Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 638-645.doi: 10.12122/j.issn.1673-4254.2026.03.17
Jie LI( ), Yinan LIU, Dong WANG, Haopeng LI, Xijing HE, Teng LU(
), Yinan LIU, Dong WANG, Haopeng LI, Xijing HE, Teng LU( )
)
Received:2025-09-24
Online:2026-03-20
Published:2026-03-26
Contact:
Teng LU
E-mail:15191589376@163.com;luteng656@126.com
Supported by:Jie LI, Yinan LIU, Dong WANG, Haopeng LI, Xijing HE, Teng LU. In vitro mechanical testing and finite element analysis of a novel β-Titanium alloy pedicle screw-rod fixation system[J]. Journal of Southern Medical University, 2026, 46(3): 638-645.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.17

Fig.2 Static mechanical testing of β-titanium alloy screws and rods. A: Tensile test of the β-titanium alloy rod. B: Compression-torsion test of the β-titanium alloy screw-rod system. C: Torsion test of the β-titanium alloy screw. D: Bending test of the β-titanium alloy screw.
| Materials | Element type | Constitutive equation | Young's modulus | Poisson's ratio (μ) |
|---|---|---|---|---|
| Cortical bone | C3D4 | Isotropic elastic | 12 GPa | 0.3 |
| Cancellous bone | C3D4 | neo-Hookean | C10 = 19.38 MPa, D = 0.0252 MPa | |
| Bony endplate | C3D8I | Isotropic elastic | 1.2 GPa | 0.29 |
| Cartilage endplate | C3D8I | neo-Hookean | C10 = 4.10 MPa, D =0.03 MPa | |
| Nucleus pulposus | C3D8H | Mooney-Rivlin | C10 = 0.12 MPa, C01 = 0.03 MPa | |
| Annulus ground | C3D8H | Mooney-Rivlin | C10 = 0.18 MPa, C01 = 0.045 MPa | |
