南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 861-870.doi: 10.12122/j.issn.1673-4254.2026.04.14
• • 上一篇
邓子豪1( ), 王转1(
), 王转1( ), 蒙森雄2, 杨玉生1, 杨运平1(
), 蒙森雄2, 杨玉生1, 杨运平1( ), 范刘奕1(
), 范刘奕1( ), 王磊1(
), 王磊1( )
)
收稿日期:2025-11-09
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
杨运平,范刘奕,王磊
E-mail:2313588093@qq.com;1113014681@qq.com;yangyp2012@126.com;378280282@qq.com;conan213@smu.edu.cn
作者简介:邓子豪,在读硕士研究生,E-mail: 2313588093@qq.com基金资助:
Zihao DENG1( ), Zhuan WANG1(
), Zhuan WANG1( ), Senxiong MENG2, Yusheng YANG1, Yunping YANG1(
), Senxiong MENG2, Yusheng YANG1, Yunping YANG1( ), Liuyi FAN1(
), Liuyi FAN1( ), Lei WANG1(
), Lei WANG1( )
)
Received:2025-11-09
Online:2026-04-20
Published:2026-04-24
Contact:
Yunping YANG, Liuyi FAN, Lei WANG
E-mail:2313588093@qq.com;1113014681@qq.com;yangyp2012@126.com;378280282@qq.com;conan213@smu.edu.cn
Supported by:摘要:
目的 验证细胞衰老在糖皮质激素性骨质疏松中的作用,探究金天格胶囊治疗糖皮质激素性骨质疏松的机制。 方法 复苏人脐静脉内皮细胞(HUVEC)和小鼠胚胎前成骨细胞(MC3T3),诱导生成破骨细胞,将上述细胞均分为对照组(CN组)、模型组(MPS组)和金天格处理组(MPS+JTG组),使用稀释后的生理盐水溶解甲基强的松龙和金天格胶囊,待细胞贴壁后分别使用相应的药物处理对应组别的细胞。通过蛋白印迹、SA-β-Gal染色、DCFH-DA法染色、划痕实验和RT-PCR检测HUVEC细胞的衰老通路、修复能力;通过免疫荧光染色、ALP染色、Alizarin Red S染色和RT-PCR检测MC3T3细胞成骨能力;通过TRAP染色检测破骨细胞分化程度。将30只3周龄SPF级雌性SD大鼠随机分为对照组(CN组)、模型组(MPS组)和金天格处理组(MPS+ JTG组),10只/组。建立糖皮质激素性骨质疏松模型并连续灌胃给药3个月。人道主义处死大鼠后取股骨及静脉血,进行micro-CT扫描、检测大鼠股骨骨体积分数变化情况,并检测大鼠血清骨代谢指标。 结果 网络药理学结果显示,虎骨有效成分与糖皮质激素性骨质疏松存在大量交集靶点,并与P53等细胞衰老相关靶点具有较强的结合力。在细胞水平,与模型组相比,在金天格处理后的HUVEC细胞模型中,P53、P21等衰老蛋白表达被抑制(P<0.01)、氧化应激减弱(P<0.01),而细胞迁移、成血管能力增强(P<0.05)。同样,金天格处理组的MC3T3细胞成骨和矿化能力也明显增强(P<0.001),并且抑制了破骨细胞的分化。在动物水平,经过14 d MPS腹腔注射的大鼠股骨骨量较对照组明显丢失,而金天格胶囊可以缓解骨量的丢失及血清骨代谢指标紊乱(P<0.05)。 结论 金天格胶囊可以通过抑制HUVEC细胞的衰老、增强MC3T3细胞的成骨能力并抑制破骨,促进骨髓内相关血管的迁移和生成,改善成骨能力和骨骼血供并促进成骨、抑制破骨,从而缓解糖皮质激素性骨质疏松。
邓子豪, 王转, 蒙森雄, 杨玉生, 杨运平, 范刘奕, 王磊. 金天格胶囊通过抑制血管内皮细胞衰老并促进骨形成以缓解大鼠糖皮质激素性骨质疏松[J]. 南方医科大学学报, 2026, 46(4): 861-870.
Zihao DENG, Zhuan WANG, Senxiong MENG, Yusheng YANG, Yunping YANG, Liuyi FAN, Lei WANG. Jintiange Capsule alleviates glucocorticoid-induced osteoporosis in rats by inhibiting vascular endothelial cell senescence and promoting bone formation[J]. Journal of Southern Medical University, 2026, 46(4): 861-870.
