南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 956-966.doi: 10.12122/j.issn.1673-4254.2026.04.24
• • 上一篇
刘坤1,2( ), 李晓莹1,2(
), 李晓莹1,2( ), 张玉罡2, 孙嘉2(
), 张玉罡2, 孙嘉2( ), 廖文镇3(
), 廖文镇3( )
)
收稿日期:2026-01-05
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
孙嘉,廖文镇
E-mail:liukun_gdou@163.com;xiaoyinglee0814@163.com;sunjia@smu.edu.cn;wenzhenliao@163.com
作者简介:刘 坤,副研究员,硕士生导师,E-mail: liukun_gdou@163.com基金资助:
Kun LIU1,2( ), Xiaoying LI1,2(
), Xiaoying LI1,2( ), Yugang ZHANG2, Jia SUN2(
), Yugang ZHANG2, Jia SUN2( ), Wenzhen LIAO3(
), Wenzhen LIAO3( )
)
Received:2026-01-05
Online:2026-04-20
Published:2026-04-24
Contact:
Jia SUN, Wenzhen LIAO
E-mail:liukun_gdou@163.com;xiaoyinglee0814@163.com;sunjia@smu.edu.cn;wenzhenliao@163.com
Supported by:摘要:
代谢性疾病如肥胖、糖尿病、心血管疾病的全球流行与营养过剩及饮食模式失衡密切相关。淀粉作为人类主要能量来源的碳水化合物,其消化特性直接影响糖脂代谢稳态。抗消化性淀粉(RS)因其独特的抗消化特性与益生元功能,成为当前改善糖脂代谢紊乱的膳食营养研究的热点。本文综述了淀粉消化特性以及RS改善代谢性疾病的综合作用及相关机制。高RS的膳食不仅通过延缓葡萄糖释放优化血糖稳态,而且未消化的部分进入结肠通过驱动肠道菌群-肠-脑轴代谢调控网络,其中包括激活腺苷酸活化蛋白激酶/乙酰辅酶A羧化酶(AMPK/ACC)通路减少脂肪蓄积、提升短链脂肪酸(SCFAs)介导的肠屏障功能与胰高血糖素样肽-1/肽YY(GLP-1/PYY)神经信号传导。这不仅将为新型健康食品设计提供研究基础,也为优化膳食营养,调控高碳水饮食引起的糖脂代谢紊乱提供了新的研究思路。
刘坤, 李晓莹, 张玉罡, 孙嘉, 廖文镇. 高抗消化性淀粉膳食调控糖脂代谢研究进展[J]. 南方医科大学学报, 2026, 46(4): 956-966.
Kun LIU, Xiaoying LI, Yugang ZHANG, Jia SUN, Wenzhen LIAO. Research progress in mechanisms of dietary resistant starch for regulating glucose and lipid metabolism[J]. Journal of Southern Medical University, 2026, 46(4): 956-966.
| Starch | Morphology | Size | Crystaline |
|---|---|---|---|
| Rice starch | Uniform polyhedron | 3~8 μm | Type A |
| Wheat starch | Polyhedral or spherical | 2~40 μm | Type A |
| Maize starch | Polyhedral or spherical | 5~25 μm | Type A & C |
| Potato starch | Ovoid or elliptical | 15~100 μm | Type B |
| Barley starch | Round or polygonal | 5~25 μm | Type A |
| Highland barley starch | Irregular polygonal | 10~30 μm | Hybrid A&B |
| Mung bean starch | Irregular polygonal | 15~45 μm | Type C |
| Sorghum starch | Polygonal or spherical | 5~20 μm | Type A |
| Millet starch | Polygonal | 2~10 μm | Type A |
| Cassava starch | Round or shell-like pattern | 5~35 μm | Type A or C |
| Sweet potato starch | Elliptical or bell-shaped | 10~50 μm | Type A or C |
| kudzu vine starch | Polygonal or ovoid | 5~30 μm | Type B |
表1 人类主食主要淀粉及其特性
Tab.1 Main dietary starch in human staple foods and their characteristics
| Starch | Morphology | Size | Crystaline |
|---|---|---|---|
| Rice starch | Uniform polyhedron | 3~8 μm | Type A |
| Wheat starch | Polyhedral or spherical | 2~40 μm | Type A |
| Maize starch | Polyhedral or spherical | 5~25 μm | Type A & C |
| Potato starch | Ovoid or elliptical | 15~100 μm | Type B |
| Barley starch | Round or polygonal | 5~25 μm | Type A |
| Highland barley starch | Irregular polygonal | 10~30 μm | Hybrid A&B |
