南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (6): 1153-1162.doi: 10.12122/j.issn.1673-4254.2025.06.05
王心恒1,2( ), 邵小涵3, 李童童3, 张璐1, 杨勤军1,2,5, 叶卫东1, 童佳兵1,2,4,5, 李泽庚1,2,4,5, 方向明1(
), 邵小涵3, 李童童3, 张璐1, 杨勤军1,2,5, 叶卫东1, 童佳兵1,2,4,5, 李泽庚1,2,4,5, 方向明1( )
)
收稿日期:2024-12-25
出版日期:2025-06-20
发布日期:2025-06-27
通讯作者:
方向明
E-mail:wangxinheng@ahtcm.edu.cn;fxm.bsh@163.com
作者简介:王心恒,讲师,博士,E-mail: wangxinheng@ahtcm.edu.cn
基金资助:
Xinheng WANG1,2( ), Xiaohan SHAO3, Tongtong LI3, Lu ZHANG1, Qinjun YANG1,2,5, Weidong YE1, Jiabing TONG1,2,4,5, Zegeng LI1,2,4,5, Xiangming FANG1(
), Xiaohan SHAO3, Tongtong LI3, Lu ZHANG1, Qinjun YANG1,2,5, Weidong YE1, Jiabing TONG1,2,4,5, Zegeng LI1,2,4,5, Xiangming FANG1( )
)
Received:2024-12-25
Online:2025-06-20
Published:2025-06-27
Contact:
Xiangming FANG
E-mail:wangxinheng@ahtcm.edu.cn;fxm.bsh@163.com
Supported by:摘要:
目的 基于高迁移率族蛋白 B1(HMGB1)/自噬相关基因Beclin-1(Beclin-1)轴探讨平喘宁方(PCN)调控细胞自噬减轻寒哮证大鼠气道炎症的作用。 方法 105只SPF级SD大鼠,随机分为对照组(CON)、模型组(MOD)、平喘宁高、中、低剂量组(PCN-H、PCN-M、PCN-L)、桂龙咳喘宁组(GLCKN)和地塞米松组(DEX),15只/组。除CON组外,其余大鼠构建寒哮证模型,并从第29天开始灌胃PCN、DEX及GLCKN。观察各组大鼠体质量、摄食量、摄水量,肺功能、肺组织氧化应激水平、肺泡灌洗液(BALF)炎性因子、肺组织病理、肺组织超微结构,细胞因子水平以及HMGB1/Beclin-1信号轴和细胞自噬相关基因和蛋白的表达。 结果 与CON组比较,MOD组大鼠表现出呼吸急促、喘息、流涕、腹部收缩明显、反应迟钝、反应缓慢,进食量减少,明显的皮毛枯槁或脱落等表现;体质量、摄食量、摄水量明显下降(P<0.05);第0.3秒用力呼气量(FEV0.3)、用力肺活量(FVC)、FEV0.3/FVC明显降低(P<0.05);肺组织表现出明显的炎性浸润,肺泡炎症评分明显上升(P<0.05);BALF中性粒细胞、嗜酸性粒细胞、淋巴细胞、巨噬细胞、白细胞数量明显上升(P<0.05);肺组织MDA明显上升,SOD明显下降(P<0.05);透射电镜(TEM)观察发现MOD组有自噬小泡数量较多,线粒体排列有紊乱及水肿现象,嗜锇性板层小体排空比较明显;肺组织HMGB1、Beclin-1、ATG5蛋白及mRNA表达明显上升,Bcl-2蛋白表达及mRNA明显降低,LC3II/I比值明显上升(P<0.05);大鼠肺组织细胞因子IFN-γ明显降低,TNF-α、IL-6IL-1β、IL-13明显上升(P<0.05)。与MOD组比较,PCN能明显改善各项指标的病理改变,且作用效果优于GLKCN组,不亚于DEX组(P<0.05)。 结论 PCN能有效改善寒哮证模型大鼠气道炎症,其机制可能与调控HMGB1/Beclin-1信号轴,抑制细胞自噬从而缓解炎症损伤有关。
王心恒, 邵小涵, 李童童, 张璐, 杨勤军, 叶卫东, 童佳兵, 李泽庚, 方向明. 平喘宁方通过调控HMGB1/Beclin-1轴介导的自噬改善患寒哮证大鼠的气道炎症[J]. 南方医科大学学报, 2025, 45(6): 1153-1162.
Xinheng WANG, Xiaohan SHAO, Tongtong LI, Lu ZHANG, Qinjun YANG, Weidong YE, Jiabing TONG, Zegeng LI, Xiangming FANG. Pingchuanning Formula suppresses airway inflammation in a rat model of asthmatic cold syndrome by regulating the HMGB1/Beclin-1 axis-mediated autophagy[J]. Journal of Southern Medical University, 2025, 45(6): 1153-1162.
