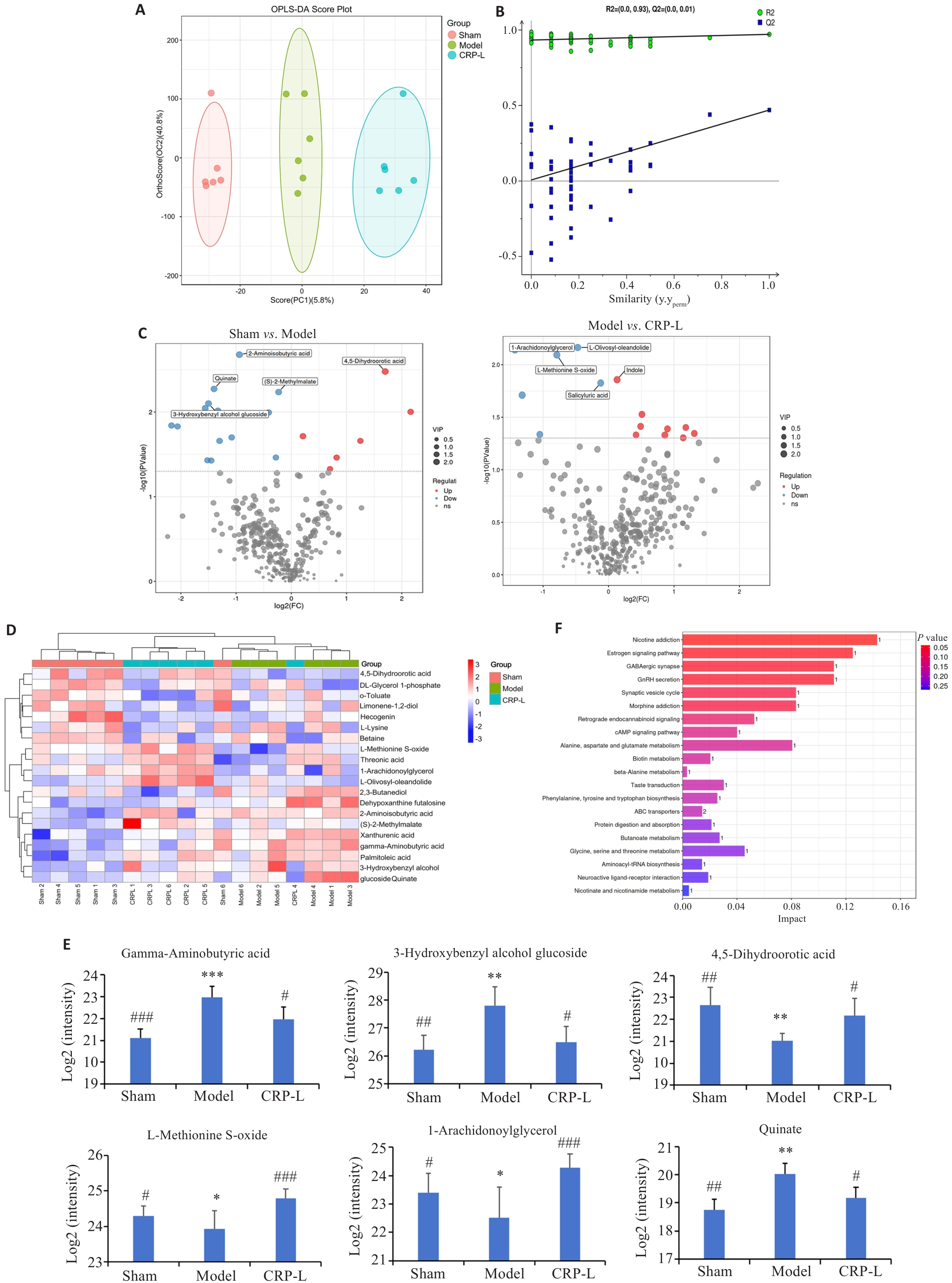
Fig.4 Effects of CRP on brain metabolomics in UCCA rats. A: OPLS-DA score plot. B: Permutation test. C: Volcano plot. D: Differential molecule heat map. E: Relative contents of representative differential molecules. F: KEGG enrichment analysis. Data are presented as Mean±SD (n=6). *P<0.05, **P<0.01, ***P<0.001 vs Sham, #P<0.05, ##P<0.01, ###P<0.001 vs Model.