| Mung bean starch | Irregular polygonal | 15~45 μm | Type C |
| Sorghum starch | Polygonal or spherical | 5~20 μm | Type A |
| Millet starch | Polygonal | 2~10 μm | Type A |
| Cassava starch | Round or shell-like pattern | 5~35 μm | Type A or C |
| Sweet potato starch | Elliptical or bell-shaped | 10~50 μm | Type A or C |
| kudzu vine starch | Polygonal or ovoid | 5~30 μm | Type B |
| Source | RDS | SDS | RS |
|---|---|---|---|
| Rice[ | 44-45% | 37-41% | 17-18% |
| Wheat[ | 61.02% | 15.55% | 23.43% |
| Maize[ | 79.71% | 2.54% | 17.75% |
| Potato[ | 69.91% | 21.54% | 8.55% |
| Barley[ | 96.19 % | 1.54 % | 2.27 % |
| Cassava[ | 76-80% | 12-17% | 1.6-7.3% |
| Millet[ | 46.37% | 28.03% | 25.61% |
表2 主食淀粉RDS、SDS和RS比例
Tab.2 Proportions of RDS, SDS and RS in staple food starches
| Source | RDS | SDS | RS |
|---|---|---|---|
| Rice[ | 44-45% | 37-41% | 17-18% |
| Wheat[ | 61.02% | 15.55% | 23.43% |
| Maize[ | 79.71% | 2.54% | 17.75% |
| Potato[ | 69.91% | 21.54% | 8.55% |
| Barley[ | 96.19 % | 1.54 % | 2.27 % |
| Cassava[ | 76-80% | 12-17% | 1.6-7.3% |
| Millet[ | 46.37% | 28.03% | 25.61% |
| Classification of carbohydrate diets | Carbohydrate proportion |
|---|---|
| High-carbohydrate diet | >65% total calories |
| Typical carbohydrate diet | 45%~65% total calories |
| Moderately restricted carbohydrate diet | 26%~44% total calories |
| Low-carbohydrate diet | <130 g/d (equivalent to 26% of a 2000-calorie diet) |
| Very low-carbohydrate diet | Usually provides 20-50 g of carbohydrates; depending on total caloric intake, carbohydrates may contribute 5%-15% of total calories. |
表3 不同类型饮食的特征
Tab.3 Characteristics of different types of diets
| Classification of carbohydrate diets | Carbohydrate proportion |
|---|---|
| High-carbohydrate diet | >65% total calories |
| Typical carbohydrate diet | 45%~65% total calories |
| Moderately restricted carbohydrate diet | 26%~44% total calories |
| Low-carbohydrate diet | <130 g/d (equivalent to 26% of a 2000-calorie diet) |
| Very low-carbohydrate diet | Usually provides 20-50 g of carbohydrates; depending on total caloric intake, carbohydrates may contribute 5%-15% of total calories. |
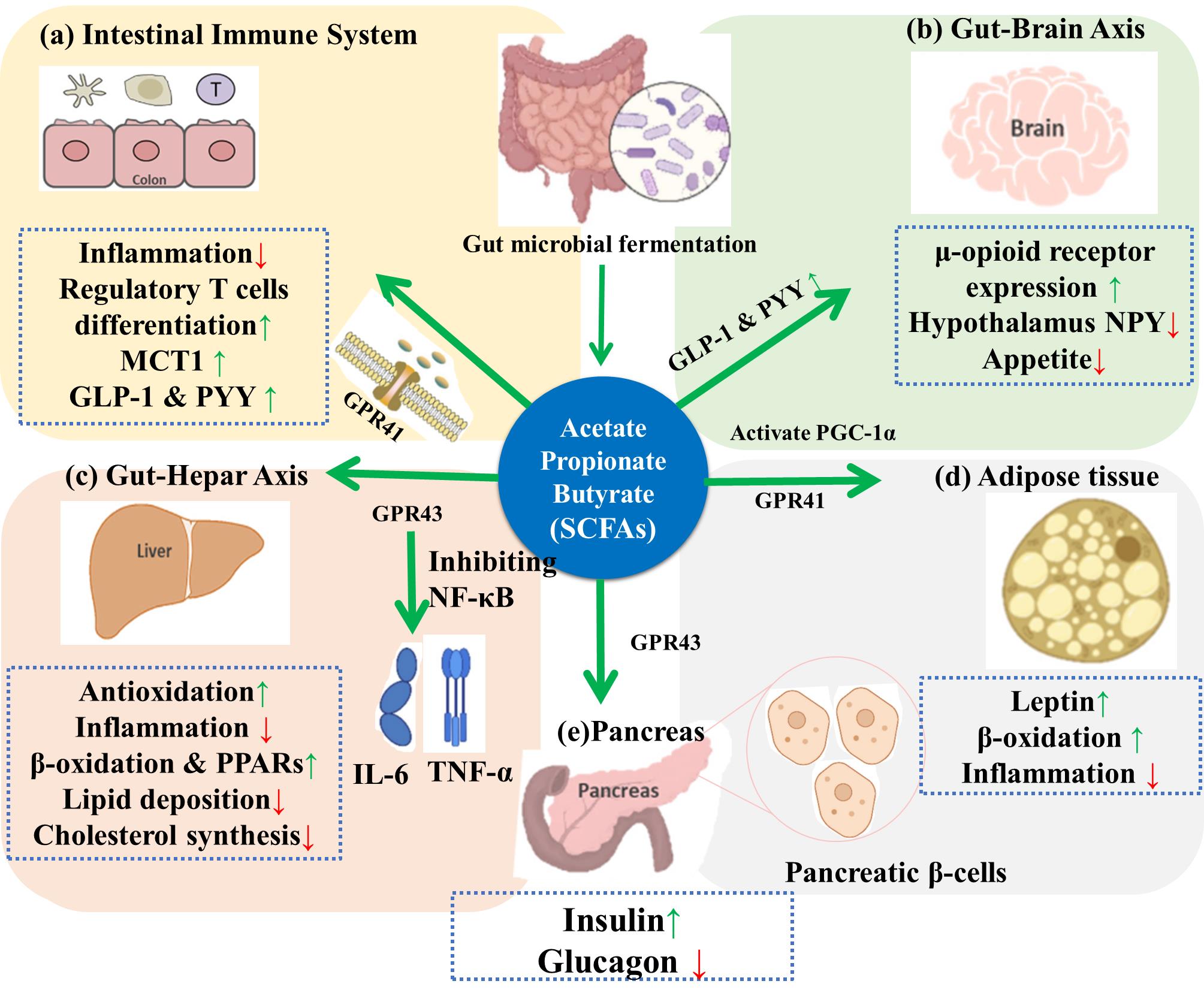
图1 高抗消化性淀粉对肥胖的调控作用
Fig.1 Regulatory effects of dietary resistant starch on obesity: (a) Short-chain fatty acids alleviate intestinal inflammation through GPR41 and promote the secretion of GLP-1 and PYY by L cells; (b) GLP-1 and PYY act on the brain to regulate appetite; (c) SCFAs regulate bile acid metabolism and cholesterol synthesis through the gut-liver axis; (d) SCFAs promote the production and secretion of leptin in adipose tissue by activating PGC-1α receptors; (e) SCFAs act on insulin β cells to promote insulin secretion and regulate blood glucose levels.