| Scientific name | Family | Drug name |
|---|---|---|
| Ephedra sinica stapf | Ephedraceae | Chinese ephedra |
| Prunus armeniaca L. | Rosaceae | Apricot |
| Citrus reticulata blanco | Rutaceae | Satsuma orenge |
| Bulbus fritillariae thunbergii | Liliaceae | Bulb of thunberg fritillary |
Asarum heterotropoides Fr. Schmidt var. | Aristolochiaceae | Manchur wildginger |
| Perilla frutescens (L.) Britt. | Lamiaceae | Common perilla |
| Astragali radix | Fabaceae | Milkvetch root |
| Radix pseudoxtellariae | Caryophyllaceae | Heterophylly falsestarwort root |
| Pheretima | Megascolecidae | Earthworm |
| Pinelliae rhizoma | Araceae | Pinellia ternata |
| Saposhnikoviae radix | Umbelliferae | Divaricate saposhniovia root |
表1 PCN方的中药组成
Tab.1 Composition of Pingchuanning Formula (PCN)
| Scientific name | Family | Drug name |
|---|---|---|
| Ephedra sinica stapf | Ephedraceae | Chinese ephedra |
| Prunus armeniaca L. | Rosaceae | Apricot |
| Citrus reticulata blanco | Rutaceae | Satsuma orenge |
| Bulbus fritillariae thunbergii | Liliaceae | Bulb of thunberg fritillary |
Asarum heterotropoides Fr. Schmidt var. | Aristolochiaceae | Manchur wildginger |
| Perilla frutescens (L.) Britt. | Lamiaceae | Common perilla |
| Astragali radix | Fabaceae | Milkvetch root |
| Radix pseudoxtellariae | Caryophyllaceae | Heterophylly falsestarwort root |
| Pheretima | Megascolecidae | Earthworm |
| Pinelliae rhizoma | Araceae | Pinellia ternata |
| Saposhnikoviae radix | Umbelliferae | Divaricate saposhniovia root |
| Gene | Amplicon size (bp) | Forward primer (5'→3') | Reverse primer (5'→3') |
|---|---|---|---|
| GAPDH | 127 | CAAGGCTGAGAATGGGAAGC | GAAGACGCCAGTAGACTCCA |
| Beclin-1 | 134 | GAATGAGGGCGACAGTGAAC | CCTGGACCTTCTCCAGGTTT |
| ATG5 | 71 | TCCTGCCACAGTAAGGGAAG | TCACAGTCAATGGCAGGAGT |
| HMGB1 | 85 | CCTAAGAAGCCGAGAGGCAA | CATCCGGGTGCTTCTTCTTG-3' |
| Bcl-2 | 84 | GCCTTCTTTGAGTTCGGTG | GCCAGGAGAAATCAAACAGAG |
表2 各检测指标引物
Tab.2 Primers for amplification of the target mRNAs
| Gene | Amplicon size (bp) | Forward primer (5'→3') | Reverse primer (5'→3') |
|---|---|---|---|
| GAPDH | 127 | CAAGGCTGAGAATGGGAAGC | GAAGACGCCAGTAGACTCCA |
| Beclin-1 | 134 | GAATGAGGGCGACAGTGAAC | CCTGGACCTTCTCCAGGTTT |
| ATG5 | 71 | TCCTGCCACAGTAAGGGAAG | TCACAGTCAATGGCAGGAGT |
| HMGB1 | 85 | CCTAAGAAGCCGAGAGGCAA | CATCCGGGTGCTTCTTCTTG-3' |
| Bcl-2 | 84 | GCCTTCTTTGAGTTCGGTG | GCCAGGAGAAATCAAACAGAG |
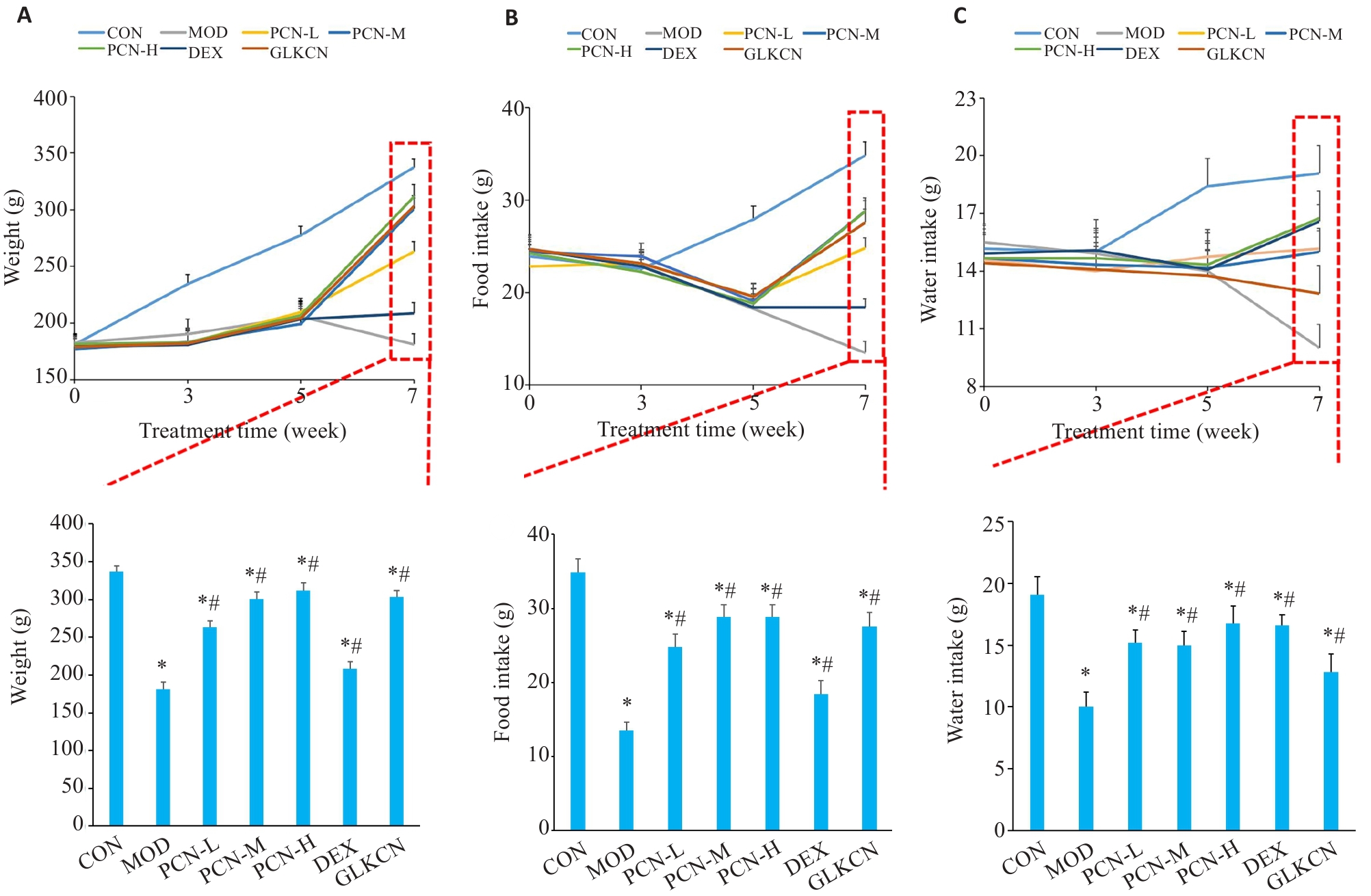
图2 各组大鼠大鼠体质量、摄食量和摄水量比较
Fig.2 Comparison of body mass (A), food intake (B), and water intake (C) of the rats among the groups. *P<0.05 vs control group, #P<0.05 vs model group.