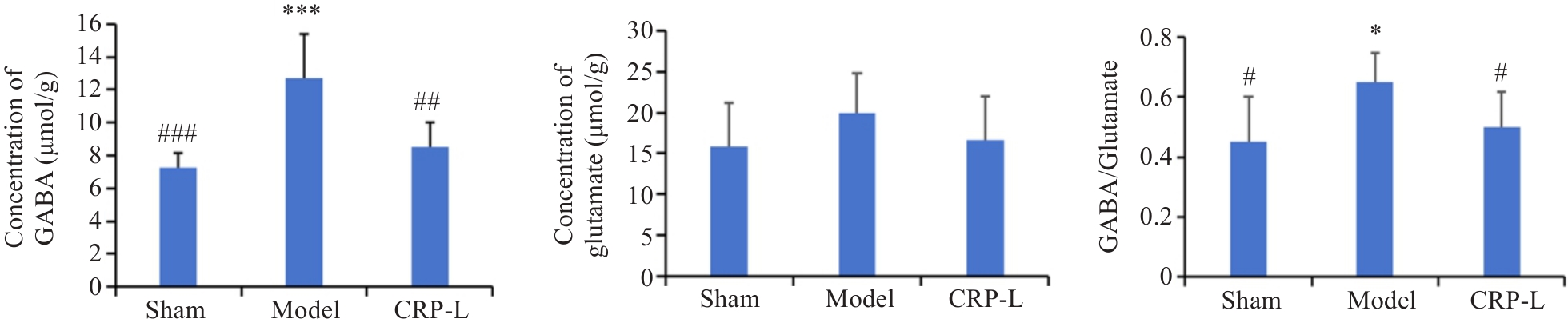
Fig.5 Effect of CRP on GABA and glutamate contents in the brain of UCCA rats. Data are presented as Mean±SD (n=6). *P<0.05, ***P<0.001 vs Sham; #P<0.05, ##P<0.01, ###P<0.001 vs Model.
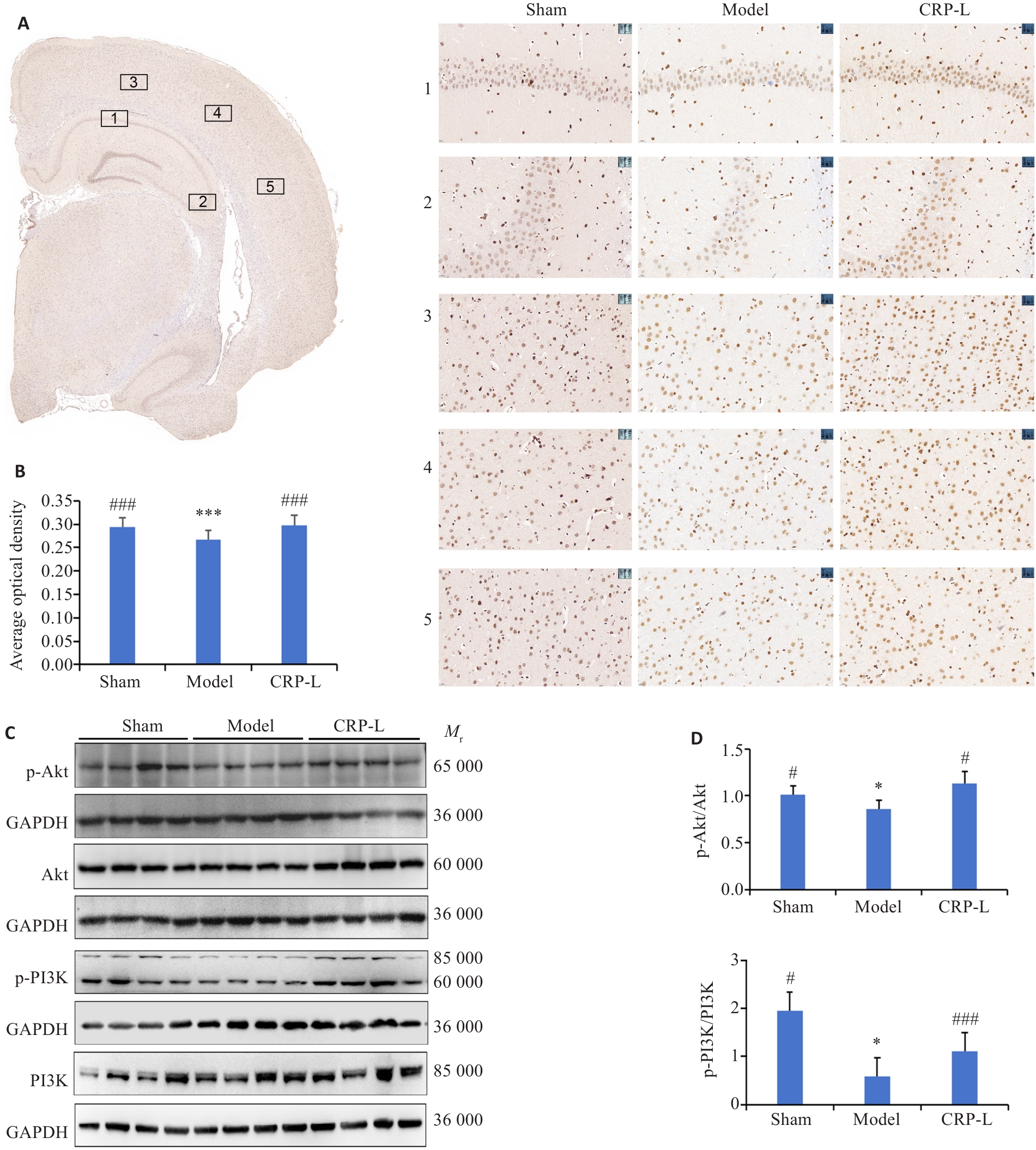
Fig.6 Effect of CRP on the ERα/PI3K/Akt signaling pathway in the brain of UCCA rats. A: Anatomical regions selected for ERα expression analysis and representative images from each group (×1; ×40). 1: Cornu Ammonis area 1; 2: Cornu Ammonis area 3; 3: Visual area 2, Mediolateral part; 4: Visual area 2, Lateral part; 5: Auditory Dorsal.B: Average optical density of ERα. C: Representative protein bands. D: Relative expression levels of p-Akt and p-PI3K. Data are presented as Mean±SD (n=6). *P<0.05, ***P<0.001 vs Sham; #P<0.05, ###P<0.001 vs Model.