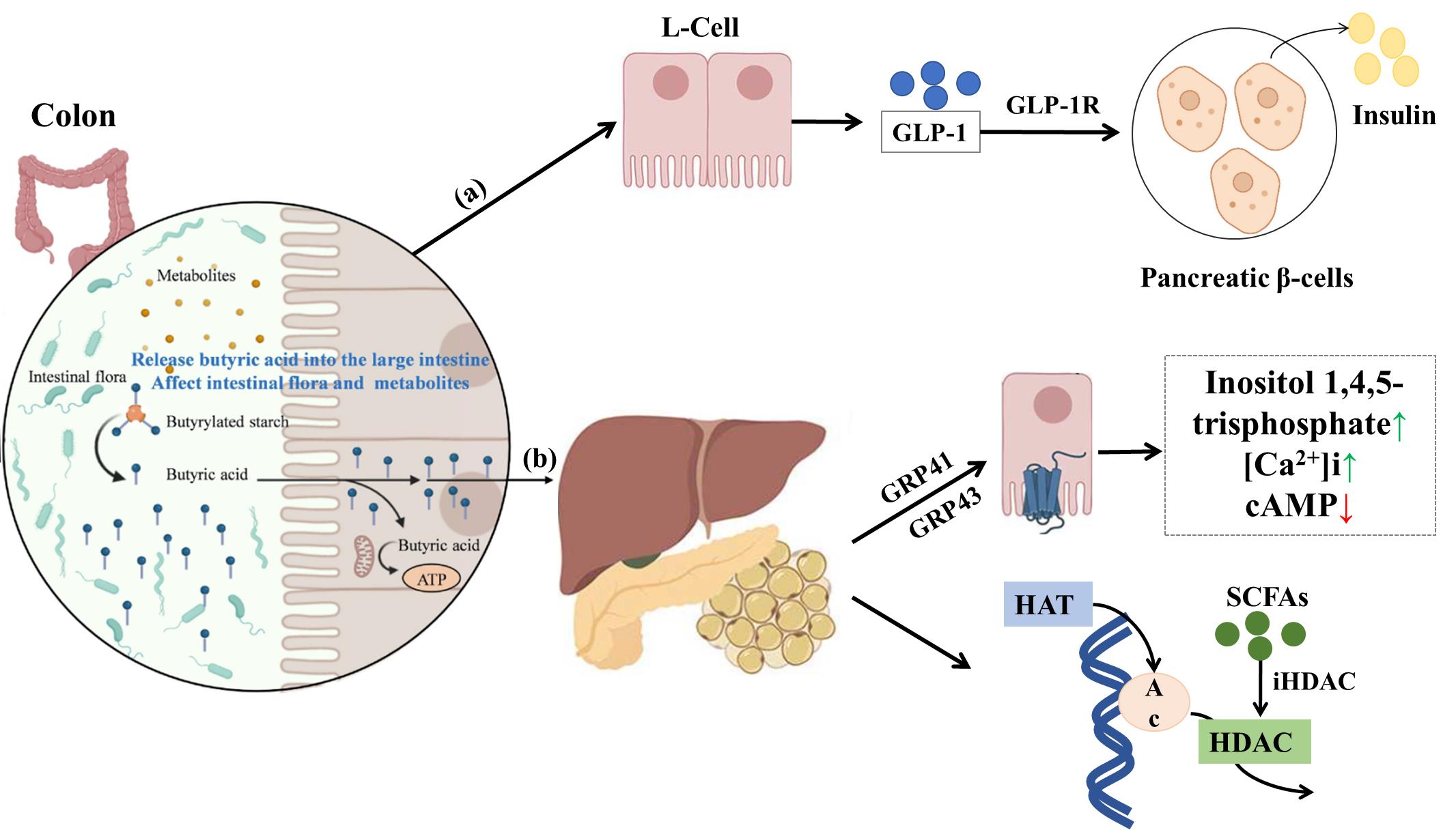
图2 高抗消化性淀粉对糖尿病的调控作用
Fig.2 Regulatory effects of dietary resistant starch on diabetes. (a) Short-chain fatty acids, the metabolites of resistant starch, promote the release of GLP-1 from L cells, which can act on pancreatic β cells to promote insulin release and reduce glucagon production; (b) Short-chain fatty acids can promote the production of cAMP through GPR41 and GPR43, thereby increasing glucose-stimulated insulin secretion. As histone deacetylase inhibitors, short-chain fatty acids also target class I and class II HDACs and regulate the expression of glucose-6-phosphate and gluconeogenesis to improve hyperglycemia.
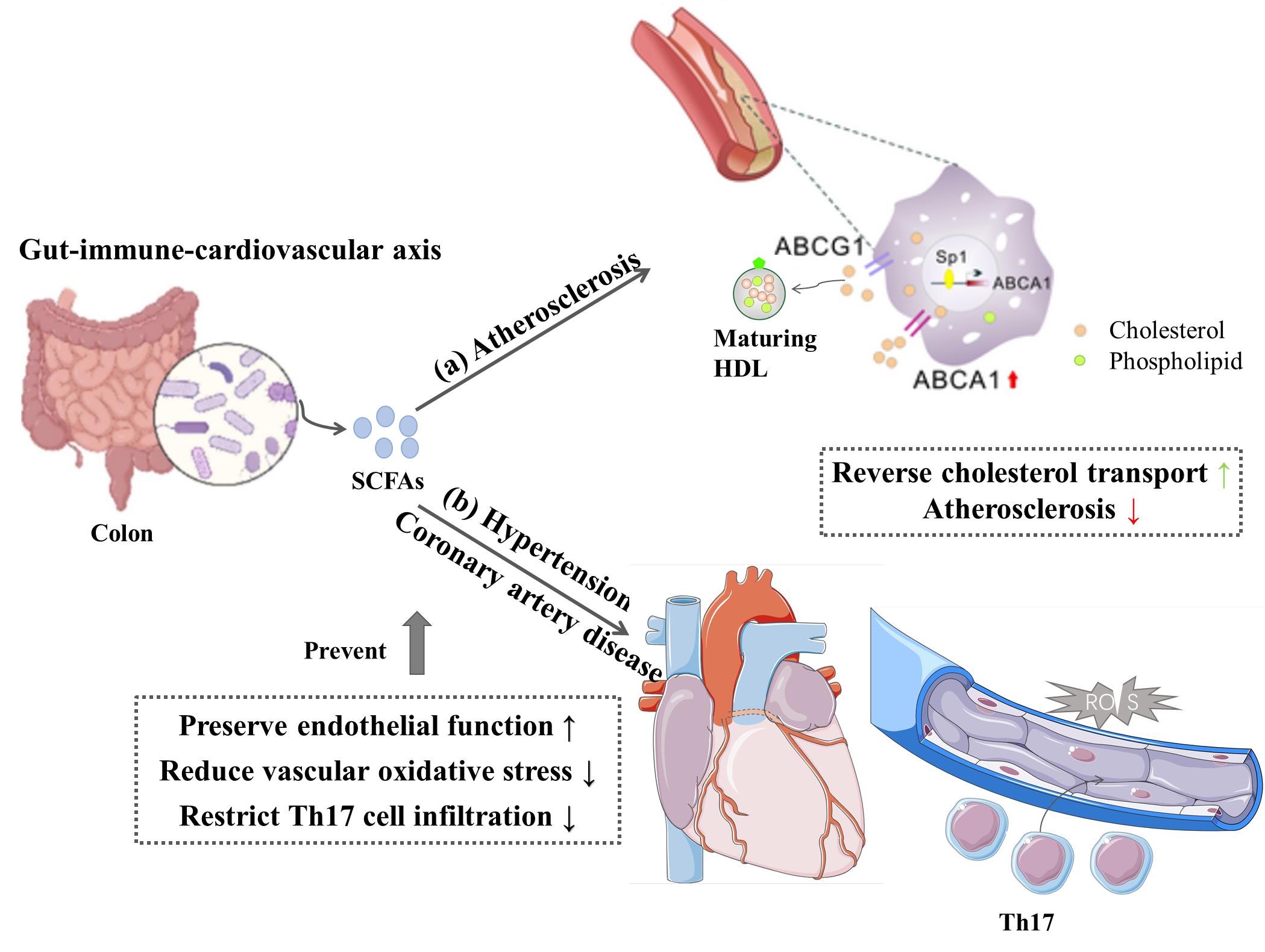
图3 高抗消化性淀粉对心血管系统调控作用
Fig.3 Regulatory effects of resistant starch on the cardiovascular system. (a) Short-chain fatty acids can accelerate reverse cholesterol transport (RCT) by activating ABCA1 expression, thereby preventing the development of atherosclerosis; (b) Short-chain fatty acids stabilize vascular endothelial function, reduce oxidative stress and limit Th17 cell infiltration, thereby lowering the risks of cardiovascular diseases.
