Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 497-504.doi: 10.12122/j.issn.1673-4254.2026.03.03
Yan ZHANG1( ), Qingqiu WEN2, Haibo HUANG1, Meijuan ZHOU2(
), Qingqiu WEN2, Haibo HUANG1, Meijuan ZHOU2( ), Jianyu WANG1(
), Jianyu WANG1( )
)
Received:2025-10-15
Online:2026-03-20
Published:2026-03-26
Contact:
Meijuan ZHOU, Jianyu WANG
E-mail:18863301@qq.com;lkzmj@smu.edu.cn;13430230612@126.com
Supported by:Yan ZHANG, Qingqiu WEN, Haibo HUANG, Meijuan ZHOU, Jianyu WANG. Mefloquine HCl promotes DNA repair and alleviates radiation-induced lung epithelial cell injury[J]. Journal of Southern Medical University, 2026, 46(3): 497-504.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.03
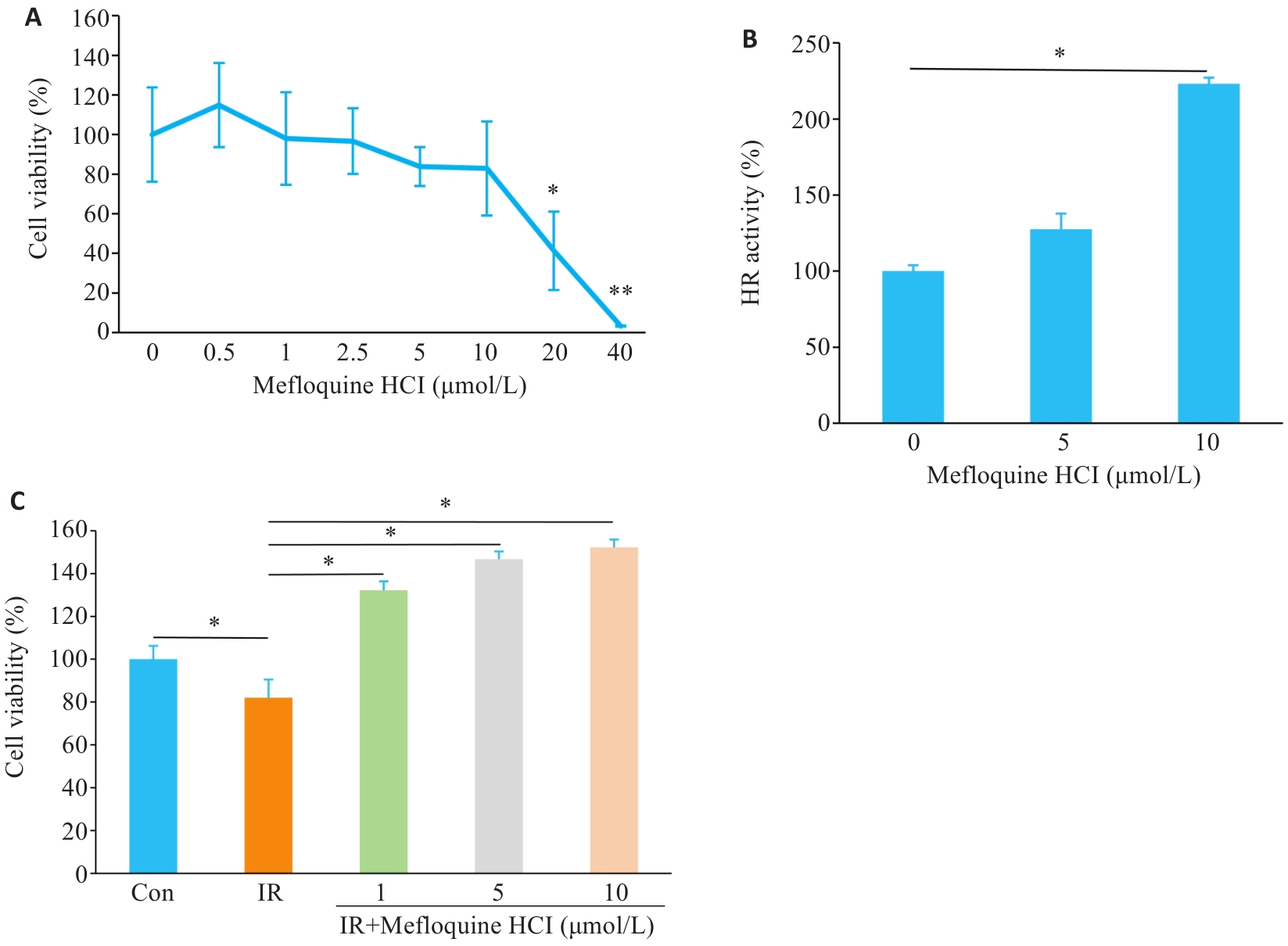
Fig.1 Effects of different concentrations of MQ on cell viability and HR repair efficiency. A: Cell viability curve. *P<0.05, **P<0.01 vs 0 μmol/L. B: Effect of MQ on HR repair efficiency. C: Effect of MQ on cell viability after irradiation. Data are presented as Mean±SD (n=3). *P<0.05.
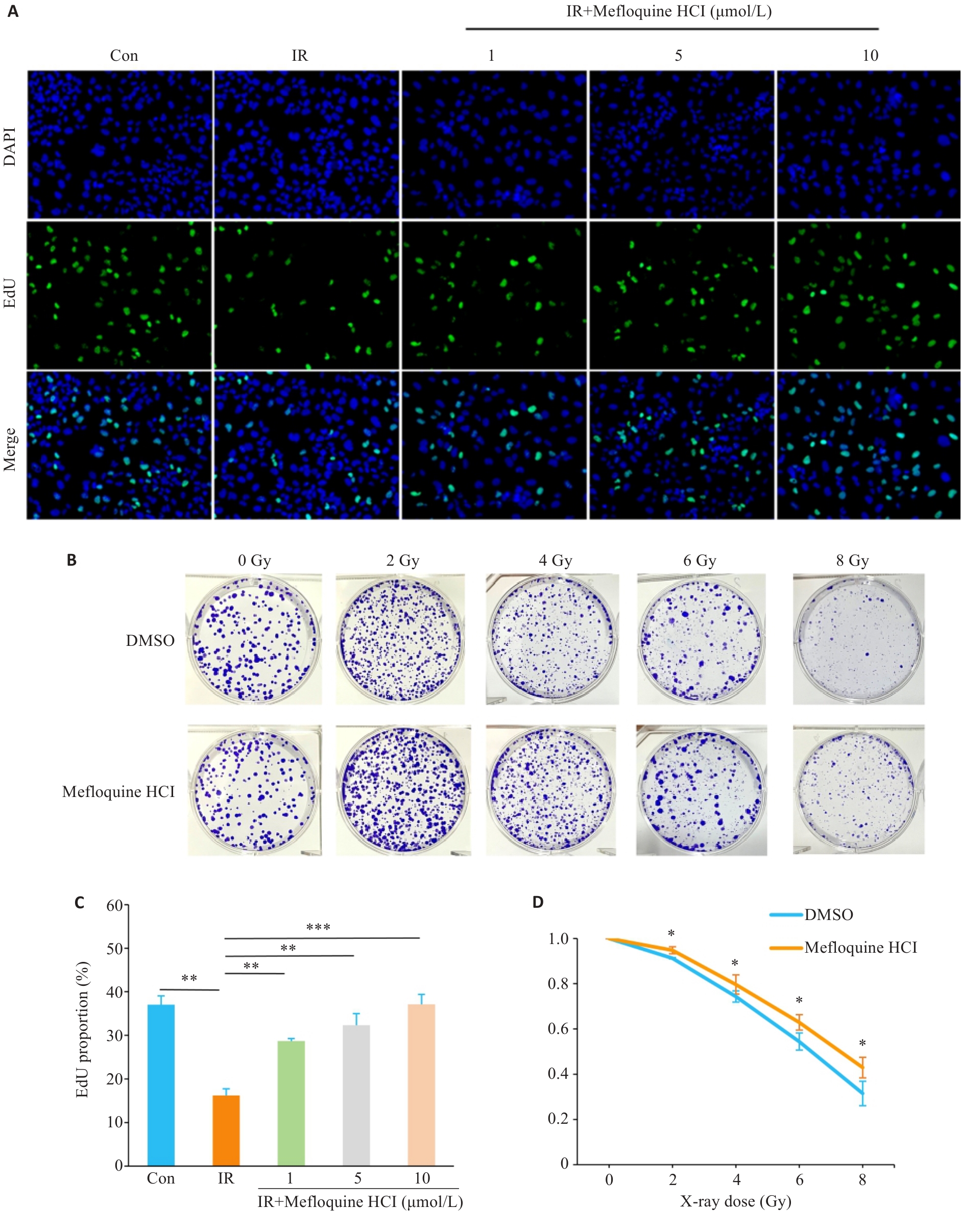
Fig.2 Effects of MQ on proliferation and colony formation capacity of X-ray-irradiated BEAS-2B cells. A: Cell proliferation after irradiation (Original magnification: ×50); B: BEAS-2B colony formation diagram. C, D: Statistical analysis diagrams. Data are presented as Mean±SD (n=3). *P<0.05, **P<0.01, ***P<0.001.