| [1] | Zhi N, Ren RJ, Qi JL, et al. The China Alzheimer report 2025[J]. Gen Psych, 2025, 38(4): e102020. 12-26. doi:10.1136/gpsych-2024-102020 |
| [2] | 刘雨辉, 卜先乐, 马 辛, 等. 阿尔茨海默病药物治疗指南[J]. 阿尔茨海默病及相关病杂志, 2025, 8(1): 8-16. |
| [3] | Cummings JL, Zhou YD, Lee G, et al. Alzheimer's disease drug development pipeline: 2025[J]. lzheimers Dement (N Y), 2025, 11(2): e70098. doi:10.1002/trc2.70098 |
| [4] | Law BYK, Wu AG, Wang MJ, et al. Chinese medicine: a hope for neurodegenerative diseases [J]. J Alzheimers Dis, 2017, 60(s1): S151-60. doi:10.3233/jad-170374 |
| [5] | 魏江平, 赵子瑄, 曾 静, 等. 基于神经活性配体受体交互作用探讨党参-茯苓配伍改善痴呆小鼠学习记忆能力的作用机制[J]. 中药新药与临床药理, 2023, 34(11): 1514-24. |
| [6] | 徐 杨, 何芷绮, 刘晓凤, 等. 代谢组学在中药复方制剂领域的研究进展及面临的挑战[J]. 中草药, 2024, 55(4): 1354-64. |
| [7] | 刘玥欣, 徐 岩, 赵昕彤, 等. 鹿茸多肽对单侧颈总动脉结扎所致轻度认知功能障碍模型大鼠的保护作用研究[J]. 人参研究, 2019, 31(6): 42-4. |
| [8] | 曾 静, 陈 荣, 任香怡, 等. 异功散通过调控肠道微生物改善痴呆症模型大鼠的认知下降[J]. 南方医科大学学报, 2024, 44(7): 1297-305. |
| [9] | Shi ZS, Zhu LH, Li TT, et al. Neuroprotective mechanisms of Lycium barbarum polysaccharides against ischemic insults by regulating NR2B and NR2A containing NMDA receptor signaling pathways[J]. Front Cell Neurosci, 2017, 11: 288. doi:10.3389/fncel.2017.00288 |
| [10] | 王胜鹏, 胡杨洋, 陈锐娥, 等. 中药药对的系统研究(Ⅱ): 药效与药动学研究[J]. 世界科学技术: 中医药现代化, 2012, 14(2): 1322-8. |
| [11] | 牛 明, 张斯琴, 张 博, 等. 《网络药理学评价方法指南》解读[J]. 中草药, 2021, 52(14): 4119-29. |
| [12] | 武晓伟, 郝艳艳, 聂春霞, 等. 中药血清药物化学的方法学概述及应用领域研究进展[J]. 中国实验方剂学杂志, 2019, 25(3): 173-9. |
| [13] | 王一凯, 董金旭, 白玉涛, 等. 基于超高效液相色谱-四极杆飞行时间质谱和靶向网络药理学技术结合分子对接研究薤白治疗高血脂症的药效成分[J]. 分析化学, 2024, 52(4): 578-86. |
| [14] | Wang J, Chen Q, Sheng RL, et al. Integration of transdermal chemistry and network pharmacology to decipher the mechanism of ShexiangZhuifeng analgesic plaster to treat rheumatoid arthritis[J]. Phytomedicine, 2023, 108: 154507. doi:10.1016/j.phymed.2022.154507 |
| [15] | 朱星晔, 于嘉祥, 袁子越, 等. 基于LC-MS技术联合生物信息学预测半夏白术天麻汤和当归芍药散干预梅尼埃病的机制[J]. 中国实验方剂学杂志, 2025, 31(24): 50-60. |
| [16] | 杨 丽, 李孟莹, 胡文青, 等. 基于HPLC-Q-TOF-MS/MS和网络药理学的补肺丸入血成分、肺病治疗潜能及新型冠状病毒感染恢复期的应用机制研究[J]. 中国中药杂志, 2024, 49(20): 5598-609. |
| [17] | 唐志苓, 李 瑶, 支 颢, 等. 基于UPLC-Q-TOF/MS分析炙甘草汤体内成分及心脏组织分布特征[J]. 南京中医药大学学报, 2025, 41(7): 896-903. |
| [18] | Wang J, Wen W, Gong DY, et al. SZAP exerts analgesic effects on rheumatalgia in CIA rats by suppressing pain hyperalgesia and inhibiting TRPV1 and P2X3[J]. J Ethnopharmacol, 2022, 284: 114780. doi:10.1016/j.jep.2021.114780 |
| [19] | Samakashvili S, Ibáñez C, Simó C, et al. Analysis of chiral amino acids in cerebrospinal fluid samples linked to different stages of Alzheimer disease[J]. Electrophoresis, 2011, 32(19): 2757-64. doi:10.1002/elps.201100139 |
| [20] | Jo S, Yarishkin O, Hwang YJ, et al. GABA from reactive astrocytes impairs memory in mouse models of Alzheimer's disease[J]. Nat Med, 2014, 20(8): 886-96. doi:10.1038/nm.3639 |