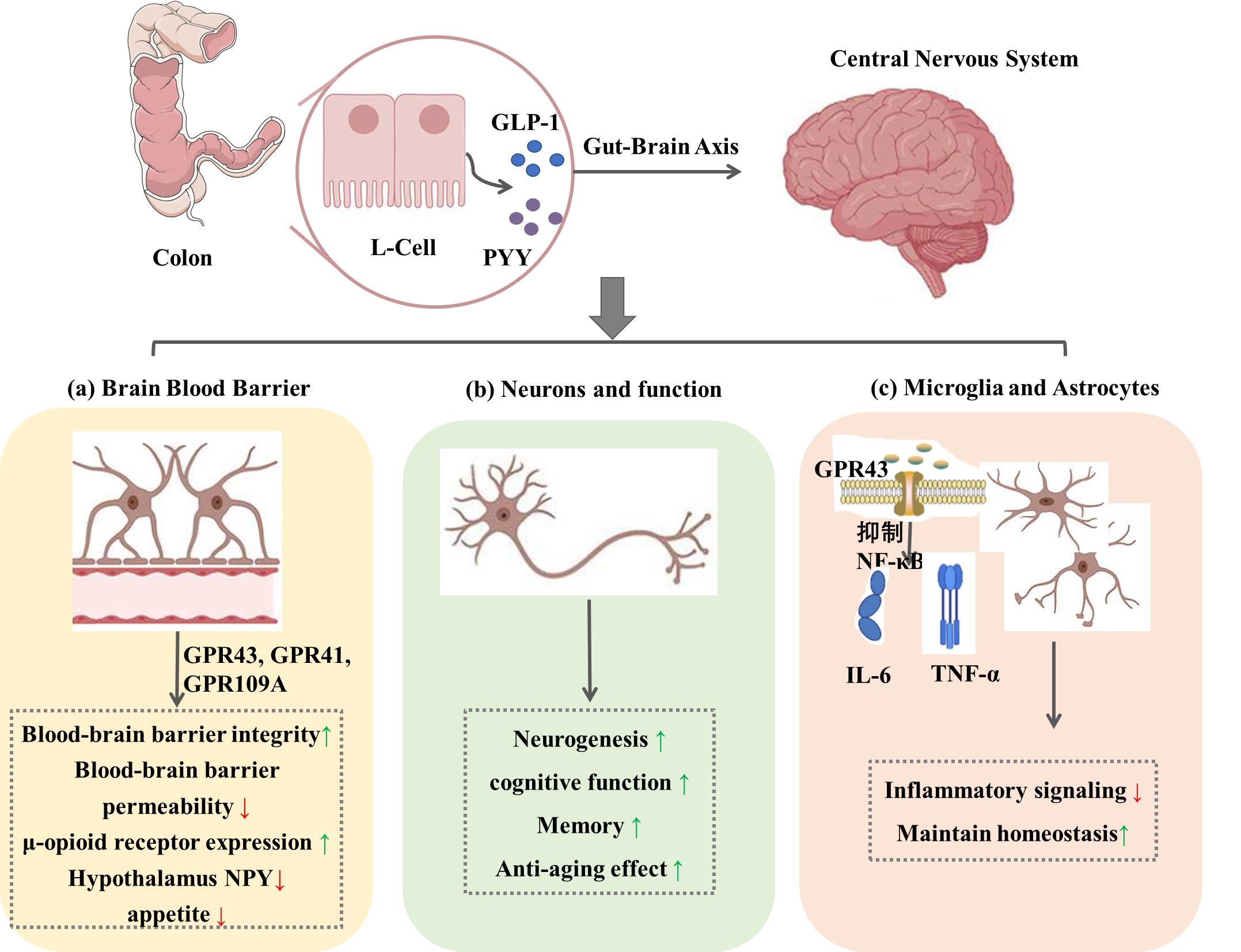
图4 高抗消化性淀粉对肠-脑轴的调控作用
Fig.4 Regulatory effects of resistant starch on the gut-brain axis. (a) Short-chain fatty acids can stimulate L cells to produce GLP-1, which acts on brain GLP-1 receptors to regulate appetite and glucose-lipid metabolism. SCFAs can also act on CPR41, GPR43, and GPR109A receptors to reduce blood-brain barrier permeability and maintain blood-brain barrier homeostasis; (b) SCFAs promote neurogenesis, which has a positive effect for improving cognition and memory; (c) SCFAs can affect the steady-state environment of glial cells and reduce oxidative stress and inflammatory responses.