| Gene | Primer sequences 5'-3' |
|---|---|
| P16-forward | ACCAGAGGCAGTAACCATGC |
| P16-reverse | GTGAAAAGGCAGAAGCGGTG |
| P21-forward | AGTCAGTTCCTTGTGGAGCC |
| P21-reverse | AGGAGAACACGGGATGAGGA |
| PLNB-forward | AGACTCTCCCAGCAACAAGC |
| PLNB-reverse | CTCTACTGCATCCTGAGGCG |
| ANG-forward | AGATTCTTCCTCCTGGGAGCC |
| ANG-reverse | CCAGCACGAAGACCAACAAC |
| BMP-2-forward | ACTCGAAATTCCCCGTGACC |
| BMP-2-reverse | CCACTTCCACCACGAATCCA |
| OCN-forward | ATGAGAGCCCTCACACTCCT |
| OCN-reverse | CTTGGACACAAAGGCTGCAC |
| RUNX2-forward | CGCCTCACAAACAACCACAG |
| RUNX2-reverse | TCACTGTGCTGAAGAGGCTG |
表1 MC3T3细胞成骨及HUVEC细胞衰老相关引物序列
Tab.1 Primer sequences for amplification of genes related with MC3T3 osteogenic differentiation and HUVEC cellular senescence
| Gene | Primer sequences 5'-3' |
|---|---|
| P16-forward | ACCAGAGGCAGTAACCATGC |
| P16-reverse | GTGAAAAGGCAGAAGCGGTG |
| P21-forward | AGTCAGTTCCTTGTGGAGCC |
| P21-reverse | AGGAGAACACGGGATGAGGA |
| PLNB-forward | AGACTCTCCCAGCAACAAGC |
| PLNB-reverse | CTCTACTGCATCCTGAGGCG |
| ANG-forward | AGATTCTTCCTCCTGGGAGCC |
| ANG-reverse | CCAGCACGAAGACCAACAAC |
| BMP-2-forward | ACTCGAAATTCCCCGTGACC |
| BMP-2-reverse | CCACTTCCACCACGAATCCA |
| OCN-forward | ATGAGAGCCCTCACACTCCT |
| OCN-reverse | CTTGGACACAAAGGCTGCAC |
| RUNX2-forward | CGCCTCACAAACAACCACAG |
| RUNX2-reverse | TCACTGTGCTGAAGAGGCTG |
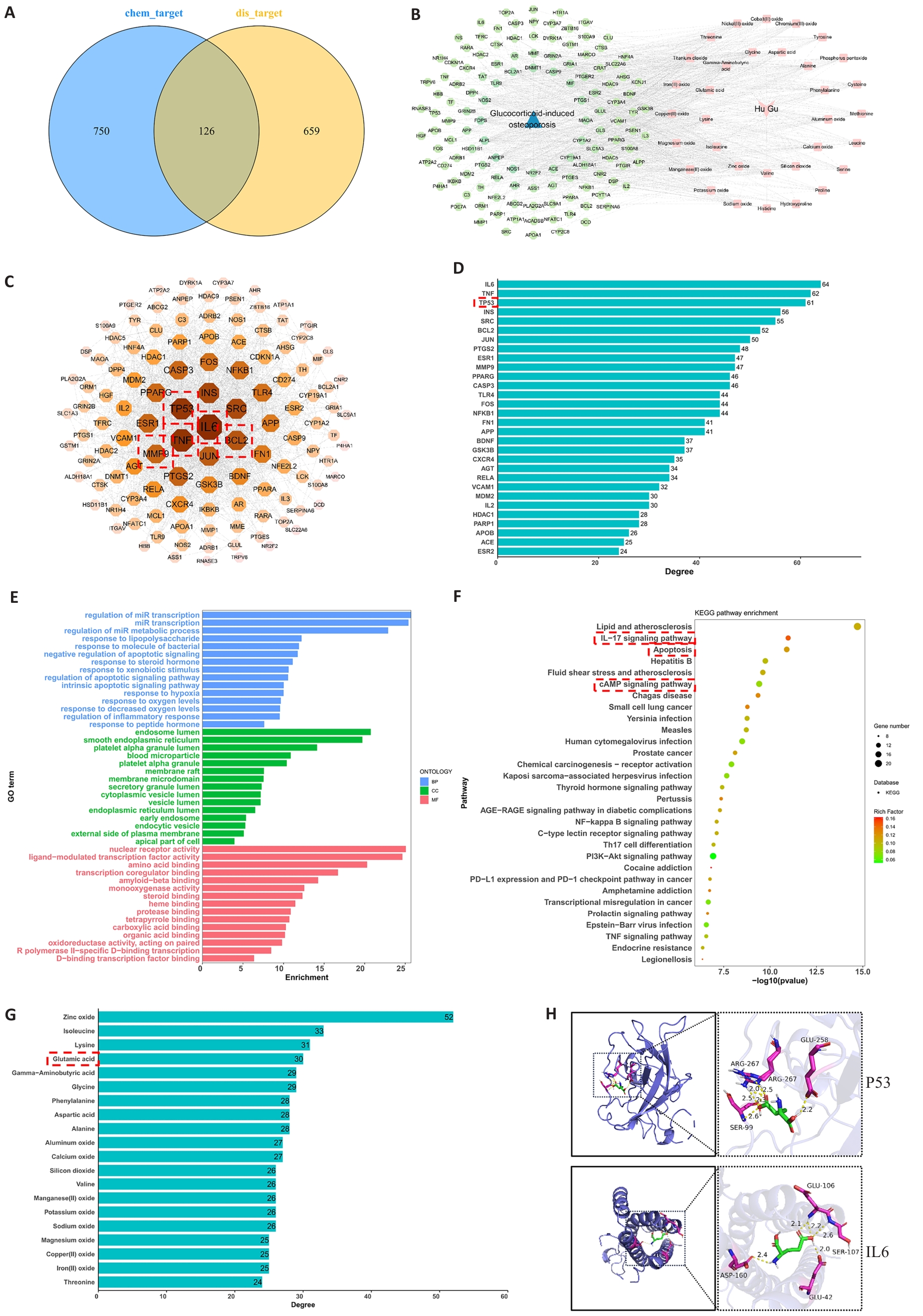
图1 虎骨与GIO之间可能存在的潜在治疗靶向作用
Fig.1 Potential therapeutic targeting mechanism between HU and glucocorticoid-induced osteoporosis (GIO). A: Collection of GIO-related targets and screening of potential therapeutic targets. B: Construction and analysis of the active TB components-targets-GIO network. C: Protein-protein interaction (PPI) network of overlapping targets between TB active components and GIO treatment. D: Bar graph showing connectivity ranking of the core targets. E: Gene Ontology (GO) enrichment analysis. F: Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis. G: Compound connectivity ranking of active ingredients in Hu Gu. H: Molecular docking results of glutamic acid (a key active component of TB) with P53 and IL6.