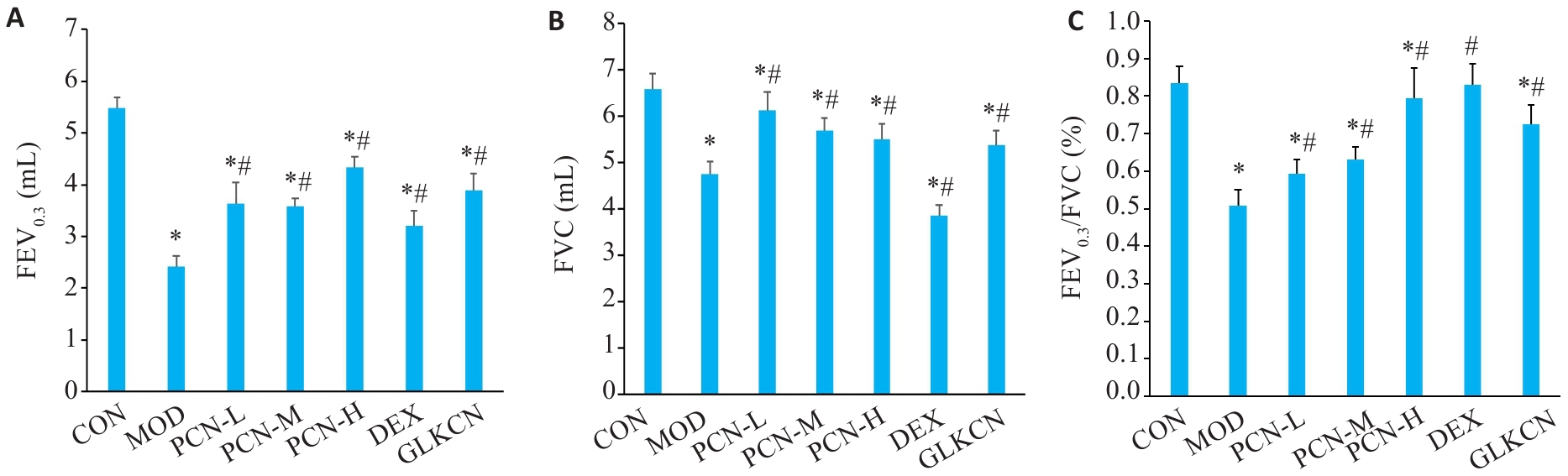
图3 各组大鼠肺功能指标比较
Fig.3 Comparison of lung function of the rats among the groups. A: FEV0.3; B: FVC; C: FEV0.3/FVC; *P<0.05 vs control group, #P<0.05 vs model group.
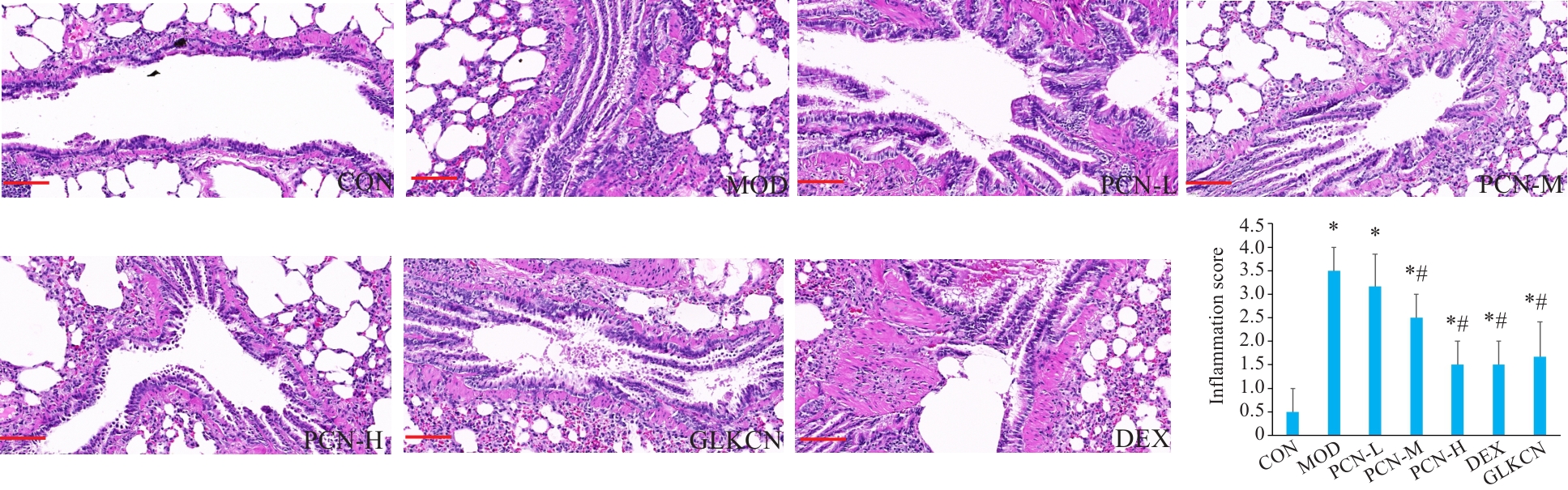
图4 各组大鼠肺组织病理学形态和炎症评分比较
Fig.4 Lung histopathology and inflammation scores of the rats in each group (HE staining, scale bar=40 μm). *P<0.05 vs control group, #P<0.05 vs model group (n=6).