| [1] | de Lima EP, Moretti RC Jr, Torres Pomini K, et al. Glycolipid metabolic disorders, metainflammation, oxidative stress, and cardiovascular diseases: unraveling pathways[J]. Biology, 2024, 13(7): 519. doi:10.3390/biology13070519 |
| [2] | Choi YJ, Kim GS, Chu SH, et al. Metabolic syndrome clustering patterns and the association with cardiovascular disease among post-menopausal Korean women[J]. Sci Rep, 2024, 14(1): 22702. doi:10.1038/s41598-024-72403-1 |
| [3] | Miao M, Hamaker BR. Food matrix effects for modulating starch bioavailability[J]. Annu Rev Food Sci Technol, 2021, 12: 169-91. doi:10.1146/annurev-food-070620-013937 |
| [4] | Lin YD, Zhang ZW, Wang SY, et al. Hypothalamus-pituitary-adrenal axis in glucolipid metabolic disorders[J]. Rev Endocr Metab Disord, 2020, 21(4): 421-9. doi:10.1007/s11154-020-09586-1 |
| [5] | Reddy CK, Suriya M, Vidya PV, et al. Synthesis and physico-chemical characterization of modified starches from banana (Musa AAB) and its biological activities in diabetic rats[J]. Int J Biol Macromol, 2017, 94: 500-7. doi:10.1016/j.ijbiomac.2016.10.050 |
| [6] | Bergeron N, Williams PT, Lamendella R, et al. Diets high in resistant starch increase plasma levels of trimethylamine-N-oxide, a gut microbiome metabolite associated with CVD risk[J]. Br J Nutr, 2016, 116(12): 2020-9. doi:10.1017/s0007114516004165 |
| [7] | Yin XX, Hu ZJ, Zheng YX, et al. Multi-scale structure characterization and in vivo digestion of parboiled rice[J]. Food Chem, 2023, 402: 134502. doi:10.1016/j.foodchem.2022.134502 |
| [8] | Awais M, Virk MS, Rehman A, et al. Emerging insights on the ultrasonication assisted modification of multi-scale structure-digestibility relationships in starches: a review[J]. Int J Biol Macromol, 2025, 317(Pt 1): 144759. doi:10.1016/j.ijbiomac.2025.144759 |
| [9] | Shi PW, Zhao YT, Qin F, et al. Understanding the multi-scale structure and physicochemical properties of millet starch with varied amylose content[J]. Food Chem, 2023, 410: 135422. doi:10.1016/j.foodchem.2023.135422 |
| [10] | Wu ZT, Qiao DL, Zhao SM, et al. Nonthermal physical modification of starch: an overview of recent research into structure and property alterations[J]. Int J Biol Macromol, 2022, 203: 153-75. doi:10.1016/j.ijbiomac.2022.01.103 |
| [11] | Zhang YJ, Mei XY, Li WQ, et al. Mechanisms of starchy foods glycemic index reduction under different means and their impacts on food sensory qualities: a review[J]. Food Chem, 2025, 467: 142351. doi:10.1016/j.foodchem.2024.142351 |
| [12] | Zhu RX, Li XX, Ren JL, et al. Manipulating starch crystalline structure for controlled digestibility and glycemic response[J]. Carbohydr Polym, 2026, 373: 124662. doi:10.1016/j.carbpol.2025.124662 |
| [13] | Meenu M, Xu BJ. A critical review on anti-diabetic and anti-obesity effects of dietary resistant starch[J]. Crit Rev Food Sci Nutr, 2019, 59(18): 3019-31. doi:10.1080/10408398.2018.1481360 |
| [14] | Wang ZG, Wang SL, Xu QH, et al. Synthesis and functions of resistant starch[J]. Adv Nutr, 2023, 14(5): 1131-44. doi:10.1016/j.advnut.2023.06.001 |
| [15] | Feng SJ, Wang YF, Cheng XJ, et al. Interaction of Lactic Acid Bacteria and Bifidobacterium with starch-lipid complex and effects on structure and digestibility of starch-lipid complex[J]. Int J Biol Macromol, 2026, 335(Pt 1): 149302. doi:10.1016/j.ijbiomac.2025.149302 |
| [16] | Livesey G, Taylor R, Livesey HF, et al. Dietary glycemic index and load and the risk of type 2 diabetes: a systematic review and updated meta-analyses of prospective cohort studies[J]. Nutrients, 2019, 11(6): 1280. doi:10.3390/nu11061280 |
| [17] | Ye SJ, Choi HW, Baik MY. A new physical starch modification method: Pressure annealing treatment (PAT)[J]. Int J Biol Macromol, 2026, 338(Pt 1): 149757. doi:10.1016/j.ijbiomac.2025.149757 |
| [18] | Lee CM, Jeong OY, Park HS, et al. Evaluation of glycemic response and starch digestibility in Korean rice toward the development of low GI rice[J]. Front Plant Sci, 2025, 16: 1724565. doi:10.3389/fpls.2025.1724565 |
| [19] | Zhang YG, He H, Feng SY, et al. Effect of grapefruit peel pectin on the structure, pasting characteristics, and in vitro digestibility of starch under different moisture content and temperature[J]. Int J Biol Macromol, 2025, 307(Pt 4): 142284. doi:10.1016/j.ijbiomac.2025.142284 |
| [20] | Wang JH, Huang JH, Liang Q, et al. Effects of heat-moisture treatment on structural characteristics and in vitro digestibility of A- and B-type wheat starch[J]. Int J Biol Macromol, 2024, 256(Pt 2): 128012. doi:10.1016/j.ijbiomac.2023.128012 |
| [21] | Liu K, Chi CD, Huang XY, et al. Synergistic effect of hydrothermal treatment and lauric acid complexation under different pressure on starch assembly and digestion behaviors[J]. Food Chem, 2019, 278: 560-7. doi:10.1016/j.foodchem.2018.11.097 |
| [22] | Liu T, Zhang BJ, Wang LL, et al. Microwave reheating increases the resistant starch content in cooked rice with high water contents[J]. Int J Biol Macromol, 2021, 184: 804-11. doi:10.1016/j.ijbiomac.2021.06.136 |