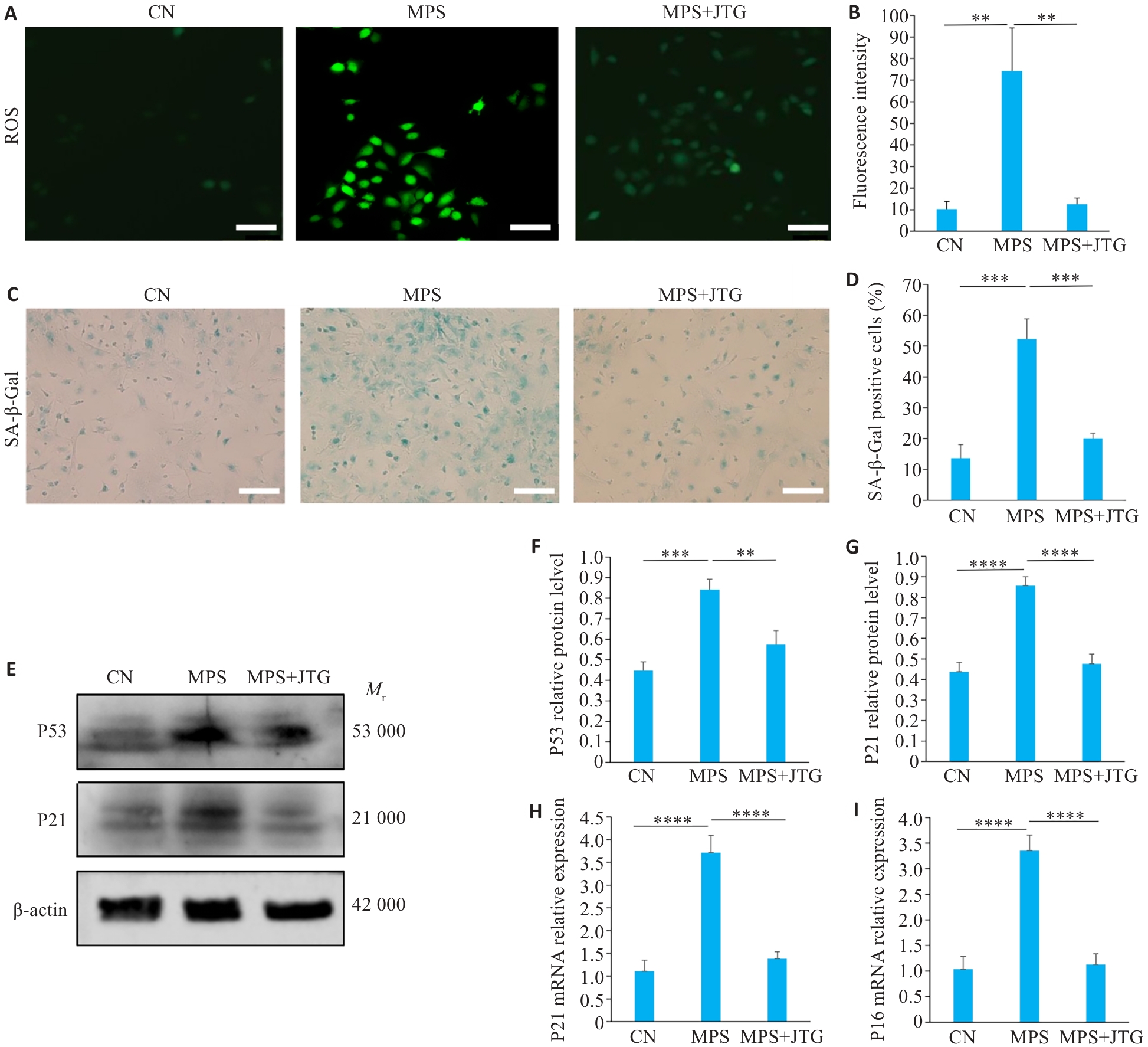
图2 金天格胶囊对血管内皮细胞氧化应激及衰老的作用
Fig.2 Effects of Jintiange (JTG) Capsule on oxidative stress and senescence in vascular endothelial cells. A, B: DCFH-DA staining for intracellular ROS detection (Original magnification: ×20). C, D: Senescence-associated β-galactosidase (SA-β-gal) staining for assessing the number of senescent cells in the 3 groups (×10). E-G: Western blotting for detecting P53 and P21 protein expression levels in the 3 groups. H, I: Quantitative PCR analysis of p16 and p21 mRNA expressions in the 3 groups. **P<0.01, ***P<0.001, ****P<0.0001.