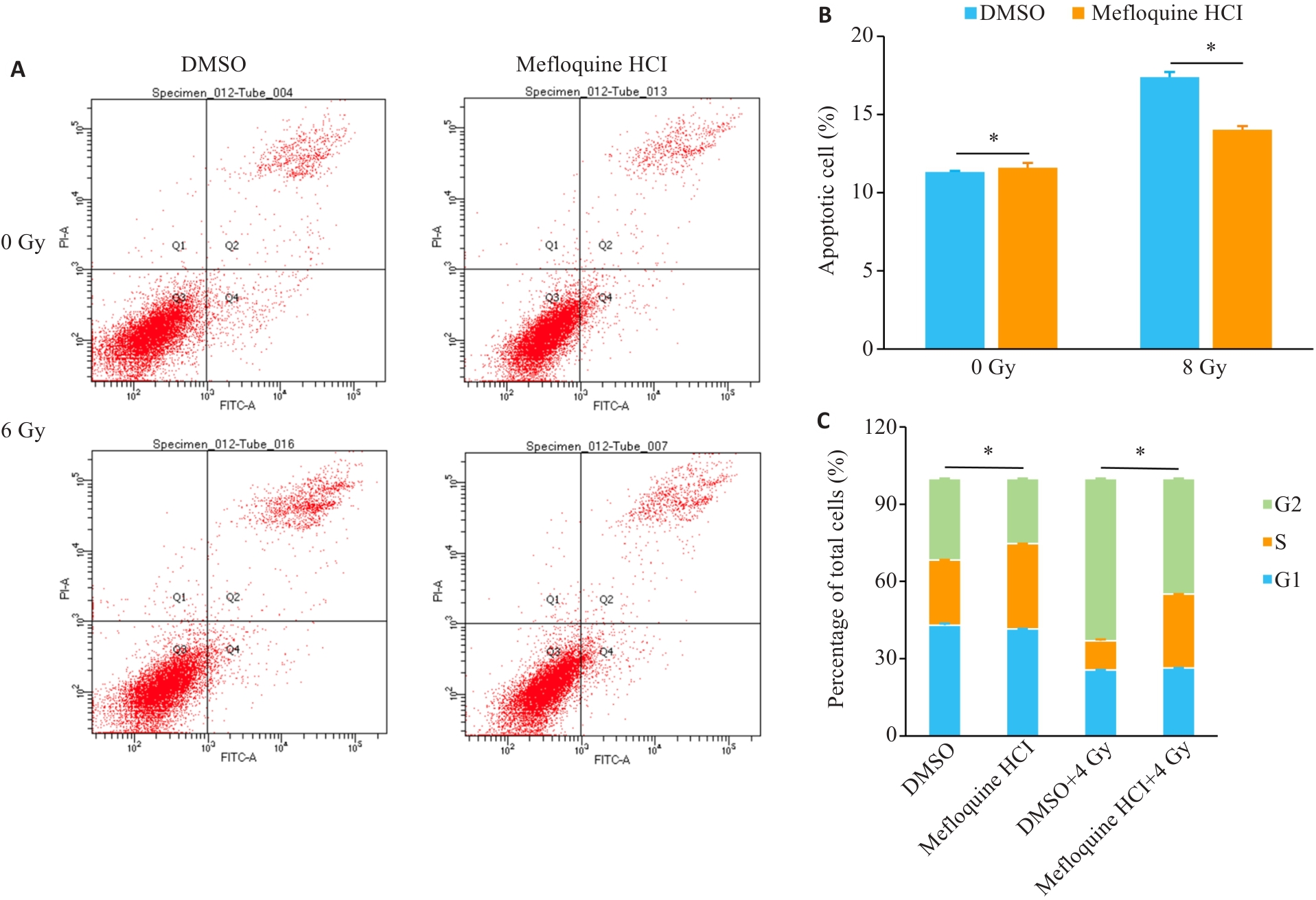
Fig.3 Flow cytometry analysis of the effects of MQ on apoptosis and cell cycle in X-ray-irradiated BEAS-2B cells. A, B: Apoptosis analysis by flow cytometry and statistical data. C. Statistical analysis of cell cycle. Data are presented as Mean±SD (n=3). *P<0.05.
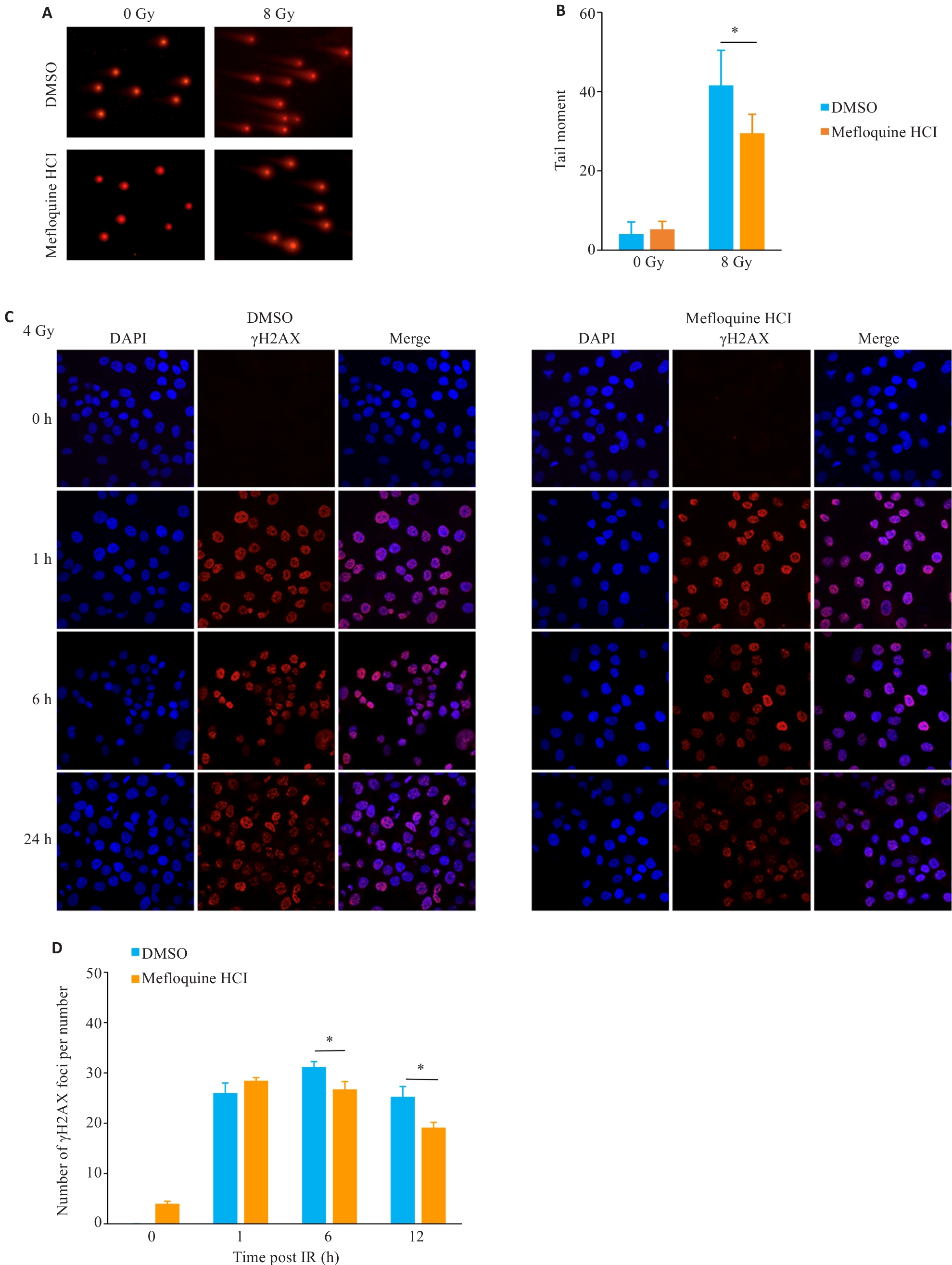
Fig.4 Effect of MQ on DNA double-strand break (DSB) repair following irradiation. A, B: Comet assay images and statistical analysis of irradiated BEAS-2B cells (×100). C, D: Immunofluorescence staining and quantification of γH2AX foci in BEAS-2B cells after irradiation (×50). Data are presented as Mean±SD (n=3). *P<0.05.
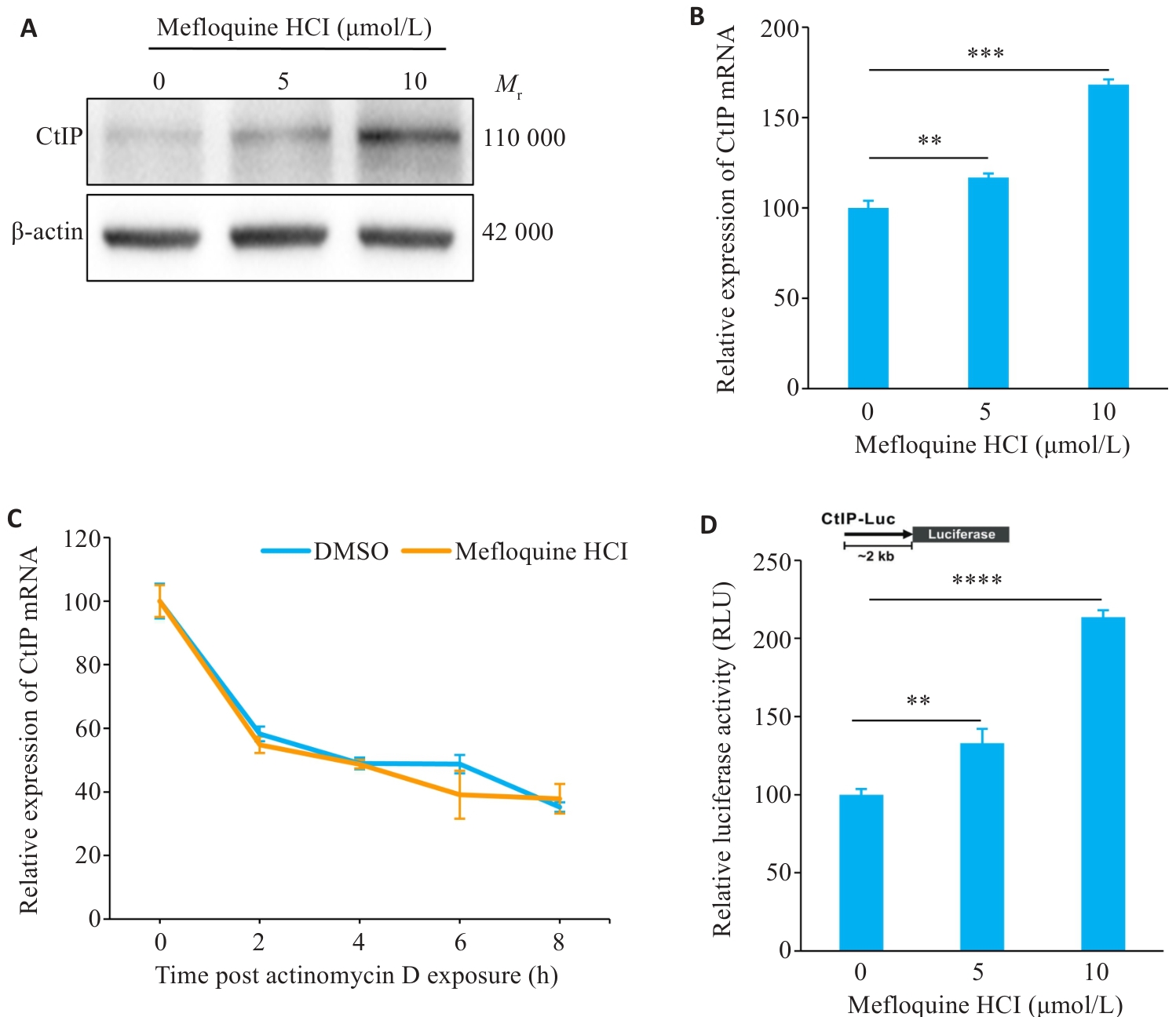
Fig.5 Effect of MQ on CtIP protein and mRNA levels. A: Protein expression of CtIP after MQ treatment. B: mRNA level of CtIP after MQ treatment. C: Effect of MQ on CtIP mRNA stability. D: Effect of MQ on CtIP promoter activity. Data are presented as Mean±SD (n=3). **P<0.01, ***P<0.001, ****P<0.0001.
| [1] | Baskar R, Lee KA, Yeo R, et al. Cancer and radiation therapy: current advances and future directions[J]. Int J Med Sci, 2012, 9(3): 193-9. doi:10.7150/ijms.3635 |