| [21] | Conn KA, Borsom EM, Cope EK. Implications of microbe-derived ɣ-aminobutyric acid (GABA) in gut and brain barrier integrity and GABAergic signaling in Alzheimer's disease[J]. Gut Microbes, 2024, 16(1): 2371950. doi:10.1080/19490976.2024.2371950 |
| [22] | Kwakowsky A, Calvo-Flores Guzmán B, Pandya M, et al. GABAA receptor subunit expression changes in the human Alzheimer's disease hippocampus, subiculum, entorhinal cortex and superior temporal gyrus[J]. J Neurochem, 2018, 145(5): 374-92. doi:10.1111/jnc.14325 |
| [23] | Wu Z, Guo ZY, Gearing M, et al. Tonic inhibition in dentate gyrus impairs long-term potentiation and memory in an Alzheimer's [corrected] disease model[J]. Nat Commun, 2014, 5: 4159. doi:10.1038/ncomms5159 |
| [24] | Herbison AE. Estrogen regulation of GABA transmission in rat preoptic area[J]. Brain Res Bull, 1997, 44(4): 321-6. doi:10.1016/s0361-9230(97)00210-4 |
| [25] | Hara Y, Waters EM, McEwen BS, et al. Estrogen effects on cognitive and synaptic health over the lifecourse[J]. Physiol Rev, 2015, 95(3): 785-807. doi:10.1152/physrev.00036.2014 |
| [26] | Malyala A, Zhang CG, Bryant DN, et al. PI3K signaling effects in hypothalamic neurons mediated by estrogen[J]. J Comp Neurol, 2008, 506(6): 895-911. doi:10.1002/cne.21584 |
| [27] | Knowlton AA, Lee AR. Estrogen and the cardiovascular system[J]. Pharmacol Ther, 2012, 135(1): 54-70. doi:10.1016/j.pharmthera.2012.03.007 |
| [28] | Han X, Cheng XL, Xu JY, et al. Activation of TREM2 attenuates neuroinflammation via PI3K/Akt signaling pathway to improve postoperative cognitive dysfunction in mice[J]. Neuropharm-acology, 2022, 219: 109231. doi:10.1016/j.neuropharm.2022.109231 |
| [29] | Yang CZ, Wang SH, Zhang RH, et al. Neuroprotective effect of astragalin via activating PI3K/Akt-mTOR-mediated autophagy on APP/PS1 mice[J]. Cell Death Discov, 2023, 9(1): 15. doi:10.1038/s41420-023-01324-1 |
| [30] | Fu C, Wu YF, Liu SJ, et al. Rehmannioside A improves cognitive impairment and alleviates ferroptosis via activating PI3K/AKT/Nrf2 and SLC7A11/GPX4 signaling pathway after ischemia[J]. J Ethnopharmacol, 2022, 289: 115021. doi:10.1016/j.jep.2022.115021 |
| [31] | 刘志华, 孙晓波. 网络药理学: 中医药现代化的新机遇[J]. 药学学报, 2012, 47(6): 696-703. |
| [32] | Taleb O, Maammar M, Klein C, et al. A role for xanthurenic acid in the control of brain dopaminergic activity[J]. Int J Mol Sci, 2021, 22(13): 6974. doi:10.3390/ijms22136974 |
| [33] | Yang ZJ, Huang SY, Zhong KY, et al. Betaine alleviates cognitive impairment induced by homocysteine through attenuating NLRP3-mediated microglial pyroptosis in an m6A-YTHDF2-dependent manner[J]. Redox Biol, 2024, 69: 103026. doi:10.1016/j.redox.2024.103026 |
| [34] | Shi MM, Xu XF, Sun QM, et al. Betaine prevents cognitive dysfunction by suppressing hippocampal microglial activation in chronic social isolated male mice[J]. Phytother Res, 2023, 37(10): 4755-70. doi:10.1002/ptr.7944 |