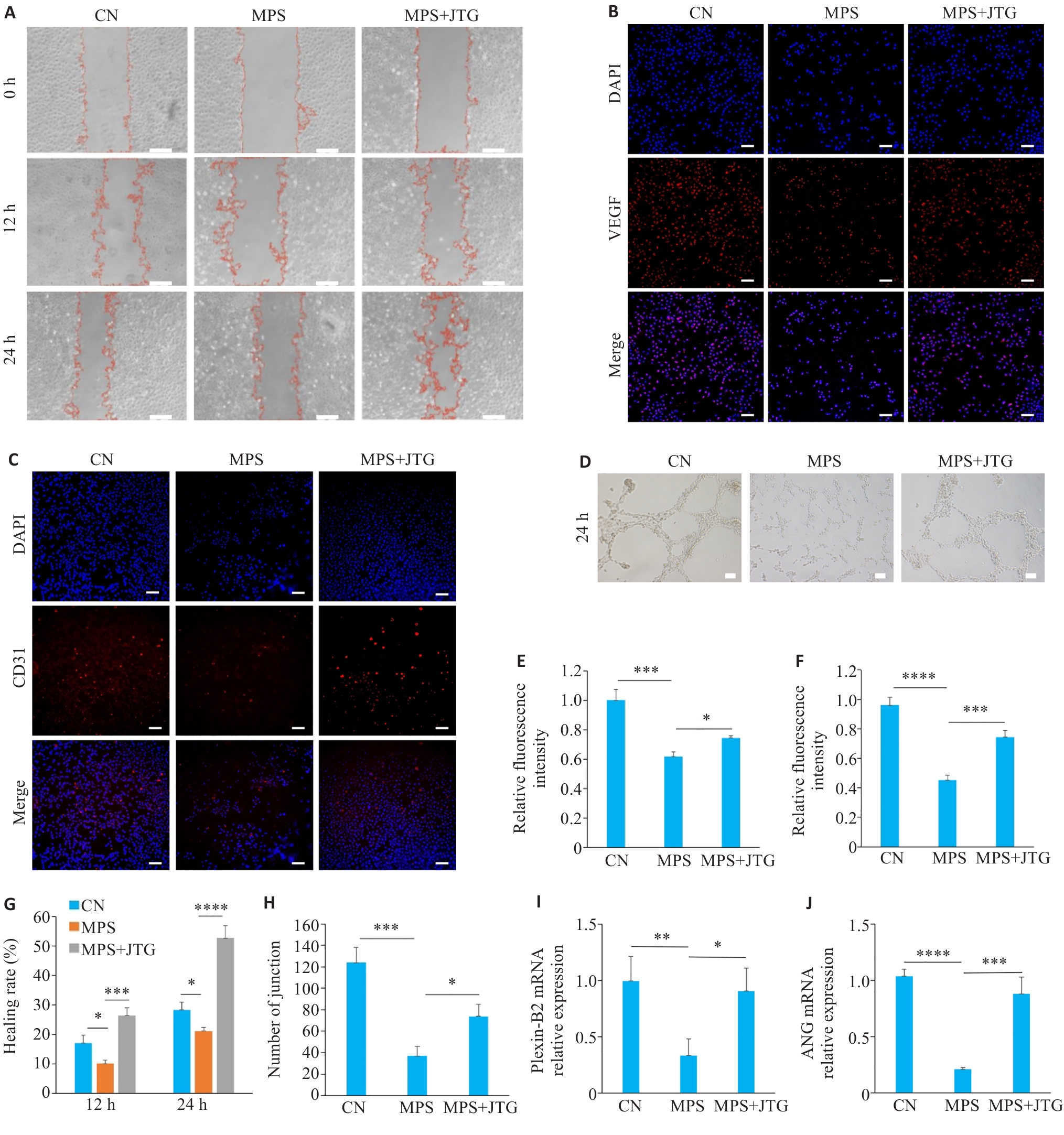
图3 金天格胶囊对血管内皮细胞增殖及成血管作用的影响
Fig.3 Effects of JTG Capsule on proliferation and angiogenic activity of vascular endothelial cells. A, G: Scratch assay of HUVECs with different treatments for analyzing cell migration and proliferation. B, E: VEGF immunofluorescence staining in HUVECs in the 3 groups (×10). C, F: CD31 immunofluorescence staining in HUVECs with different treatments (×10). D, H: Tube formation assay of HUVECs cultured in conditioned media from each group for 24 h (×20). I, J: qPCR for detecting mRNA expression levels of angiogenesis-related markers Plexin-B2 (I) and ANG (J) in HUVECs. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