| Annulus fiber | T3D2 | Hypoelastic | 360-550 MPa | |
| Screws and rods | C3D4 | Isotropic elastic | 110 GPa, 55 GPa | 0.3 |
| Cage | C3D4 | Isotropic elastic | 3.6 GPa | 0.25 |
| Bone grafts | C3D4 | Isotropic elastic | 100 MPa | 0.2 |
Tab.1 Material properties and element types in the finite element models
| Materials | Element type | Constitutive equation | Young's modulus | Poisson's ratio (μ) |
|---|---|---|---|---|
| Cortical bone | C3D4 | Isotropic elastic | 12 GPa | 0.3 |
| Cancellous bone | C3D4 | neo-Hookean | C10 = 19.38 MPa, D = 0.0252 MPa | |
| Bony endplate | C3D8I | Isotropic elastic | 1.2 GPa | 0.29 |
| Cartilage endplate | C3D8I | neo-Hookean | C10 = 4.10 MPa, D =0.03 MPa | |
| Nucleus pulposus | C3D8H | Mooney-Rivlin | C10 = 0.12 MPa, C01 = 0.03 MPa | |
| Annulus ground | C3D8H | Mooney-Rivlin | C10 = 0.18 MPa, C01 = 0.045 MPa | |
| Annulus fiber | T3D2 | Hypoelastic | 360-550 MPa | |
| Screws and rods | C3D4 | Isotropic elastic | 110 GPa, 55 GPa | 0.3 |
| Cage | C3D4 | Isotropic elastic | 3.6 GPa | 0.25 |
| Bone grafts | C3D4 | Isotropic elastic | 100 MPa | 0.2 |
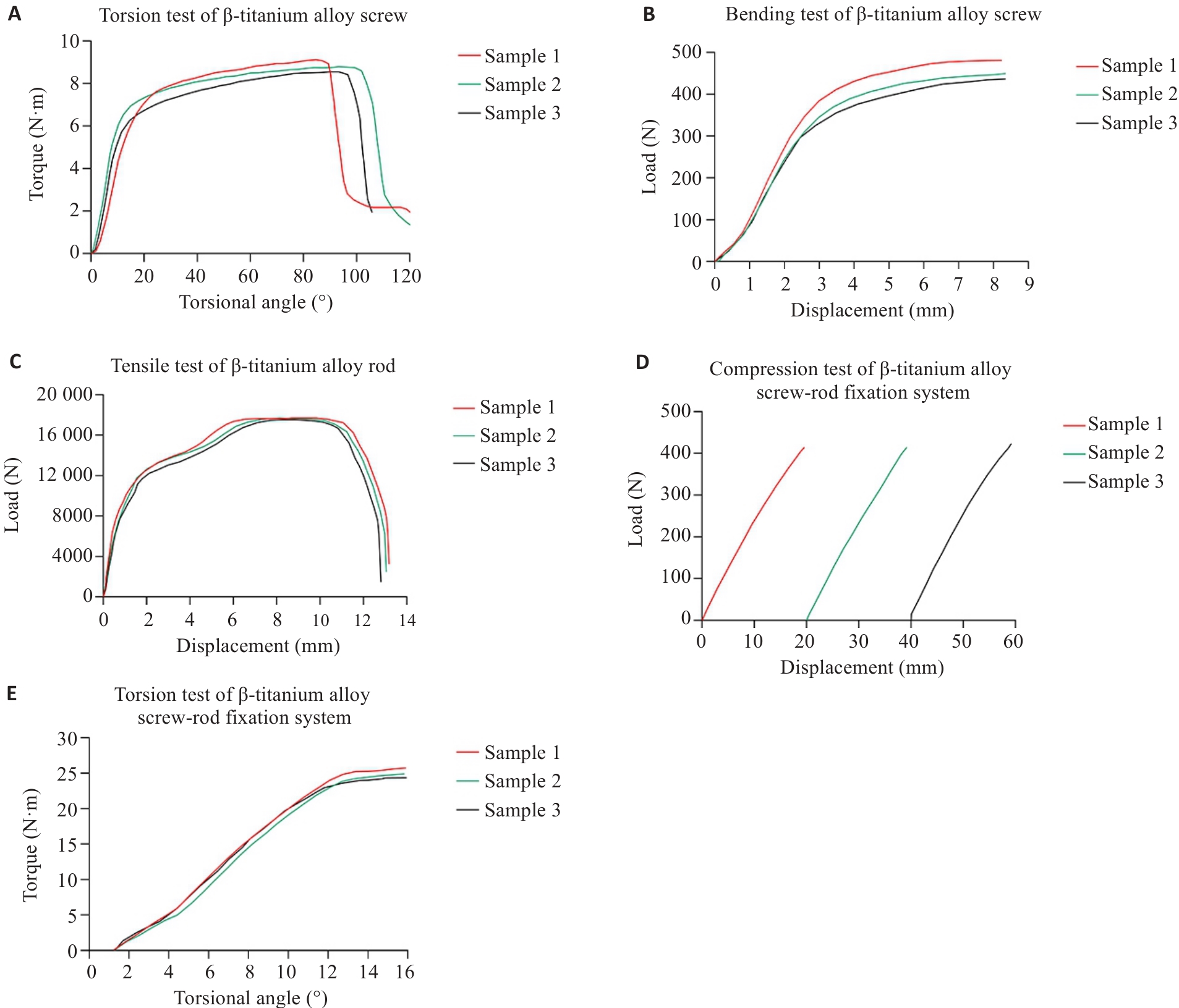
Fig.5 Results of in vitro mechanical testing of the β-titanium alloy pedicle screw-rod fixation system. A: Torsion test of the β‑titanium alloy screw. B: Bending test of the β‑titanium alloy screw. C: Tensile test of the β‑titanium alloy rod. D: Compression test of the β-titanium alloy screw-rod fixation system. E: Torsion test of the β‑titanium alloy screw-rod fixation system.