| [23] | Geng J, Luo Y, Liu LP, et al. Effect of extraction methods on Lyophyllum decastes crude polysaccharides for modifying wheat starch pasting, rheology, and in vitro digestibility[J]. Int J Biol Macromol, 2026, 335(Pt 1): 149328. doi:10.1016/j.ijbiomac.2025.149328 |
| [24] | Xu S, Gu ZB, Cheng L, et al. Regulation mechanisms and effects of thermal processing methods on starch digestibility in cornmeal staples[J]. Food Res Int, 2026, 226: 118192. doi:10.1016/j.foodres.2025.118192 |
| [25] | Cui LL, Wang XZ, Ge SH, et al. Mechanism of physicochemical changes in potato starch gel induced by citric acid-autoclaving for texture improvement and resistant starch formation[J]. Food Chem, 2026, 501: 147646. doi:10.1016/j.foodchem.2025.147646 |
| [26] | Wang X, Hao ZW, Liu NN, et al. Influence of the structure and physicochemical properties of OSA modified highland barley starch based on ball milling assisted treatment[J]. Int J Biol Macromol, 2024, 259: 129243. doi:10.1016/j.ijbiomac.2024.129243 |
| [27] | Liu K, Zu YY, Chi CD, et al. Modulation of the digestibility and multi-scale structure of cassava starch by controlling the cassava growth period[J]. Int J Biol Macromol, 2018, 120: 346-53. doi:10.1016/j.ijbiomac.2018.07.184 |
| [28] | Fan YT, Chang L, Yao Y, et al. Structural, physicochemical, and functional properties of waxy and non-waxy foxtail millet starches[J]. Foods, 2025, 14(17): 3034. doi:10.3390/foods14173034 |
| [29] | Seidelmann SB, Claggett B, Cheng SS, et al. Dietary carbohydrate intake and mortality: a prospective cohort study and meta-analysis[J]. Lancet Public Health, 2018, 3(9): e419-28. doi:10.1016/s2468-2667(18)30135-x |
| [30] | Liu YS, Wu QJ, Xia Y, et al. Carbohydrate intake and risk of metabolic syndrome: a dose-response meta-analysis of observational studies[J]. Nutr Metab Cardiovasc Dis, 2019, 29(12): 1288-98. doi:10.1016/j.numecd.2019.09.003 |
| [31] | Park SH, Lee KS, Park HY. Dietary carbohydrate intake is associated with cardiovascular disease risk in Korean: analysis of the third Korea National Health and Nutrition Examination Survey (KNHANES III)[J]. Int J Cardiol, 2010, 139(3): 234-40. doi:10.1016/j.ijcard.2008.10.011 |
| [32] | Zhao ML, Zhang QP, Lin Y, et al. The association between dietary carbohydrate intake and the risk of hyperlipidemia among reproductive-aged women in the US: a cross-sectional study[J]. PLoS One, 2024, 19(10): e0310184. doi:10.1371/journal.pone.0310184 |
| [33] | Ludwig DS, Aronne LJ, Astrup A, et al. The carbohydrate-insulin model: a physiological perspective on the obesity pandemic[J]. Am J Clin Nutr, 2021, 114(6): 1873-85. doi:10.1093/ajcn/nqab270 |
| [34] | Yin HT, Zhou YQ, Zhou YJ, et al. Retrospective analysis of omega-3 fatty acids and the DASH diet in hyperlipidemia and hypertension management among obese individuals[J]. Front Nutr, 2025, 12: 1704552. doi:10.3389/fnut.2025.1704552 |
| [35] | Zheng LY, Zeng AQ, Liu L, et al. Metabolic syndrome: molecular mechanisms and therapeutic interventions[J]. Mol Biomed, 2025, 6(1): 59. doi:10.1186/s43556-025-00303-5 |
| [36] | Zhang Y, Zhang WQ, Tang WW, et al. The prevalence of obesity-related hypertension among middle-aged and older adults in China[J]. Front Public Health, 2022, 10: 865870. doi:10.3389/fpubh.2022.865870 |
| [37] | Lu XQ, Jin YX, Li DX, et al. Multidisciplinary progress in obesity research[J]. Genes, 2022, 13(10): 1772. doi:10.3390/genes13101772 |
| [38] | Fu QS, Li TT, Zhang C, et al. Butyrate mitigates metabolic dysfunctions via the ERα-AMPK pathway in muscle in OVX mice with diet-induced obesity[J]. Cell Commun Signal, 2023, 21(1): 95. doi:10.1186/s12964-023-01119-y |
| [39] | Hong J, Jia YM, Pan SF, et al. Butyrate alleviates high fat diet-induced obesity through activation of adiponectin-mediated pathway and stimulation of mitochondrial function in the skeletal muscle of mice[J]. Oncotarget, 2016, 7(35): 56071-82. doi:10.18632/oncotarget.11267 |
| [40] | den Besten G, Bleeker A, Gerding A, et al. Short-chain fatty acids protect against high-fat diet-induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation[J]. Diabetes, 2015, 64(7): 2398-408. doi:10.2337/db14-1213 |
| [41] | Chen BR, Guan LZ, Wu C, et al. Gut microbiota-butyrate-PPARγ axis modulates adipose regulatory T cell population[J]. Adv Sci, 2025, 12(20): 2411086. doi:10.1002/advs.202570151 |
| [42] | Zhang L, Ouyang Y, Li HT, et al. Metabolic phenotypes and the gut microbiota in response to dietary resistant starch type 2 in normal-weight subjects: a randomized crossover trial[J]. Sci Rep, 2019, 9(1): 4736. doi:10.1038/s41598-018-38216-9 |
| [43] | Higgins JA. Resistant starch and energy balance: impact on weight loss and maintenance[J]. Crit Rev Food Sci Nutr, 2014, 54(9): 1158-66. doi:10.1080/10408398.2011.629352 |
| [44] | Wang D, Liu CD, Li HF, et al. LSD1 mediates microbial metabolite butyrate-induced thermogenesis in brown and white adipose tissue[J]. Metabolism, 2020, 102: 154011. doi:10.1016/j.metabol.2019.154011 |