| [2] | Zhang K, Yang SH, Zhu Y, et al. Protection against acute radiation-induced lung injury: a novel role for the anti-angiogenic agent Endostar[J]. Mol Med Rep, 2012, 6(2): 309-15. doi:10.3892/mmr.2012.903 |
| [3] | Hanania AN, Mainwaring W, Ghebre YT, et al. Radiation-induced lung injury: assessment and management[J]. Chest, 2019, 156(1): 150-62. doi:10.1016/j.chest.2019.03.033 |
| [4] | Xu TK, Zhang YY, Chang PY, et al. Mesenchymal stem cell-based therapy for radiation-induced lung injury[J]. Stem Cell Res Ther, 2018, 9(1): 18. doi:10.1186/s13287-018-0776-6 |
| [5] | Zhen S, Qiang R, Lu JJ, et al. TGF-β1-based CRISPR/Cas9 gene therapy attenuates radiation-induced lung injury[J]. Curr Gene Ther, 2022, 22(1): 59-65. doi:10.2174/1566523220666201230100523 |
| [6] | Beach TA, Groves AM, Williams JP, et al. Modeling radiation-induced lung injury: lessons learned from whole Thorax irradiation[J]. Int J Radiat Biol, 2020, 96(1): 129-44. doi:10.1080/09553002.2018.1532619 |
| [7] | Wei YY, Gong YQ, Wei S, et al. Protection of the hematopoietic system against radiation-induced damage: drugs, mechanisms, and developments[J]. Arch Pharm Res, 2022, 45(8): 558-71. doi:10.1007/s12272-022-01400-7 |