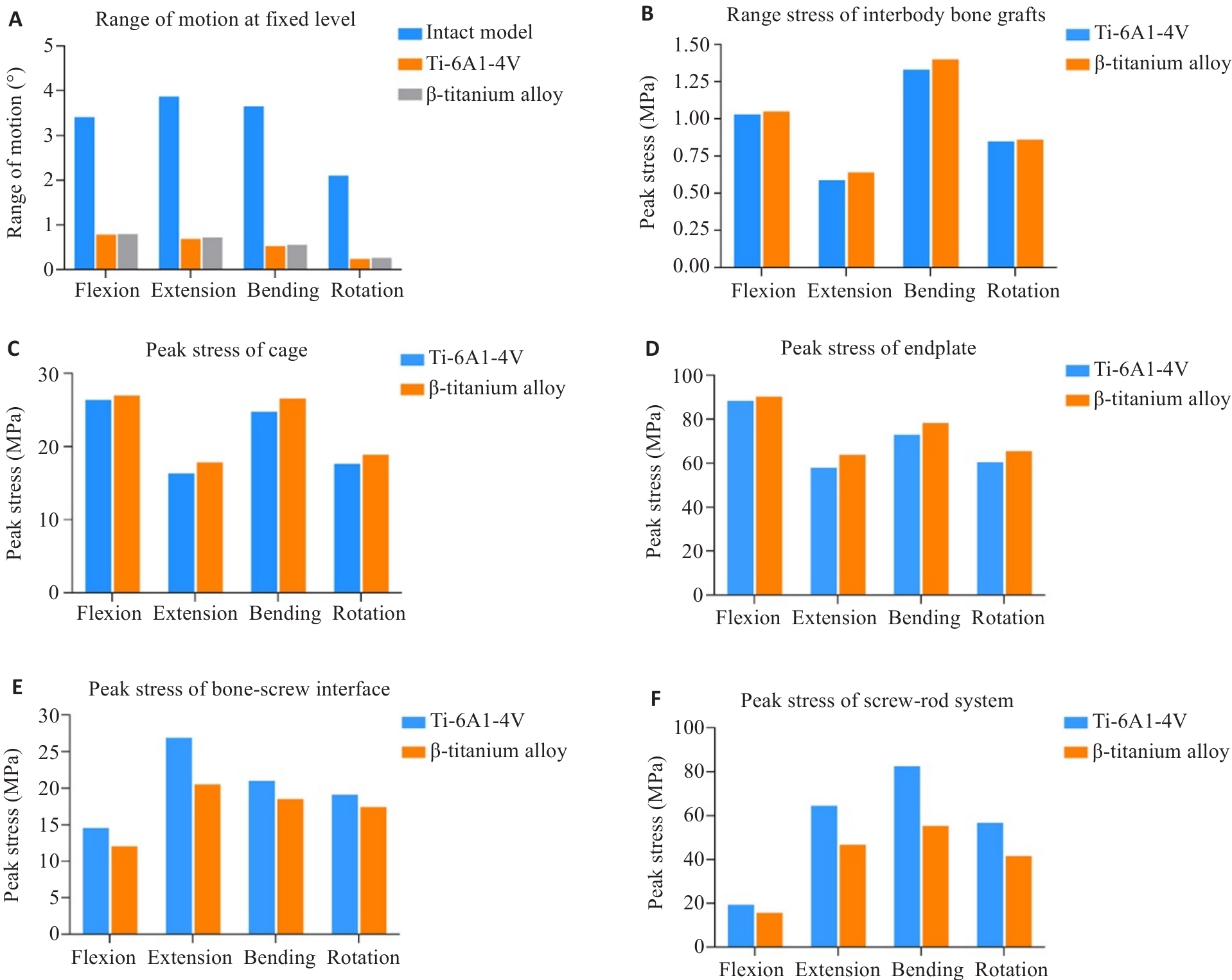
Fig.6 Results of segmental range of motion and stress within components. A: Segment range of motion for each model. B: Peak stress in the interbody bone graft. C: Peak stress in the cage. D: Peak stress in the endplate. E: Peak stress at the bone-screw interface. F: Peak stress in the screw-rod system.