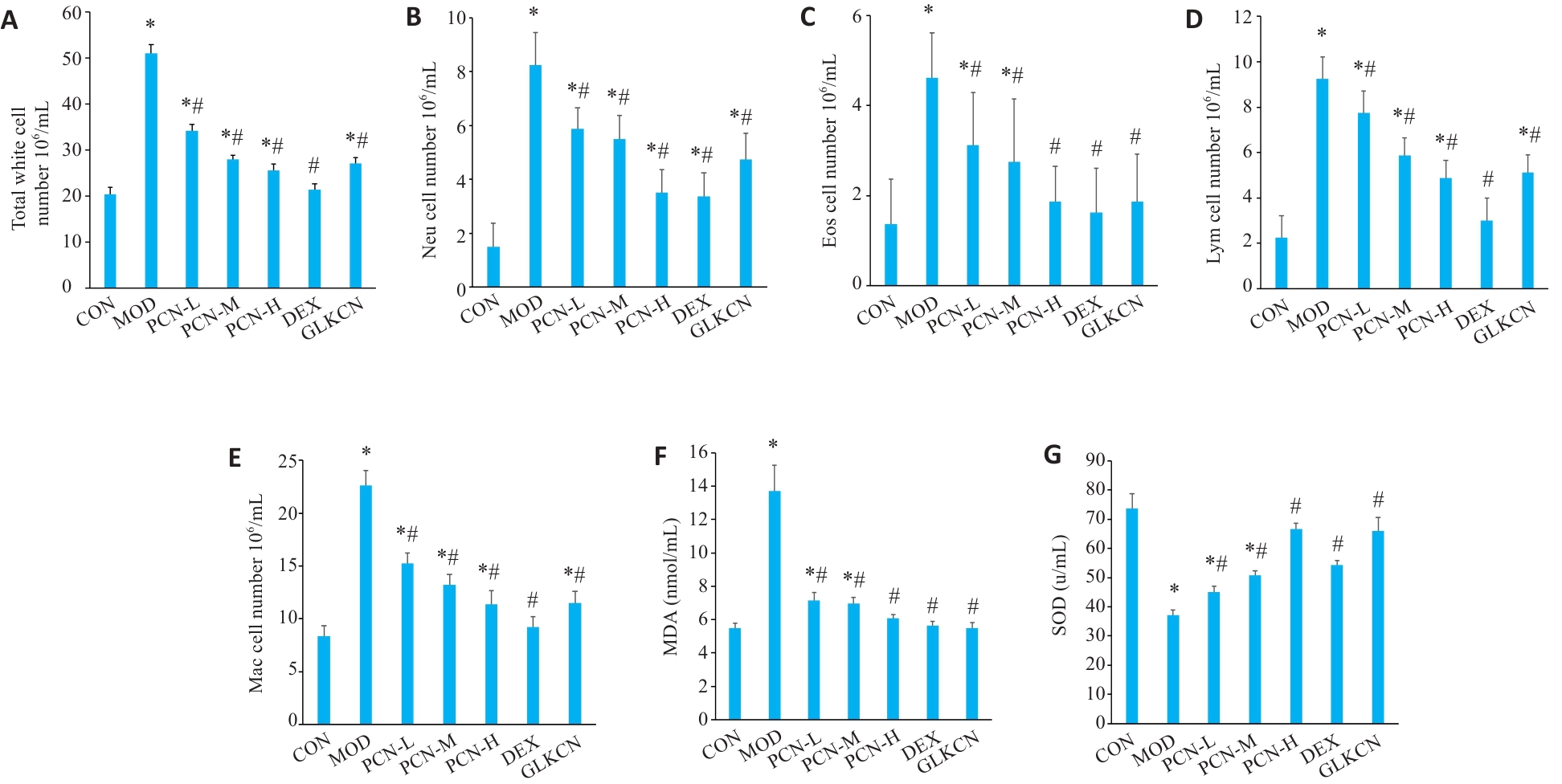
图5 各组大鼠氧化应激和炎症因子指标比较
Fig.5 Comparison of inflammatory cell counts in the BALF and oxidative stress factors among the groups. A: Total white cell counts. B: Neu cell counts. C: Eos cell counts. D: Lym cell counts. E: Mac cell counts. F: MDA level. G: SOD level. *P<0.05 vs control group, #P<0.05 vs model group
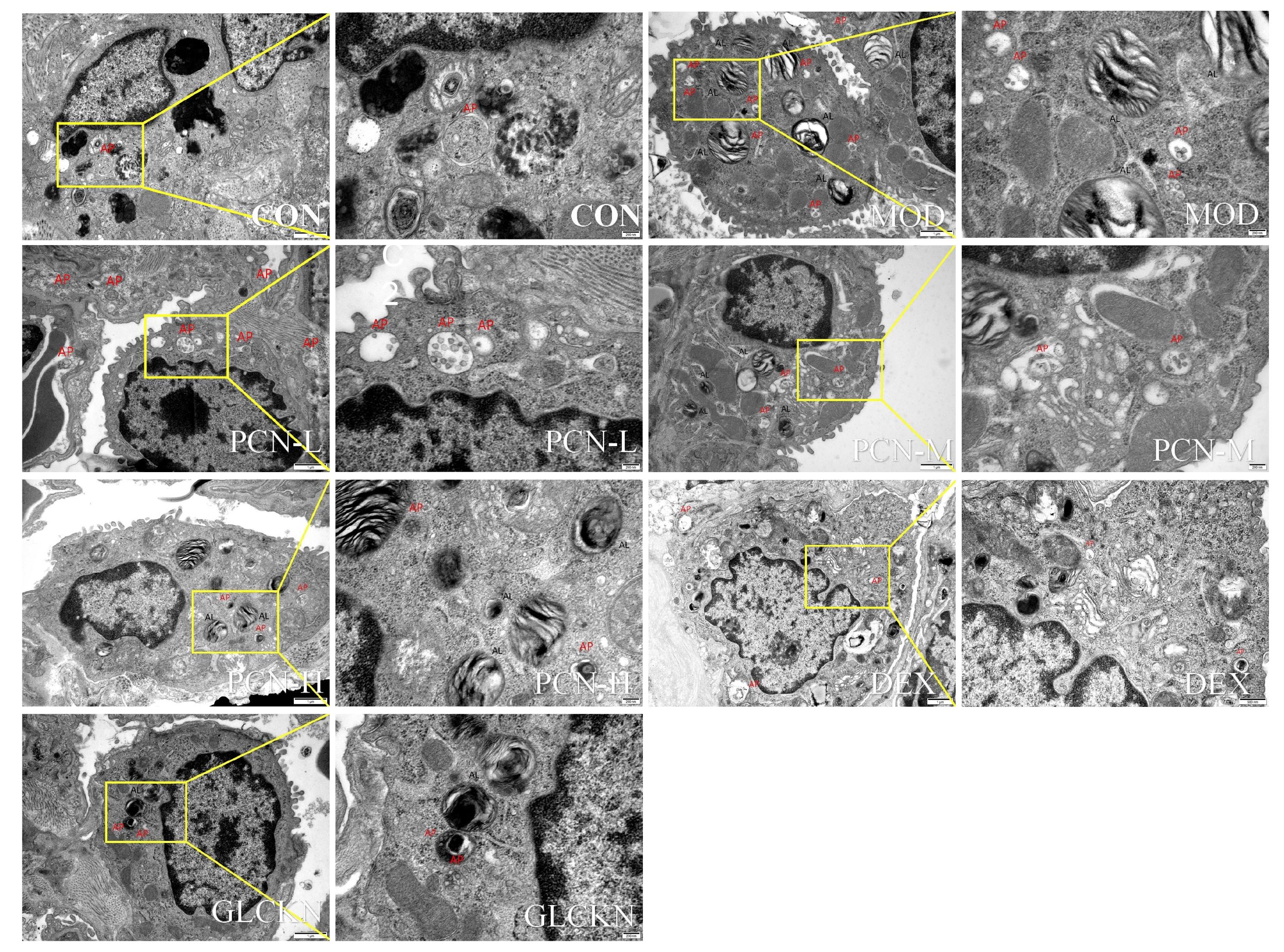
图6 TEM观察各组大鼠肺组织超微结构