| [45] | Jia YM, Hong J, Li HF, et al. Butyrate stimulates adipose lipolysis and mitochondrial oxidative phosphorylation through histone hyperacetylation-associated β(3)‑adrenergic receptor activation in high-fat diet-induced obese mice[J]. Exp Physiol, 2017, 102(2): 273-81. doi:10.1113/ep086114 |
| [46] | Li HT, Zhang L, Li J, et al. Resistant starch intake facilitates weight loss in humans by reshaping the gut microbiota[J]. Nat Metab, 2024, 6(3): 578-97. doi:10.1038/s42255-024-00988-y |
| [47] | Mollica MP, Mattace Raso G, Cavaliere G, et al. Butyrate regulates liver mitochondrial function, efficiency, and dynamics in insulin-resistant obese mice[J]. Diabetes, 2017, 66(5): 1405-18. doi:10.2337/db16-0924 |
| [48] | Sun B, Jia YM, Hong J, et al. Sodium butyrate ameliorates high-fat-diet-induced non-alcoholic fatty liver disease through peroxisome proliferator-activated receptor α-mediated activation of β oxidation and suppression of inflammation[J]. J Agric Food Chem, 2018, 66(29): 7633-42. doi:10.1021/acs.jafc.8b01189 |
| [49] | Zhang JM, Sun YS, Zhao LQ, et al. SCFAs-induced GLP-1 secretion links the regulation of gut microbiome on hepatic lipogenesis in chickens[J]. Front Microbiol, 2019, 10: 2176. doi:10.3389/fmicb.2019.02176 |
| [50] | Zhou D, Chen YW, Zhao ZH, et al. Sodium butyrate reduces high-fat diet-induced non-alcoholic steatohepatitis through upregulation of hepatic GLP-1R expression[J]. Exp Mol Med, 2018, 50(12): 157. doi:10.1038/s12276-018-0183-1 |
| [51] | Wang R, Luo SM, Wang P, et al. Estimating the burden of adult diabetes in China: a systematic review and meta-analysis with attributable risk assessment[J]. BMC Public Health, 2025, 25(1): 3734. doi:10.1186/s12889-025-24626-8 |
| [52] | Kabisch S, Hajir J, Sukhobaevskaia V, et al. Impact of dietary fiber on inflammation in humans[J]. Int J Mol Sci, 2025, 26(5): 2000. doi:10.3390/ijms26052000 |
| [53] | Hughes RL, Horn WF, Wen A, et al. Resistant starch wheat increases PYY and decreases GIP but has no effect on self-reported perceptions of satiety[J]. Appetite, 2022, 168: 105802. doi:10.1016/j.appet.2021.105802 |
| [54] | Hughes RL, Horn WH, Finnegan P, et al. Resistant starch type 2 from wheat reduces postprandial glycemic response with concurrent alterations in gut microbiota composition[J]. Nutrients, 2021, 13(2): 645. doi:10.3390/nu13020645 |
| [55] | Meng Y, Bai H, Yu QT, et al. High-resistant starch, low-protein flour intervention on patients with early type 2 diabetic nephropathy: a randomized trial[J]. J Ren Nutr, 2019, 29(5): 386-93. doi:10.1053/j.jrn.2018.12.005 |
| [56] | Belobrajdic DP, King RA, Christophersen CT, et al. Dietary resistant starch dose-dependently reduces adiposity in obesity-prone and obesity-resistant male rats[J]. Nutr Metab, 2012, 9(1): 93. doi:10.1186/1743-7075-9-93 |
| [57] | Husted AS, Trauelsen M, Rudenko O, et al. GPCR-mediated signaling of metabolites[J]. Cell Metab, 2017, 25(4): 777-96. doi:10.1016/j.cmet.2017.03.008 |
| [58] | Roshanravan N, Mahdavi R, Alizadeh E, et al. Effect of butyrate and inulin supplementation on glycemic status, lipid profile and glucagon-like peptide 1 level in patients with type 2 diabetes: a randomized double-blind, placebo-controlled trial[J]. Horm Metab Res, 2017, 49(11): 886-91. doi:10.1055/s-0043-119089 |
| [59] | Arora T, Tremaroli V. Therapeutic potential of butyrate for treatment of type 2 diabetes[J]. Front Endocrinol, 2021, 12: 761834. doi:10.3389/fendo.2021.761834 |
| [60] | Mihaylova MM, Vasquez DS, Ravnskjaer K, et al. Class IIa histone deacetylases are hormone-activated regulators of FOXO and mammalian glucose homeostasis[J]. Cell, 2011, 145(4): 607-21. doi:10.1016/j.cell.2011.03.043 |
| [61] | Su SY, Wang WY, Lin ZN, et al. Circulating ceramides and sphingomyelins: key biomarkers and targets in cardiovascular prevention[J]. Sci China Life Sci, 2026. DOI:10.1007/s11427-025-3050-y . |
| [62] | Du Y, Li XX, Su CY, et al. Butyrate protects against high-fat diet-induced atherosclerosis via up-regulating ABCA1 expression in apolipoprotein E-deficiency mice[J]. Br J Pharmacol, 2020, 177(8): 1754-72. doi:10.1111/bph.14933 |
| [63] | Haghikia A, Zimmermann F, Schumann P, et al. Propionate attenuates atherosclerosis by immune-dependent regulation of intestinal cholesterol metabolism[J]. Eur Heart J, 2022, 43(6): 518-33. doi:10.1093/eurheartj/ehab644 |
| [64] | González-Correa C, Moleón J, Miñano S, et al. Protective effect of dietary fiber on blood pressure and vascular dysfunction through regulation of sympathetic tone and immune response in genetic hypertension[J]. Phytother Res, 2025, 39(4): 1858-75. doi:10.1002/ptr.8484 |
| [65] | Marx N, Federici M, Schütt K, et al. 2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes[J]. Eur Heart J, 2023, 44(39): 4043-140. |