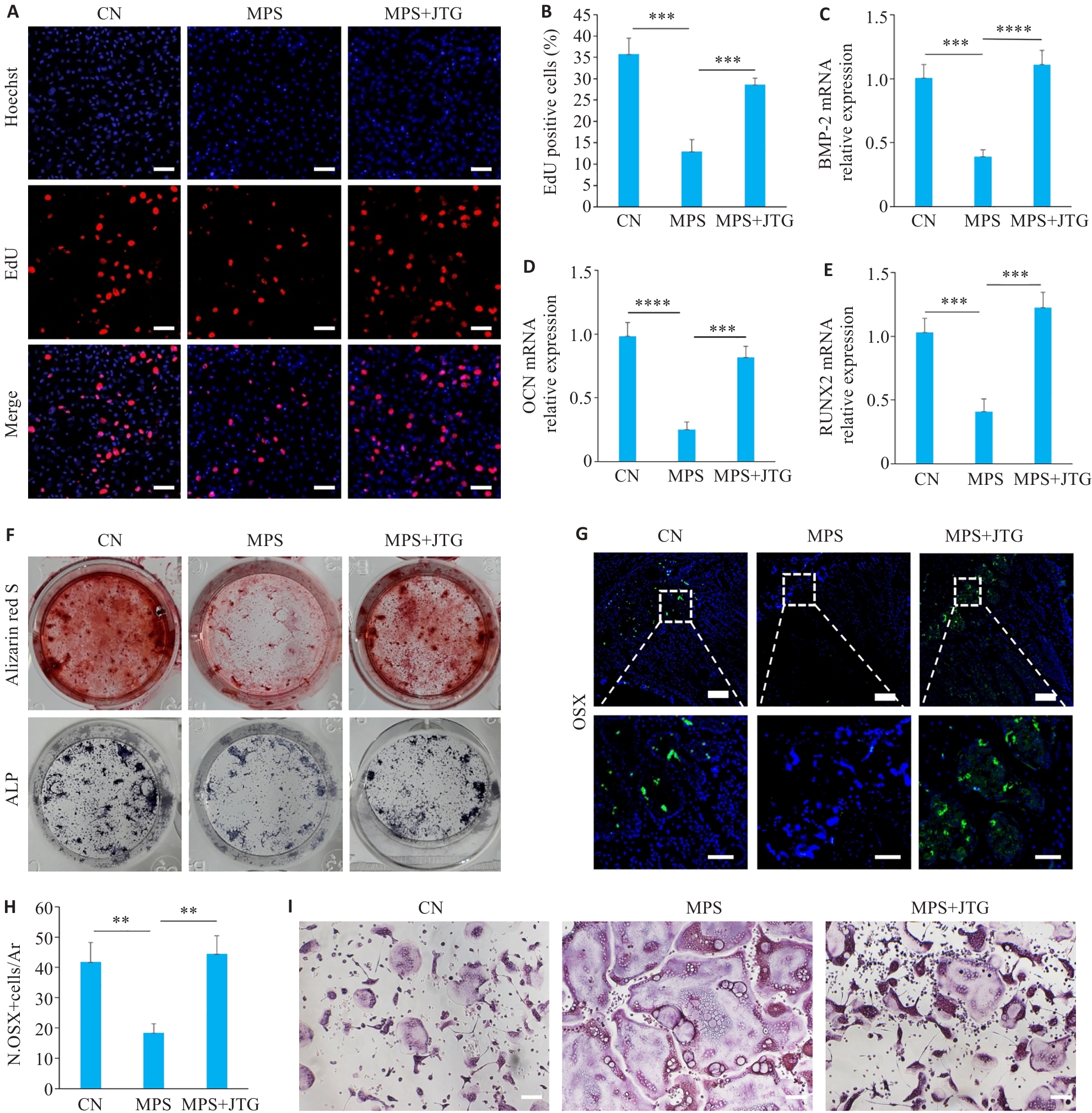
图4 金天格胶囊对成骨细胞及破骨细胞的影响
Fig.4 Effects of JTG Capsule on osteoblasts and osteoclasts. A, B: EdU labeling of MC3T3 cells with different treatment for analysis of cell proliferation (×10). C-E: Results of qPCR for detecting mRNA expressions of osteogenesis-related genes BMP-2 (C), OCN (D), and RUNX2 (E) in MC3T3 cells in the 3 groups. F: Osteogenic induction of MC3T3 cells in the 3 groups followed by alkaline phosphatase (ALP) staining and Alizarin Red S staining. G, H: Immunofluorescence staining of Osterix in MC3T3 cells with different treatments (×10). I: TRAP staining of the osteoclasts in the 3 treatment groups (×20). **P<0.01, ***P<0.001, ****P<0.0001.