Fig.6 Ultrastructure of lung tissue of rats in each group observed by TEM (Original magnification:×10 000, ×25 000). AP: Autophagosome; AL: Autophagolysosome.
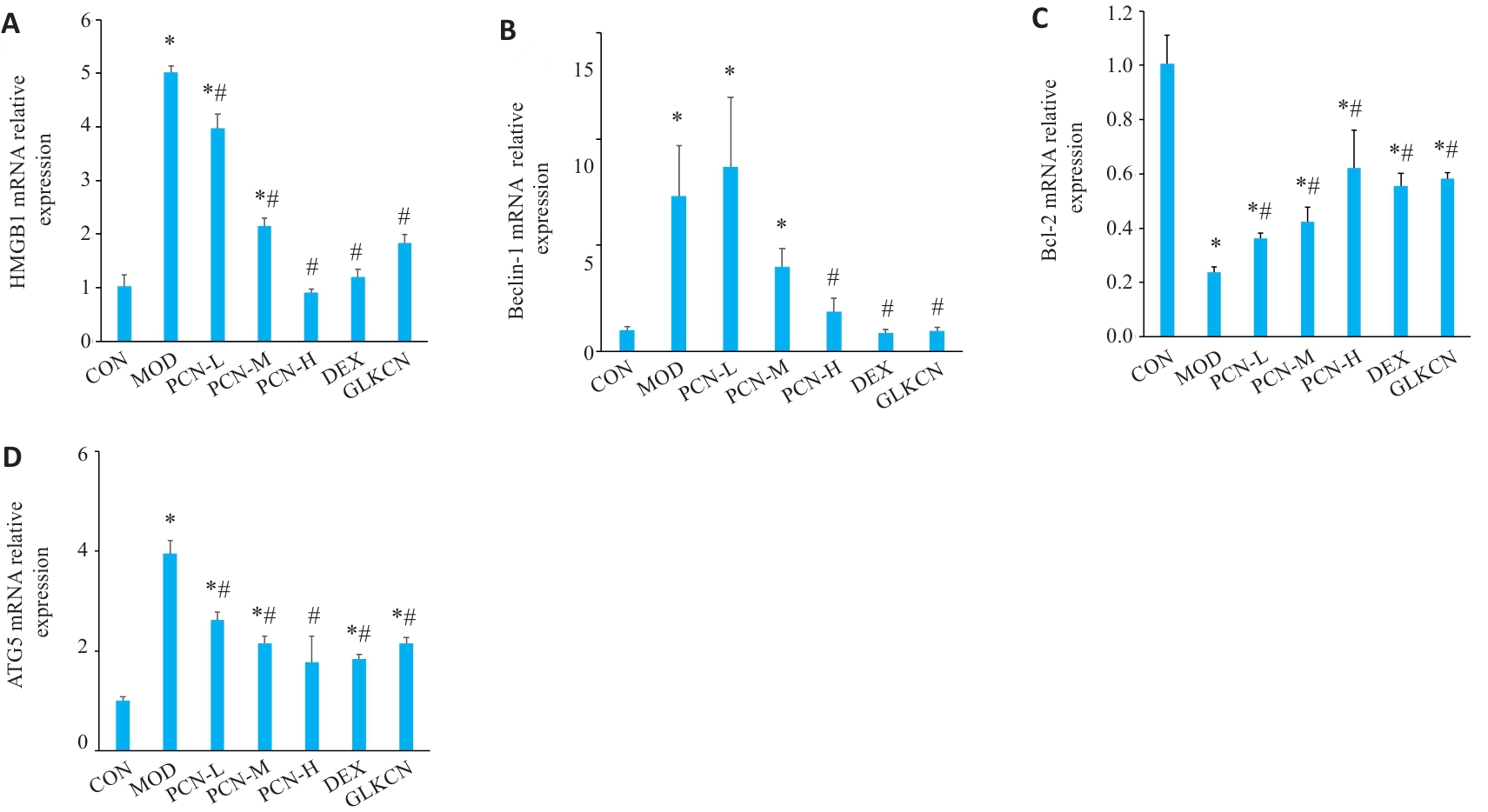
图7 各组大鼠自噬相关mRNA的表达
Fig.7 Expression of autophagy-related mRNAs in the lung tissue of the rats in each group. A: HMGB1 mRNA expression; B: Beclin-1 mRNA expression; C: Bcl-2 mRNA expression; D: ATG5 mRNA expression. *P<0.05 vs control group; #P<0.05 vs model group.