| [1] | Chang MC, Choo YJ, Lee GW. Pedicle screws versus cortical screws in posterior lumbar interbody fusion surgery for degenerative spondylolisthesis: a systematic review and meta-analysis[J]. Spine J, 2021, 21(7): 1126-34. doi:10.1016/j.spinee.2021.02.019 |
| [2] | Eastlack RK, Nunley PD, Poelstra KA, et al. Finite element analysis comparing a PEEK posterior fixation device versus pedicle screws for lumbar fusion[J]. J Orthop Surg Res, 2023, 18(1): 855. doi:10.1186/s13018-023-04349-5 |
| [3] | Son HJ, Choi SH, Heo DR, et al. Outcomes of the use of cement-augmented cannulated pedicle screws in lumbar spinal fusion[J]. Spine J, 2021, 21(11): 1857-65. doi:10.1016/j.spinee.2021.05.005 |
| [4] | Lam TN, Trinh MG, Huang CC, et al. Investigation of bone growth in additive-manufactured pedicle screw implant by using Ti-6Al-4V and bioactive glass powder composite[J]. Int J Mol Sci, 2020, 21(20): E7438. doi:10.3390/ijms21207438 |
| [5] | Li J, Cao S, Zhao B. Biomechanical comparison of polyetheretherketone rods and titanium alloy rods in transforaminal lumbar interbody fusion: a finite element analysis[J]. BMC Surg, 2024, 24(1): 169. doi:10.1186/s12893-024-02462-8 |
| [6] | Zhang H, Wang Z, Wang Y, et al. Biomaterials for interbody fusion in bone tissue engineering[J]. Front Bioeng Biotechnol, 2022, 10: 900992. doi:10.3389/fbioe.2022.900992 |
| [7] | Loenen ACY, Peters MJM, Bevers RTJ, et al. Early bone ingrowth and segmental stability of a trussed titanium cage versus a polyether ether ketone cage in an ovine lumbar interbody fusion model[J]. Spine J, 2022, 22(1): 174-82. doi:10.1016/j.spinee.2021.07.011 |
| [8] | Wong KK, Hsu HC, Wu SC, et al. A review: design from beta titanium alloys to medium-entropy alloys for biomedical applications[J]. Materials: Basel, 2023, 16(21): 7046. doi:10.3390/ma16217046 |
| [9] | Xiang W, Yuan W, Deng H, et al. Effect of aging temperature on the microstructure and mechanical properties of a novel β titanium alloy[J]. Materials: Basel, 2023, 16(23): 7393. doi:10.3390/ma16237393 |
| [10] | Hua Z, Zhang D, Guo L, et al. Negative mixing enthalpy medium-entropy Ti-Zr-Nb-Al alloys with ultrahigh elastic admissible strain, strength-elongation product, and biocompatibility for bone implant applications[J]. Acta Biomater, 2025, 197: 476-94. doi:10.1016/j.actbio.2025.03.030 |
| [11] | Kacsó AB, Peter I. A review of past research and some future perspectives regarding titanium alloys in biomedical applications[J]. J Funct Biomater, 2025, 16(4): 144. doi:10.3390/jfb16040144 |
| [12] | Song T, Chen Z, Cui X, et al. Strong and ductile titanium-oxygen-iron alloys by additive manufacturing[J]. Nature, 2023, 618(7963): 63-8. doi:10.1038/s41586-023-05952-6 |
| [13] | Calazans Neto JV, Celles CAS, de Andrade CSAF, et al. Recent advances and prospects in β-type titanium alloys for dental implants applications[J]. ACS Biomater Sci Eng, 2024, 10(10): 6029-60. doi:10.1021/acsbiomaterials.4c00963 |
| [14] | Tshephe TS, Akinwamide SO, Olevsky E, et al. Additive manufacturing of titanium-based alloys-A review of methods, properties, challenges, and prospects[J]. Heliyon, 2022, 8(3): e09041. doi:10.1016/j.heliyon.2022.e09041 |