| [8] | Dobbs LG, Johnson MD, Vanderbilt J, et al. The great big alveolar TI cell: evolving concepts and paradigms[J]. Cell Physiol Biochem, 2010, 25(1): 55-62. doi:10.1159/000272063 |
| [9] | Chen FQ, Zhao WN, Du CH, et al. Bleomycin induces senescence and repression of DNA repair via downregulation of Rad51[J]. Mol Med, 2024, 30(1): 54. doi:10.1186/s10020-024-00821-y |
| [10] | Du J, Chen FQ, Du CH, et al. Amodiaquine ameliorates stress-induced premature cellular senescence via promoting SIRT1-mediated HR repair[J]. Cell Death Discov, 2024, 10(1): 434. doi:10.1038/s41420-024-02201-1 |
| [11] | Arroyo-Hernández M, Maldonado F, Lozano-Ruiz F, et al. Radiation-induced lung injury: current evidence[J]. BMC Pulm Med, 2021, 21(1): 9. doi:10.1186/s12890-020-01376-4 |
| [12] | Zhou SH, Zhu JJ, Zhou PK, et al. Alveolar type 2 epithelial cell senescence and radiation-induced pulmonary fibrosis[J]. Front Cell Dev Biol, 2022, 10: 999600. doi:10.3389/fcell.2022.999600 |
| [13] | Dasgupta Q, Jiang A, Wen AM, et al. A human lung alveolus-on-a-chip model of acute radiation-induced lung injury[J]. Nat Commun, 2023, 14(1): 6506. doi:10.1038/s41467-023-42171-z |