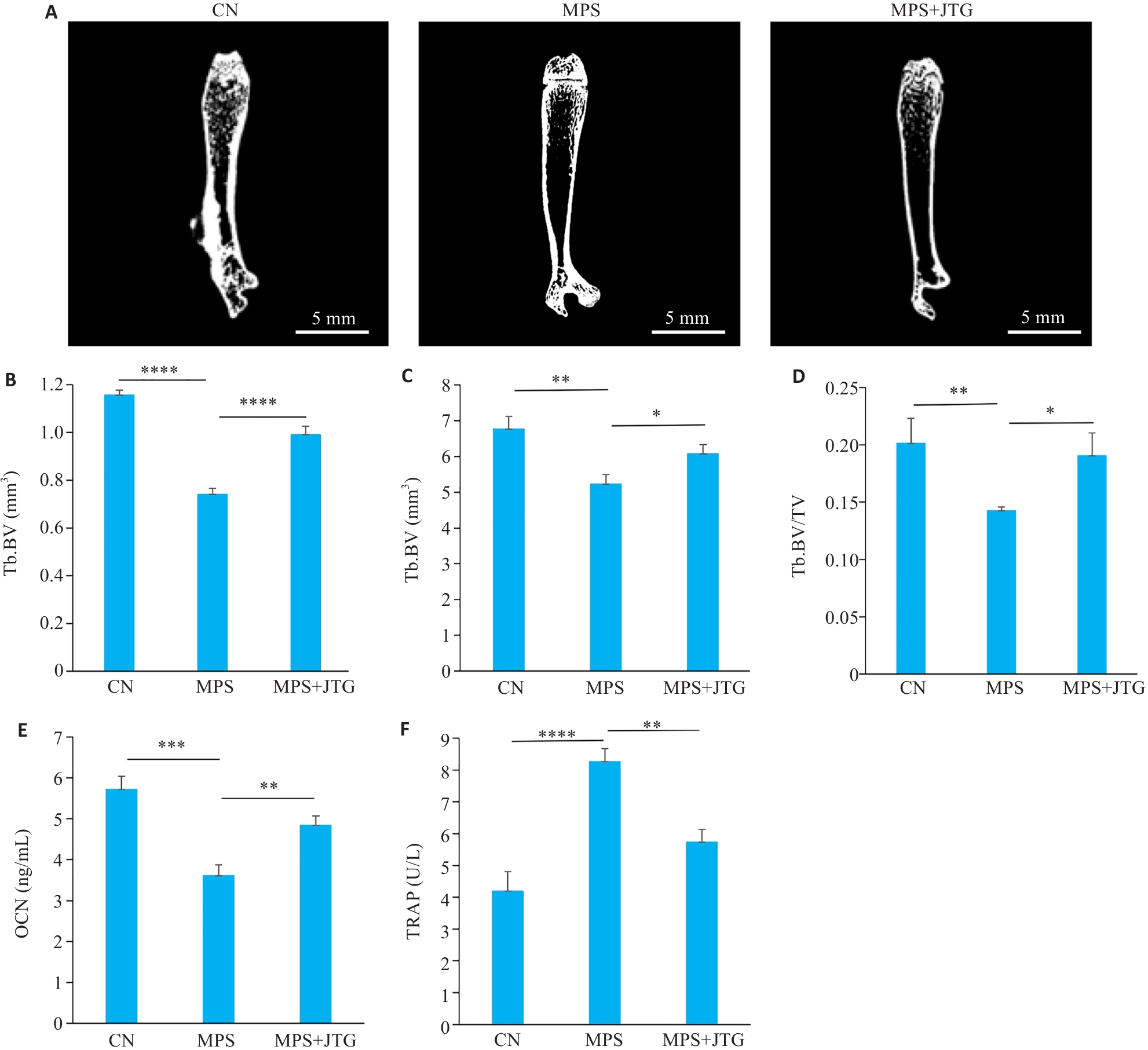
图5 金天格胶囊在体外实验中对GIO患病大鼠的缓解作用
Fig.5 Therapeutic effects of JTG Capsule on glucocorticoid-induced osteoporosis (GIO) in rats. A: Micro-CT scanning of the femoral bones from rats in the 3 treatment groups. B-D: Quantitative analysis of micro-CT scans for measurement of bone volume (BV) (B), tissue volume (TV) (C), and BV/TV ratio (D). E, F: Serum levels of osteocalcin (OCN) and tartrate-resistant acid phosphatase 5b (TRAP-5b) (F) in GIO rats. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
| [1] | Humphrey MB, Russell L, Danila MI, et al. 2022 American college of rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis[J]. Arthritis Rheum, 2023, 75(12): 2088-102. doi:10.1002/art.42646 |
| [2] | Hofbauer LC, Compston JE, Saag KG, et al. Glucocorticoid-induced osteoporosis: novel concepts and clinical implications[J]. Lancet Diabetes Endocrinol, 2025, 13(11): 964-79. doi:10.1016/s2213-8587(25)00251-7 |
| [3] | Chen M, Fu W, Xu H, et al. Pathogenic mechanisms of glucocorticoid-induced osteoporosis[J]. Cytokine Growth Factor Rev, 2023, 70: 54-66. doi:10.1016/j.cytogfr.2023.03.002 |
| [4] | Buckley L, Humphrey MB. Glucocorticoid-Induced Osteoporosis. N Engl J Med, 2018, 379(26): 2547-56. doi:10.1056/nejmcp1800214 |
| [5] | He X, Hu W, Zhang Y, et al. Cellular senescence in skeletal disease: mechanisms and treatment[J]. Cell Mol Biol Lett, 2023, 28(1): 88. doi:10.1186/s11658-023-00501-5 |
| [6] | de Magalhães JP. Cellular senescence in normal physiology[J]. Science, 2024, 384(6702): 1300-1. doi:10.1126/science.adj7050 |
| [7] | Wang F, Yang S, Zhu Y, et al. Mechanical energy-charged navigating pump repair natural vascular scaffolds to reverse osteoporosis[J]. Mater Today Bio, 2025, 35: 102431. doi:10.1016/j.mtbio.2025.102431 |
| [8] | Li K, Hu S, Chen H. Cellular senescence and other age-related mechanisms in skeletal diseases[J]. Bone Res, 2025, 13(1): 68. doi:10.1038/s41413-025-00448-7 |
| [9] | Kusumbe AP, Ramasamy SK, Adams RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone[J]. Nature, 2014, 507(7492): 323-8. doi:10.1038/nature13145 |
| [10] | Ramasamy SK, Kusumbe AP, Schiller M, et al. Blood flow controls bone vascular function and osteogenesis[J]. Nat Commun, 2016, 7: 13601. doi:10.1038/ncomms13601 |
| [11] | Liang H, Wang O, Cheng Z, et al. Jintiange combined with alfacal-cidol improves muscle strength and balance in primary osteoporosis: A randomized, double-blind, double-dummy, positive-controlled, multicenter clinical trial[J]. J Orthop Translat, 2022, 35: 53-61. doi:10.1016/j.jot.2022.05.002 |
| [12] | Shen Y, Wang N, Zhang Q, et al. Jin-Tian-Ge ameliorates ovariectomy-induced bone loss in rats and modulates osteoblasto-genesis and osteoclastogenesis in vitro [J]. Chin Med, 2022, 17(1):78. doi:10.1186/s13020-022-00627-2 |
| [13] | Liu J, Zhang X, Hou H, et al. Advances in osteoblast and mitochondrial dynamics and their transfer in osteoporosis[J]. J Cell Mol Med, 2024, 28(24): e70299. doi:10.1111/jcmm.70299 |