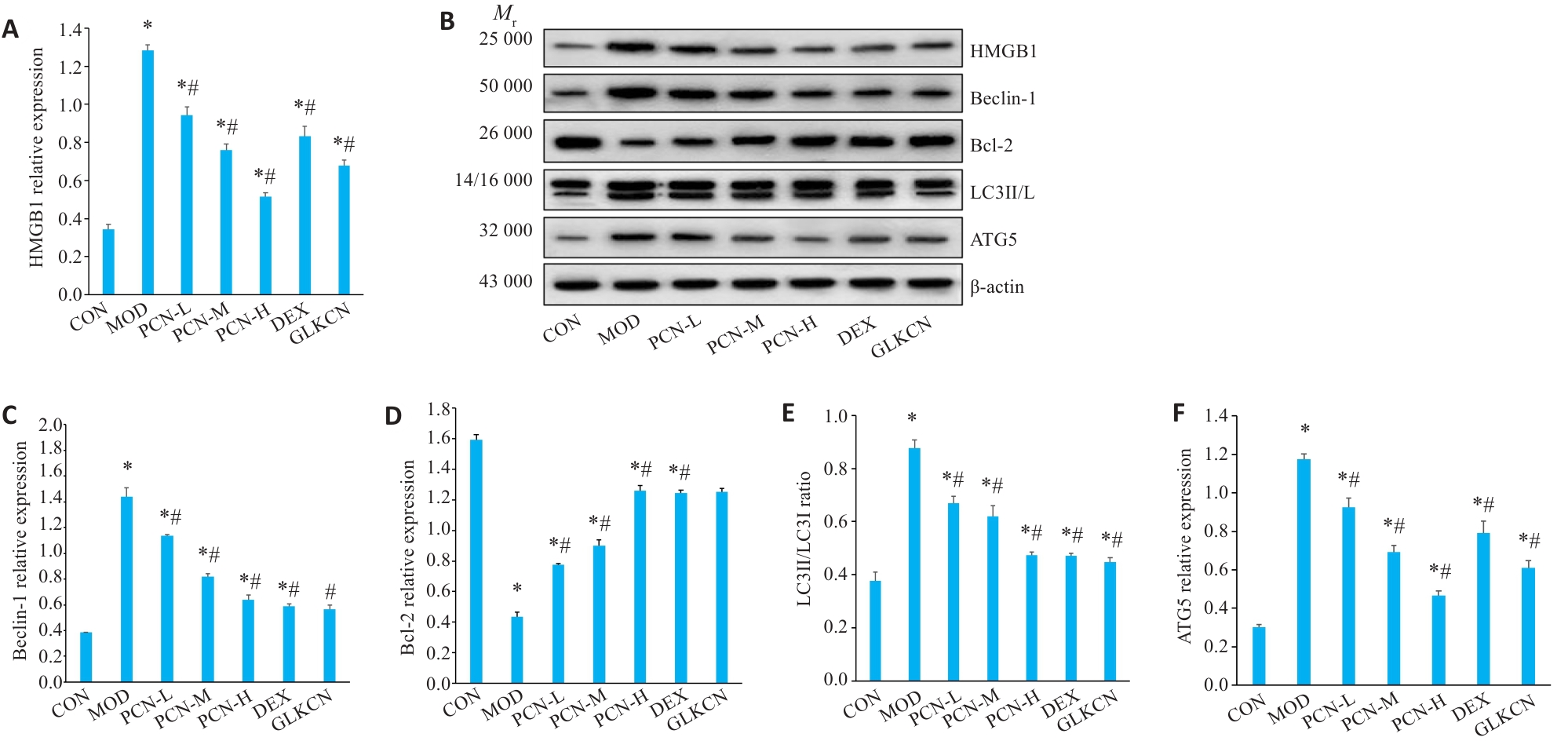
图8 各组大鼠自噬相关蛋白的表达
Fig.8 Expression levels of autophagy-associated proteins in the lung tissue of the rats in the groups. A: HMGB1 expression; B: Western blotting for analyzing HMGB1, Beclin-1, Bcl-2, LC3II/LC3I, and ATG5 expressions in the lung tissue; C: Beclin-1 expression; D: Bcl-2 expression; E: LC3II/LC3I ratio; F: ATG5 expression. *P<0.05 vs control group; #P<0.05 vs model group.