| [1] | Jing ZENG, Lei HUA, Yong YANG, Xiaomei ZHANG, Jiangping WEI, Lisheng LI. Yigong San improves learning and memory functions of APP/PS1 transgenic mice by regulating brain fluid metabolism [J]. Journal of Southern Medical University, 2024, 44(10): 2015-2023. |
| [2] | CAO Fuyang, GUO Yongxin, GUO Shuting, ZHOU Zhikang, CAO Jiangbei, TONG Li, MI Weidong. Activation of GABAergic neurons in the zona incerta accelerates anesthesia induction with sevoflurane and propofol without affecting anesthesia maintenance or awakening in mice [J]. Journal of Southern Medical University, 2023, 43(5): 718-726. |
| [3] | . Communication sound recognition and response modification in the secondary auditory cortex of female mice [J]. Journal of Southern Medical University, 2021, 41(7): 1079-1086. |
| [4] | . Directed differentiation of porcine induced pluripotent stem cells into forebrain GABAergic neuron progenitors [J]. Journal of Southern Medical University, 2021, 41(6): 820-827. |
| [5] | . [J]. Journal of Southern Medical University, 2012, 32(10): 1469-. |
| [6] | CHEN Juan, ZOU Zhi-peng, WU Zhong-Hai Department of Physiology, Southern Medical University, Guangzhou 510515, China. Effects of pentobarbital sodium on rhythmical respiration of neonatal rat medullary preparations [J]. Journal of Southern Medical University, 2006, 26(09): 1273-. |
| [7] | CHEN Jing1,HUANG Yong1,WANG Sheng-xu1,LI Qiu-shi1,LIANG Yong-jiu2,GUO Yong-ning3 1Department of Traditional Chinese Medicine,Southern Medical University,Guangzhou 510515,China;2Medical Department,Millennium Rest home,Guangzhou 510407,China;3Medical Department,Xingguang Rest home,Guangzhou 510407,China. ~(18)FDG PET cerebral function imaging in 10 vascular dementia patients receiving needling at Baihui(DU20),Shuigou(DU26) and Shenmen(HT7) [J]. Journal of Southern Medical University, 2006, 26(05): 610-612. |
| [8] | HUANG Yong1, CHEN Jing1, LAI Xin-sheng2, TANG An-wu3, LI Dong-jiang3. Effects of needling in Baihui (DU20),Shuigou (DU26) and Shenmen (HT7) on glucose metabolism in the lentiform nuclus in patients with vascular dementia [J]. Journal of Southern Medical University, 2005, 25(11): 1405-1407. |
| [9] | OUYANG Shi, SUN Li-sha, GUO Sheng-lan, LIU Xu, XU Jiang-ping. Effects of timosaponins on learning and memory abilities of rats with dementia induced by lateral cerebral ventricular injection of amyloid β-peptide [J]. Journal of Southern Medical University, 2005, 25(02): 121-126. |
| [10] | YANG Guo-feng1, WANG Lu-ning1, ZHAO Xin2, NIE Yong-hui1. Two-dimensional electrophoregram for proteomic analysis of rat brain with intrahippocampal amyloid β injection and normal rat brain [J]. Journal of Southern Medical University, 2004, 24(05): 553-555. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||