| [14] | Kim H, Park SH, Han SY, et al. LXA4-FPR2 signaling regulates radiation-induced pulmonary fibrosis via crosstalk with TGF‑β/Smad signaling[J]. Cell Death Dis, 2020, 11(8): 653. doi:10.1038/s41419-020-02846-7 |
| [15] | Mu JY, Israili ZH, Dayton PG. Studies of the disposition and metabolism of mefloquine HCl (WR 142, 490), a quinolinemethanol antimalarial, in the rat. Limited studies with an analog, WR 30, 090[J]. Drug Metab Dispos, 1975, 3(3): 198-210. doi:10.1016/s0090-9556(25)05714-9 |
| [16] | Lang FC, Cornwell JA, Kaur K, et al. Abrogation of the G2/M checkpoint as a chemosensitization approach for alkylating agents[J]. Neuro Oncol, 2024, 26(6): 1083-96. doi:10.1093/neuonc/noad252 |
| [17] | Yao YH, Qiu XN, Chen M, et al. LKB1 mutations enhance radiosensitivity in non-small cell lung cancer cells by inducing G2/M cell cycle phase arrest[J]. Curr Mol Med, 2025, 25(3): 353-60. doi:10.2174/0115665240280822231221060656 |
| [18] | Bunting SF, Nussenzweig A. End-joining, translocations and cancer[J]. Nat Rev Cancer, 2013, 13(7): 443-54. doi:10.1038/nrc3537 |
| [19] | Zhou FY, Waterman DP, Ashton M, et al. Prolonged cell cycle arrest in response to DNA damage in yeast requires the maintenance of DNA damage signaling and the spindle assembly checkpoint[J]. eLife, 2024, 13: RP94334. doi:10.7554/elife.94334.3.sa0 |