| [14] | Man Y, Na J, Wang H, et al. Efficacy and safety of Jintiange in the treatment of osteoporosis: a systematic review and meta-analysis[J]. Front Pharmacol, 2025, 16: 1592184. doi:10.3389/fphar.2025.1592184 |
| [15] | Huang Y, Che X, Wang PW, Qu X. p53/MDM2 signaling pathway in aging, senescence and tumorigenesis[J]. Semin Cancer Biol, 2024,101: 44-57. doi:10.1016/j.semcancer.2024.05.001 |
| [16] | Zhang L, Liu M, Liu W, et al. Th17/IL-17 induces endothelial cell senescence via activation of NF‑κB/p53/Rb signaling pathway[J]. Lab Invest, 2021, 101(11): 1418-26. doi:10.1038/s41374-021-00629-y |
| [17] | Naderi EH, Jochemsen AG, Blomhoff HK, et al. Activation of cAMP signaling interferes with stress-induced p53 accumulation in ALL-derived cells by promoting the interaction between p53 and HDM2[J]. Neoplasia, 2011, 13(7): 653-63. doi:10.1593/neo.11542 |
| [18] | Wu J, Hu M, Jiang H, et al. Endothelial cell-derived lactate triggers bone mesenchymal stem cell histone lactylation to attenuate osteoporosis[J]. Adv Sci (Weinheim), 2023, 10(31): e2301300. doi:10.1002/advs.202301300 |
| [19] | Finkel T, Holbrook NJ. Oxidants, oxidative stress and the biology of ageing[J]. Nature, 2000, 408(6809): 239-47. doi:10.1038/35041687 |
| [20] | Zhivodernikov IV, Kirichenko TV, Markina YV, et al. Molecular and cellular mechanisms of osteoporosis[J]. Int J Mol Sci, 2023, 24(21): 15772. doi:10.3390/ijms242115772 |
| [21] | Wang B, Wang L, Gasek NS, et al. An inducible p21-Cre mouse model to monitor and manipulate p21-highly-expressing senescent cells in vivo[J]. Nat Aging, 2021, 1(10): 962-73. doi:10.1038/s43587-021-00107-6 |
| [22] | Wang Z, Yuan J, Xu Y, et al. Oleaeuropaea leaf exosome-like nanovesicles encapsulated in a hyaluronic acid / tannic acid hydrogel dressing with dual "defense-repair" effects for treating skin photoaging[J]. Mater Today Bio, 2024, 26: 101103. doi:10.1016/j.mtbio.2024.101103 |
| [23] | Wang Q, Zhang Y, Ma K, et al. Plexin B2 in physiology and pathophysiology of the central nervous system[J]. Int Immuno-pharmacol, 2025, 155: 114627. doi:10.1016/j.intimp.2025.114627 |
| [24] | Kishimoto K, Liu S, Tsuji T, et al. Endogenous angiogenin in endothelial cells is a general requirement for cell proliferation and angiogenesis[J]. Oncogene, 2005, 24(3): 445-56. doi:10.1038/sj.onc.1208223 |
| [25] | Liu X, Chai Y, Liu G, et al. Osteoclasts protect bone blood vessels against senescence through the angiogenin/plexin-B2 axis[J]. Nat Commun, 2021, 12(1): 1832. doi:10.1038/s41467-021-22131-1 |
| [26] | Cun D, Yang N, Zhou L, et al. Relationship between systemic inflammatory response index and bone mineral density in children and adolescents aged 8-19 years: a cross-sectional study based on NHANES 2011-2016[J]. Front Endocrinol, 2025, 16: 1537574. doi:10.3389/fendo.2025.1537574 |
| [27] | Ren S, Lin Y, Liu W, et al. MSC-Exos: Important active factor of bone regeneration[J]. Front Bioeng Biotechnol, 2023, 11: 1136453. doi:10.3389/fbioe.2023.1136453 |