| [15] | Wang L, Zhou W, Yu Z, et al. An in vitro evaluation of the hierarchical micro/nanoporous structure of a Ti3Zr2Sn3Mo25Nb alloy after surface dealloying[J]. ACS Appl Mater Interfaces, 2021, 13(13): 15017-30. doi:10.1021/acsami.1c02140 |
| [16] | Jing WS, Zhang MH, Jin L, et al. Assessment of osteoinduction using a porous hydroxyapatite coating prepared by micro-arc oxidation on a new titanium alloy[J]. Int J Surg, 2015, 24: 51-6. doi:10.1016/j.ijsu.2015.08.030 |
| [17] | Yu S, Yu ZT, Wang G, et al. Biocompatibility and osteoconduction of active porous calcium-phosphate films on a novel Ti-3Zr-2Sn-3Mo-25Nb biomedical alloy[J]. Colloids Surf B Biointerfaces, 2011, 85(2): 103-15. doi:10.1016/j.colsurfb.2011.02.025 |
| [18] | Li J, Cao S, Guo D, et al. Biomechanical properties of different anterior and posterior techniques for atlantoaxial fixation: a finite element analysis[J]. J Orthop Surg Res, 2023, 18(1): 456. doi:10.1186/s13018-023-03905-3 |
| [19] | Li K, Cao S, Chen J, et al. Determining a relative total lumbar range of motion to alleviate adjacent segment degeneration after transforaminal lumbar interbody fusion: a finite element analysis[J]. BMC Musculoskelet Disord, 2024, 25(1): 197. doi:10.1186/s12891-024-07322-3 |
| [20] | Lu T, Ren JK, Sun ZW, et al. Relationship between the elastic modulus of the cage material and the biomechanical properties of transforaminal lumbar interbody fusion: a logarithmic regression analysis based on parametric finite element simulations[J]. Comput Meth Programs Biomed, 2022, 214: 106570. doi:10.1016/j.cmpb.2021.106570 |
| [21] | Lu T, Sun ZW, Xia HH, et al. Comparing the osteogenesis outcomes of different lumbar interbody fusions (A/O/X/T/PLIF) by evaluating their mechano-driven fusion processes[J]. Comput Biol Med, 2024, 171: 108215. doi:10.1016/j.compbiomed.2024.108215 |
| [22] | Li J, Du Z, Cao S, et al. Quantitative relationships between elastic modulus of rod and biomechanical properties of transforaminal lumbar interbody fusion: a finite element analysis[J]. Front Bioeng Biotechnol, 2024, 12: 1510597. doi:10.3389/fbioe.2024.1510597 |
| [23] | Wong CE, Hu HT, Kao LH, et al. Biomechanical feasibility of semi-rigid stabilization and semi-rigid lumbar interbody fusion: a finite element study[J]. BMC Musculoskelet Disord, 2022, 23(1): 10. doi:10.1186/s12891-021-04958-3 |
| [24] | Xie Y, Ma L, Yang Z, et al. Biomechanical analysis of posterior, transforaminal, extreme, oblique, and anterior lumbar interbody fusion surgical models: a finite element study[J]. Comput Methods Biomech Biomed Engin, 2025: 1-14. doi:10.1080/10255842.2025.2514135 |
| [25] | Sarraf M, Rezvani Ghomi E, Alipour S, et al. A state-of-the-art review of the fabrication and characteristics of titanium and its alloys for biomedical applications[J]. Bio Des Manuf, 2022, 5(2): 371-95. doi:10.1007/s42242-021-00170-3 |
| [26] | Straumal BB, Anisimova NY, Kiselevskiy MV, et al. Influence of the phase composition of titanium alloys on cell adhesion and surface colonization[J]. Materials, 2023, 16(22): 7130. doi:10.3390/ma16227130 |