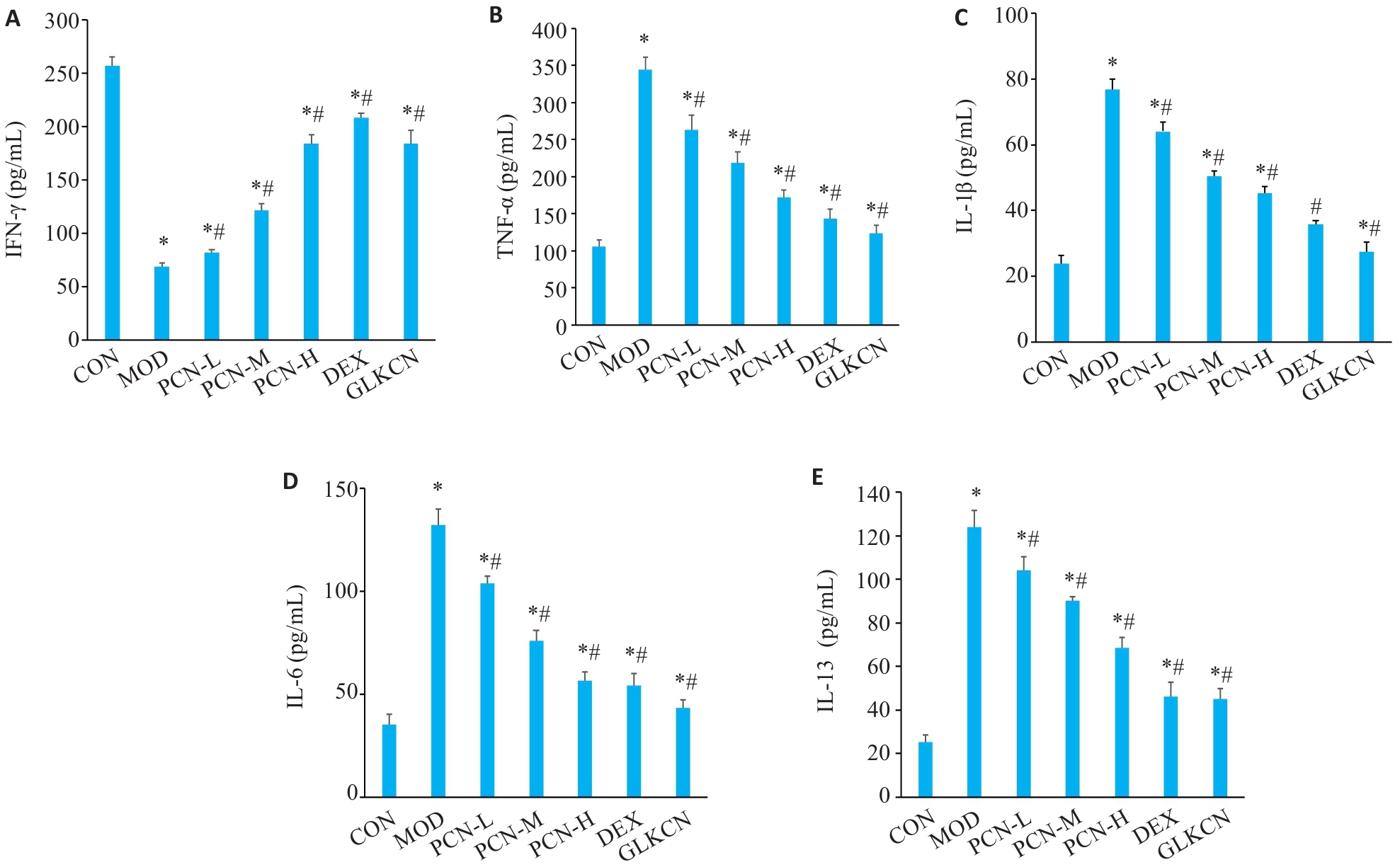
图9 各组大鼠肺组织炎症因子比较
Fig.9 Comparison of lung tissue inflammatory factors among the group. A: IFN-γ expression; B: TNF-α expression; C: IL-1β expression; D: IL-6 expression; E: IL-13 expression. *P<0.05 vs control group; #P<0.05 vs model group (n=8)
| 1 | Qin ZW, Chen YJ, Wang Y, et al. Immunometabolism in the pathogenesis of asthma[J]. Immunology, 2024, 171(1): 1-17. doi:10.1111/imm.13688 |
| 2 | Brasier AR. Innate immunity, epithelial plasticity, and remodeling in asthma[J]. Adv Exp Med Biol, 2023, 1426: 265-85. doi:10.1007/978-3-031-32259-4_13 |
| 3 | Global lnitiative for Asthma.Global strategy for asthma management and prevention[EB/OL].Revised, 2024. |
| 4 | Heffler E, Madeira LNG, Ferrando M, et al. Inhaled corticosteroids safety and adverse effects in patients with asthma[J]. J Allergy Clin Immunol Pract, 2018, 6(3): 776-81. doi:10.1016/j.jaip.2018.01.025 |
| 5 | 范晓璇, 颜培正, 张庆祥. 黄芪甲苷通过LC3/Beclin 1干预哮喘寒饮蕴肺证大鼠细胞自噬的机制[J]. 中华中医药杂志, 2024, 39(11): 6037-42. |
| 6 | Bao WP, Zhang YY, Hang JQ, et al. Short-acting beta-2 agonist prescription patterns and clinical outcomes in Chinese patients with asthma: an observational study in mainland China for the SABINA programme[J]. Ther Adv Respir Dis, 2022, 16: 17534666221115054. doi:10.1177/17534666221115054/v1/review2 |
| 7 | Haughney J, Winders T, Holmes S, et al. A charter to fundamentally change the role of oral corticosteroids in the management of asthma[J]. Adv Ther, 2023, 40(6): 2577-94. doi:10.1007/s12325-023-02479-0 |
| 8 | Sachdeva K, Do DC, Zhang Y, et al. Environmental exposures and asthma development: autophagy, mitophagy, and cellular senescence[J]. Front Immunol, 2019, 10: 2787. doi:10.3389/fimmu.2019.02787 |
| 9 | Dong HN, Yang W, Li W, et al. New insights into autophagy in inflammatory subtypes of asthma[J]. Front Immunol, 2023, 14: 1156086. doi:10.3389/fimmu.2023.1156086 |
| 10 | Wen X, Yang Y, Klionsky DJ. Moments in autophagy and disease: past and present[J]. Mol Aspects Med, 2021, 82: 100966. doi:10.1016/j.mam.2021.100966 |
| 11 | Debeuf N, Lambrecht BN. Eicosanoid control over antigen presenting cells in asthma[J]. Front Immunol, 2018, 9: 2006. doi:10.3389/fimmu.2018.02006 |
| 12 | Li BB, Chen YL, Pang FZ. microRNA-30a targets ATG5 and attenuates airway fibrosis in asthma by suppressing autophagy[J]. Inflammation, 2020, 43(1): 44-53. doi:10.1007/s10753-019-01076-0 |
| 13 | Cao XM, Wang K, Zhu HZ. Yanghepingchuan granule improves airway inflammation by inhibiting autophagy via miRNA328-3p/high mobility group box 1/Toll-like receptor 4 targeting of the pathway of signaling in rat models of asthma[J]. J Thorac Dis, 2023, 15(11): 6251-64. doi:10.21037/jtd-23-1262 |
| 14 | Tao ZP, Helms MN, Leach BCB, et al. Molecular insights into the multifaceted functions and therapeutic targeting of high mobility group box 1 in metabolic diseases[J]. J Cell Mol Med, 2022, 26(14): 3809-15. doi:10.1111/jcmm.17448 |
| 15 | Zhang D, Zhang JT, Xu CJ, et al. A humanized mouse model to study asthmatic airway remodeling and Muc-5ac secretion via the human IL-33[J]. Allergy, 2024, 79(5): 1364-7. doi:10.1111/all.16030 |