| [66] | Hu TT, Wu QQ, Yao Q, et al. Short-chain fatty acid metabolism and multiple effects on cardiovascular diseases[J]. Ageing Res Rev, 2022, 81: 101706. doi:10.1016/j.arr.2022.101706 |
| [67] | Moleón J, González-Correa C, Miñano S, et al. Protective effect of microbiota-derived short chain fatty acids on vascular dysfunction in mice with systemic lupus erythematosus induced by toll like receptor 7 activation[J]. Pharmacol Res, 2023, 198: 106997. doi:10.1016/j.phrs.2023.106997 |
| [68] | Hsu CN, Hou CY, Lee CT, et al. Maternal 3, 3-dimethyl-1-butanol therapy protects adult male rat offspring against hypertension programmed by perinatal TCDD exposure[J]. Nutrients, 2021, 13(9): 3041. doi:10.3390/nu13093041 |
| [69] | Miletta MC, Petkovic V, Eblé A, et al. Butyrate increases intracellular calcium levels and enhances growth hormone release from rat anterior pituitary cells via the G-protein-coupled receptors GPR41 and 43[J]. PLoS One, 2014, 9(10): e107388. doi:10.1371/journal.pone.0107388 |
| [70] | Xu YJ, Wei SJ, Zhu LY, et al. Low expression of the intestinal metabolite butyric acid and the corresponding memory pattern regulate HDAC4 to promote apoptosis in rat hippocampal neurons[J]. Ecotoxicol Environ Saf, 2023, 253: 114660. doi:10.1016/j.ecoenv.2023.114660 |
| [71] | Kadyan S, Park G, Hochuli N, et al. Resistant starches from dietary pulses improve neurocognitive health via gut-microbiome-brain axis in aged mice[J]. Front Nutr, 2024, 11: 1322201. doi:10.3389/fnut.2024.1322201 |
| [72] | Fock E, Parnova R. Mechanisms of blood-brain barrier protection by microbiota-derived short-chain fatty acids[J]. Cells, 2023, 12(4): 657. doi:10.3390/cells12040657 |
| [73] | Dicks LMT. Key signals produced by gut microbiota associated with metabolic syndrome, cancer, cardiovascular diseases, and brain functions[J]. Int J Mol Sci, 2025, 26(21): 10539. doi:10.3390/ijms262110539 |
| [74] | Guedes Lúcio H, Grancieri M, David Medina Martinez O, et al. Dry heat whole Sorghum BRS 305 flour modulate satiety and improves antioxidant response in brain of Wistar rats fed with high-fat high-fructose diet[J]. Food Res Int, 2023, 173(Pt 1): 113252. doi:10.1016/j.foodres.2023.113252 |
| [75] | Zhang S, Zeng L, Ma J, et al. Gut Prevotellaceae-GABAergic septohippocampal pathway mediates spatial memory impairment in high-fat diet-fed ovariectomized mice[J]. Neurobiol Dis, 2023, 177: 105993. doi:10.1016/j.nbd.2023.105993 |
| [76] | Chen WQ, Cai WK, Hoover B, et al. Insulin action in the brain: cell types, circuits, and diseases[J]. Trends Neurosci, 2022, 45(5): 384-400. doi:10.1016/j.tins.2022.03.001 |
| [77] | He QK, Wang XY, Hu W, et al. Therapeutic potential of Canna edulis RS3-resistant starch in alleviating neuroinflammation and apoptosis in a Parkinson's disease rat model[J]. Heliyon, 2024, 10(18): e38072. doi:10.1016/j.heliyon.2024.e38072 |
| [78] | Lian ZS, Xu Y, Wang C, et al. Gut microbiota-derived melatonin from Puerariae Lobatae Radix-resistant starch supplementation attenuates ischemic stroke injury via a positive microbial co-occurrence pattern[J]. Pharmacol Res, 2023, 190: 106714. doi:10.1016/j.phrs.2023.106714 |
| [79] | Wang J, He LQ, Meng WC, et al. Cooperative complexation of genistein and lauric acid with starch and its impact on starch digestibility[J]. Int J Biol Macromol, 2025, 310(Pt 3): 143318. doi:10.1016/j.ijbiomac.2025.143318 |
| [1] | 姜思捷, 宫澜, 郑华. 嗜粘蛋白阿克曼氏菌与心血管及代谢性疾病相关性的研究进展[J]. 南方医科大学学报, 2026, 46(2): 473-478. |
| [2] | 张淑芬, 黄添容, 杨灿洪, 陈家镒, 吕田明, 张嘉发. 莱菔硫烷通过抑制Aβ42寡聚体激活的U87细胞中MAPK/NF-κB信号通路降低反应性星形胶质细胞介导的SH-SY5Y凋亡[J]. 南方医科大学学报, 2026, 46(1): 191-199. |
| [3] | 李泽涵, 梁萌, 韩根成, 张学武. 菊淀粉型巴戟天寡糖降低肺炎链球菌脑膜炎小鼠的症状评分和死亡率[J]. 南方医科大学学报, 2025, 45(3): 577-586. |
| [4] | 崔佳宁, 李润霞, 刘学谦, 赵亚男, 张兴华, 刘清波, 李 涛. 心脏磁共振特征追踪技术可评估心肌淀粉样变性患者的心脏功能[J]. 南方医科大学学报, 2023, 43(4): 660-666. |
| [5] | 张嘉发, 杨灿洪, 张淑芬, 曹婷婷, 彭 瑞, 郭蔚泓, 严予苹, 谢淑婷, 彭晓佳, 吕田明, 黄添容. 莱菔硫烷通过下调MAPK/NF-κB信号通路逆转Aβ纤维介导的M1型小胶质细胞极化和神经炎症介导的神经干细胞程序性坏死[J]. 南方医科大学学报, 2023, 43(12): 2132-2138. |
| [6] | 韩 拓, 苑 昊, 叶琳樱, 江 旭, 白 亮, 王聪霞. COVID-19与心血管代谢性疾病的临床研究进展[J]. 南方医科大学学报, 2021, 41(4): 628-632. |
| [7] | 李 智, 李 虎, 姚尚龙, 程明华, 陈建颜. 右美托咪定剂量对老年脊柱手术患者术后认知功能障碍发生率及Aβ和细胞因子水平的影响:120例随机对照试验[J]. 南方医科大学学报, 2021, 41(4): 600-606. |
| [8] | 张雪卫, 傅 强. 脑脊液中β淀粉样蛋白42和神经丝轻链蛋白水平与术后神经认知功能障碍的相关性:基于90例66~78岁患者[J]. 南方医科大学学报, 2021, 41(4): 574-578. |
| [9] | 江颖仪, 李德宪, 劳穗华, 肖 芃. 血清淀粉样蛋白A、降钙素原、超敏C反应蛋白检测对肺结核合并肺炎的诊断价值[J]. 南方医科大学学报, 2021, 41(3): 453-458. |
| [10] | 蓝燕,杨梓超,刘涵,程宏彦,刘叔文,谭穗懿. PSB0739抑制精液来源的淀粉样纤维形成[J]. 南方医科大学学报, 2018, 38(11): 1338-. |
| [11] | 李锦清,宋亚丽,寻添荣,谭穗懿,刘叔文. 乳酸抑制精液来源淀粉样纤维的形成[J]. 南方医科大学学报, 2017, 37(07): 907-. |
| [12] | 吕田明,梁彦珊,史翠丽,黄小玉,潘志勇,凌容,张文玲,郝梦淅,郭秋瑶,曾宇,尹恝. 阿尔茨海默氏病大鼠淀粉样变性脑血管病的病理学变化[J]. 南方医科大学学报, 2014, 34(07): 969-. |
| [13] | 张璐,王叶,程流泉,王晶,周肖,刘淼,张威,章明,张波,智光. 左室长轴收缩期峰值应变在原发性心脏淀粉样变性与肥厚型心肌病诊断中的价值[J]. 南方医科大学学报, 2014, 34(05): 609-. |
| [14] | 朱丽娟,陈筱山,何选丽,戚韵雯,晏勇. 外源性硫化氢对高糖条件下原代神经元β淀粉样蛋白的影响[J]. 南方医科大学学报, 2014, 34(04): 504-. |
| [15] | 邓炎尧,侯德仁,田密,李维,奉夏露. AD模型小鼠海马和小脑Aβ的沉积与相关miRNAs表达的变化[J]. 南方医科大学学报, 2014, 34(03): 323-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||