| [28] | Li F, Lu Y, Ding M, et al. Runx2 contributes to murine Col10a1 gene regulation through direct interaction with its cis-enhancer[J]. J Bone Miner Res, 2011, 26(12): 2899-910. doi:10.1002/jbmr.504 |
| [29] | Zhu S, Chen W, Masson A, et al. Cell signaling and transcriptional regulation of osteoblast lineage commitment, differentiation, bone formation, and homeostasis[J]. Cell Discov, 2024, 10(1): 71. doi:10.1038/s41421-024-00689-6 |
| [30] | Tie K, Cai J, Qin J, et al. Nanog/NFATc1/Osterix signaling pathway-mediated promotion of bone formation at the tendon-bone interface after ACL reconstruction with De-BMSCs transplantation[J]. Stem Cell Res Ther, 2021, 12(1): 576. doi:10.1186/s13287-021-02643-9 |
| [31] | Liu Q, Li M, Wang S, et al. Recent advances of Osterix transcription factor in osteoblast differentiation and bone formation[J]. Front Cell Dev Biol, 2020, 8: 601224. doi:10.3389/fcell.2020.601224 |
| [32] | El-Makawy AI, Ibrahim FM, Mabrouk DM, et al. Efficiency of turnip bioactive lipids in treating osteoporosis through activation of Osterix and suppression of Cathepsin K and TNF-α signaling in rats[J]. Environ Sci Pollut Res Int, 2020, 27(17): 20950-61. doi:10.1007/s11356-020-08540-7 |
| [1] | 易明, 罗烨, 吴露, 吴泽衡, 蒋翠平, 陈史钰, 柯晓. 黄芪甲苷通过抑制PINK1/Parkin通路调控细胞线粒体自噬减轻D-半乳糖诱导的内皮细胞衰老[J]. 南方医科大学学报, 2025, 45(11): 2427-2436. |
| [2] | 何思齐, 文楠, 陈勋, 王跃, 张艇, 牟雁东. 枸杞糖肽可减轻放射治疗后人牙龈成纤维细胞来源的外泌体导致的成骨抑制[J]. 南方医科大学学报, 2024, 44(9): 1752-1759. |
| [3] | 柯志勇, 黄子城, 何若琳, 张倩, 陈思旭, 崔忠凯, 丁晶. 抑制Hmga2促进小鼠脂肪间充质干细胞成骨分化并加速骨缺损修复[J]. 南方医科大学学报, 2024, 44(7): 1227-1235. |
| [4] | 王磊, 卞芬兰, 马飞扬, 方舒, 凌梓涵, 刘梦然, 孙红燕, 付程文, 倪诗垚, 赵晓阳, 冯心茹, 孙正宇, 卢国庆, 康品方, 吴士礼. 激活ALDH2通过上调SIRT1/PGC-1α信号通路减轻小鼠缺氧性肺动脉高压[J]. 南方医科大学学报, 2024, 44(10): 1955-1964. |
| [5] | 陈梓锋, 李胜发, 张祐鸣, 杨婉雯, 王 婷. 脂质运载蛋白2自限性抑制间充质干细胞的成骨细胞分化[J]. 南方医科大学学报, 2023, 43(8): 1339-1344. |
| [6] | 刘 屿, 曾 莲, 王卫红, 杨艳玲, 王 洲, 刘建启, 李 卫, 孙婧宇, 余晓宏. 人骨髓间充质干细胞外泌体来源的miR-335-5p促进人牙周膜干细胞的成骨分化:基于下调DKK1表达[J]. 南方医科大学学报, 2023, 43(3): 420-427. |
| [7] | 金晓丽, 许 嘉, 陈煊威, 陈 瑾, 黄 慧, 张 婷, 任 军, 许 健. 冬凌草甲素逆转硫代乙酰胺对破骨和成骨细胞分化的机制研究[J]. 南方医科大学学报, 2023, 43(11): 1892-1900. |
| [8] | 路晓淼, 田瑞雪, 刘姗姗, 徐锦程. 神经生长因子联合牙髓干细胞可促进大鼠种植体周围骨结合[J]. 南方医科大学学报, 2021, 41(9): 1304-1309. |
| [9] | 罗玉婷, 杨正艳, 李 蒙, 赵曼竹, 温秀杰, 周 智. Mage-D1与活化后的p75神经营养因子受体结合可正向调节大鼠外胚间充质干细胞的矿化[J]. 南方医科大学学报, 2021, 41(10): 1547-1553. |
| [10] | 黄柳维, 沈燕婷, 刘崇斌, 李彩珍, 王 骏. Palbociclib可诱导人肾小管上皮细胞周期阻滞及衰老[J]. 南方医科大学学报, 2020, 40(12): 1784-1792. |
| [11] | 陆进,张浩轩,俞鹏,龚义凤,龚喜旺,范强强,杨月. miR-144-3p在大鼠骨髓间充质干细胞成骨分化过程中的表达及其靶向调控作用[J]. 南方医科大学学报, 2018, 38(09): 1083-. |
| [12] | 刘松,吴建群,胡稷杰,王簕,王钊,孟欢,卓灵剑,郑健雄. 神经肽Y Y1受体拮抗剂促进大鼠BMSCs成骨分化和股骨缺损修复[J]. 南方医科大学学报, 2018, 38(06): 669-. |
| [13] | 周鑫,张磊,扶世杰,刘刚,郭晓光,易刚,刘洋,汪国友. 少阳主骨方介导p19Arf-p53-p21Cip1信号通路调控食蟹猴关节软骨退变的机制[J]. 南方医科大学学报, 2018, 38(03): 346-. |
| [14] | 杜婷婷,刘娜,张维,石海刚,张彤. 年龄因素对牙周膜干细胞增殖和分化能力的影响[J]. 南方医科大学学报, 2017, 37(03): 360-. |
| [15] | 谭咏梅,侯晋,杨小军,梁悦娥,张琰,赵望泓. 侵入细胞内的牙龈卟啉单胞菌影响人牙周膜细胞的增殖及成骨分化[J]. 南方医科大学学报, 2016, 36(04): 525-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||