| [27] | Dias Corpa Tardelli J, Bolfarini C, Cândido dos Reis A. Comparative analysis of corrosion resistance between beta titanium and Ti-6Al-4V alloys: a systematic review[J]. J Trace Elem Med Biol, 2020, 62: 126618. doi:10.1016/j.jtemb.2020.126618 |
| [28] | Liu J, Wang K, Li X, et al. Biocompatibility and osseointegration properties of a novel high strength and low modulus β-Ti10Mo6Zr4Sn3Nb alloy[J]. Front Bioeng Biotechnol, 2023, 11: 1127929. doi:10.3389/fbioe.2023.1127929 |
| [29] | Sass JO, Sellin ML, Kauertz E, et al. Advanced Ti-Nb-Ta alloys for bone implants with improved functionality[J]. J Funct Biomater, 2024, 15(2): 46. doi:10.3390/jfb15020046 |
| [30] | Okulov A, Berger S, Okulov I. Influence of β-stabilizer element on microstructure and mechanical behavior of porous titanium alloy synthesized by liquid metal dealloying[J]. Materials: Basel, 2023, 16(16): 5699. doi:10.3390/ma16165699 |
| [31] | Sing SL. Perspectives on additive manufacturing enabled beta-titanium alloys for biomedical applications[J]. Int J Bioprint, 2022, 8(1): 478. doi:10.18063/ijb.v8i1.478 |
| [32] | Giorgio I, Dell'Isola F, Andreaus U, et al. An orthotropic continuum model with substructure evolution for describing bone remodeling: an interpretation of the primary mechanism behind Wolff's law[J]. Biomech Model Mechanobiol, 2023, 22(6): 2135-52. doi:10.1007/s10237-023-01755-w |
| [1] | CHEN Jianmin, LIU Guoyin, BAO Tianyi, BAI Tianting, ZHANG Erlai, ZHAO Jianning. Biomechanical analysis of miniplate fixation systems in restorative laminoplasty for spinal canal reconstruction [J]. Journal of Southern Medical University, 2023, 43(3): 331-339. |
| [2] | SUN Yue, GUO Yun, LI Jiancheng, LIU Liang, YANG Dongkun, CHEN Mo, HU Kai. Mandibular defect reconstruction using digital design-assisted free fibula flap and three-dimensional finite element analysis of stress distribution [J]. Journal of Southern Medical University, 2021, 41(12): 1892-1898. |
| [3] | . Prognostic evaluation of hip joint function following capsule repair based on a threedimensional finite element analysis model [J]. Journal of Southern Medical University, 2020, 40(12): 1826-1830. |
| [4] | . Biomechanical study of cystic lesions of the mandible based on a three-dimensional finite element model [J]. Journal of Southern Medical University, 2020, 40(06): 911-915. |
| [5] | . Impact of anterior cervical fusion surgeries on adjacent segments: a finite element analysis [J]. Journal of Southern Medical University, 2017, 37(01): 49-. |
| [6] | . Finite element modeling of material property assignment based on CT gray value and its application in simulation of osteotomy for deformities [J]. Journal of Southern Medical University, 2016, 36(07): 979-. |
| [7] | SHU Xiao-qiu1, WAN Lei1, YU Chun-tang2, YIN Tong1, JIN An-min1 1Department of Orthopedics, 2Department of Radiology, Zhujiang Hospital, Southern Medical University, Guangzhou 510282, China. Finite element analysis of a new shape memory alloy cervical hook for atlantoaxial instability [J]. Journal of Southern Medical University, 2006, 26(08): 1197-1199. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||