| [20] | Chien TM, Yang CW, Yen CH, et al. Excavatolide C/cisplatin combination induces antiproliferation and drives apoptosis and DNA damage in bladder cancer cells[J]. Arch Toxicol, 2024, 98(5): 1543-60. doi:10.1007/s00204-024-03699-1 |
| [21] | Ciccia A, Elledge SJ. The DNA damage response: making it safe to play with knives[J]. Mol Cell, 2010, 40(2): 179-204. doi:10.1016/j.molcel.2010.09.019 |
| [22] | Chapman JR, Taylor MRG, Boulton SJ. Playing the end game: DNA double-strand break repair pathway choice[J]. Mol Cell, 2012, 47(4): 497-510. doi:10.1016/j.molcel.2012.07.029 |
| [23] | Liu T, Huang J. DNA end resection: facts and mechanisms[J]. Genomics Proteomics Bioinformatics, 2016, 14(3): 126-30. doi:10.1016/j.gpb.2016.05.002 |
| [24] | Lamarche BJ, Orazio NI, Weitzman MD. The MRN complex in double-strand break repair and telomere maintenance[J]. FEBS Lett, 2010, 584(17): 3682-95. doi:10.1016/j.febslet.2010.07.029 |
| [25] | Clarke TL, Cho HM, Ceppi I, et al. ZNF280A links DNA double-strand break repair to human 22q11.2 distal deletion syndrome[J]. Nat Cell Biol, 2025, 27(6): 1006-20. doi:10.1038/s41556-025-01674-1 |
| [26] | Jiang YN, Yam JC, Tham CC, et al. RB regulates DNA double strand break repair pathway choice by mediating CtIP dependent end resection[J]. Int J Mol Sci, 2020, 21(23): 9176. doi:10.3390/ijms21239176 |
| [27] | Xie DF, Huang Q, Zhou PK. Drug discovery targeting post-translational modifications in response to DNA damages induced by space radiation[J]. Int J Mol Sci, 2023, 24(8): 7656. doi:10.3390/ijms24087656 |
| [28] | Yadav N, Dwivedi A, Mujtaba SF, et al. Photosensitized mefloquine induces ROS-mediated DNA damage and apoptosis in keratinocytes under ambient UVB and sunlight exposure[J]. Cell Biol Toxicol, 2014, 30(5): 253-68. doi:10.1007/s10565-014-9280-7 |
| [29] | Yan KH, Yao CJ, Hsiao CH, et al. Mefloquine exerts anticancer activity in prostate cancer cells via ROS-mediated modulation of Akt, ERK, JNK and AMPK signaling[J]. Oncol Lett, 2013, 5(5): 1541-5. doi:10.3892/ol.2013.1211 |
| [30] | Martins AC, Paoliello MMB, Docea AO, et al. Review of the mechanism underlying mefloquine-induced neurotoxicity[J]. Crit Rev Toxicol, 2021, 51(3): 209-16. doi:10.1080/10408444.2021.1901258 |
| [1] | XUE Jiguo, LIU Jing, GENG Miao, YUE Jingwei, HE Haochen, FAN Jiao. Identification of potential hub genes of Alzheimer's disease by weighted gene co-expression network analysis [J]. Journal of Southern Medical University, 2021, 41(12): 1752-1762. |
| [2] | . Expressions of HELQ and RAD51C in endometrial stromal sarcoma and their clinical significance [J]. Journal of Southern Medical University, 2020, 40(07): 936-941. |
| [3] | MA Yu-feng1, CAI Ji-ye2, ZHAO Qiu-xiang3, YANG Pei-hui2 1Department of Chemistry, School of Pharmaceutical Sciences, Southern Medical University, Guangzhou 510515, China; 2Department of Chemistry, Jinan University, Guangzhou 510632, China; 3 Material Testing Center of Guangdong Province, Guangzhou 510080, China. Study of DNA damage induced by potassium dichromate and glutathione with atomic force microscope [J]. Journal of Southern Medical University, 2006, 26(10): 1427-1430. |
| [4] | CHEN Ping1,YANG Yao-qin2,TAO Hui-hong2,YANG Hu-chuan2 1College of Life Science and Technology,Tongji University,Shanghai 200092,China;2Department of Tumor Cytology Research,Medical College,Tongji University,Shanghai 200092,China. Effects of electromagnetic fields of different frequencies on proliferation and DNA damage of gallbladder cancer cells [J]. Journal of Southern Medical University, 2006, 26(03): 328-330. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||