| 16 | Quan JY, Wen XX, Su GM, et al. Epithelial SIRT6 governs IL-17A pathogenicity and drives allergic airway inflammation and remodeling[J]. Nat Commun, 2023, 14(1): 8525. doi:10.1038/s41467-023-44179-x |
| 17 | Qu LH, Chen C, Chen YY, et al. High-mobility group box 1 (HMGB1) and autophagy in acute lung injury (ALI): a review[J]. Med Sci Monit, 2019, 25: 1828-37. doi:10.12659/msm.912867 |
| 18 | Yang QN, Li M, Hou YJ, et al. High-mobility group box 1 emerges as a therapeutic target for asthma[J]. Immun Inflamm Dis, 2023, 11(12): e1124. doi:10.1002/iid3.1124 |
| 19 | Chen YJ, Shimizu Bassi G, Wang Y, et al. Research hotspot and frontier analysis of traditional Chinese medicine in asthma using bibliometric methods from 1991 to 2021[J]. J Allergy Clin Immunol Glob, 2022, 1(4): 185-97. doi:10.1016/j.jacig.2022.07.004 |
| 20 | 方向明, 孙子凯. 平喘宁对哮喘患者血清嗜酸细胞阳离子蛋白及白细胞介素2受体的影响[J]. 中国中医药科技, 2002, 9(3): 160. doi:10.3969/j.issn.1005-7072.2002.03.038 |
| 21 | 方向明, 王德均. 平喘宁对哮喘患者白细胞流变特性CD18 +表达及SICAM-1水平的影响[J]. 辽宁中医杂志, 2001, 28(12): 732. doi:10.3969/j.issn.1000-1719.2001.12.016 |
| 22 | 杨勤军, 王 卉, 徐淑钰, 等. 参芪调肾方减轻慢阻肺肺肾气虚证大鼠气道炎症的机制: 基于铁死亡途径[J]. 南方医科大学学报, 2024, 44(10): 1937-46. doi:10.12122/j.issn.1673-4254.2024.10.12 |
| 23 | 杨勤军. 麻芍平喘汤治疗支气管哮喘(冷哮证)临床疗效观察及实验研究[D]. 合肥: 安徽中医药大学, 2020. |
| 24 | Ridzuan N, Zakaria N, Widera D, et al. Human umbilical cord mesenchymal stem cell-derived extracellular vesicles ameliorate airway inflammation in a rat model of chronic obstructive pulmonary disease (COPD)[J]. Stem Cell Res Ther, 2021, 12(1): 54. doi:10.1186/s13287-020-02088-6 |
| 25 | 蔡 旻, 方向明. 平喘宁调节PI3K信号通路干预寒哮大鼠肺组织气道炎症机制[J]. 辽宁中医药大学学报, 2024, 26(5): 19-24. doi:10.13194/j.issn.1673-842x.2024.05.004 |
| 26 | 陈林林, 谢金根, 樊学程, 等. 麻黄与杏仁配伍后在支气管哮喘模型大鼠体内的药动学研究[J]. 中国药房, 2024, 35(13): 1588-93. doi:10.6039/j.issn.1001-0408.2024.13.08 |
| 27 | 马继兴. 神农本草经辑注[M]. 北京: 人民卫生出版社, 1995: 74, 96, 201, 335, 398. |
| 28 | 周亚丽, 杨 萍, 李喜香, 等. 半夏化学成分与药理作用研究进展及其质量标志物(Q-Marker)预测[J]. 中草药, 2024, 55(14): 4939-52. doi:10.7501/j.issn.0253-2670.2024.14.029 |
| 29 | 金 鑫, 李春楠, 张 辉. 贝母属药材中生物碱类化学成分及其药理活性研究进展[J]. 中药材, 2022, 45(9): 2273-9. doi:10.13863/j.issn1001-4454.2022.09.044 |
| 30 | 李思维, 郝二伟, 杜正彩, 等. 广地龙化学成分和药理作用的研究进展及其质量标志物(Q-Marker)的预测分析[J]. 中草药, 2022, 53(8): 2560-71. doi:10.7501/j.issn.0253-2670.2022.08.033 |
| 31 | 王 为, 潘赐明, 胡 倩, 等. 基于网络药理学研究半夏-陈皮-茯苓角药治疗胃癌的分子生物学机制[J]. 实用中医内科杂志, 2023, 37(8): 39-42, 181-2. |
| 32 | Ying Y, Sun CB, Zhang SQ, et al. Induction of autophagy via the TLR4/NF-κB signaling pathway by astragaloside Ⅳ contributes to the amelioration of inflammation in RAW264.7 cells[J]. Biomed Pharmacother, 2021, 137: 111271. doi:10.1016/j.biopha.2021.111271 |
| 33 | Hyrkäs-Palmu H, Jaakkola MS, Mäkikyrö EMS, et al. Subtypes of asthma and cold weather-related respiratory symptoms[J]. Int J Environ Res Public Health, 2022, 19(14): 8790. doi:10.3390/ijerph19148790 |
| 34 | Michaeloudes C, Abubakar-Waziri H, Lakhdar R, et al. Molecular mechanisms of oxidative stress in asthma[J]. Mol Aspects Med, 2022, 85: 101026. doi:10.1016/j.mam.2021.101026 |
| 35 | Ong G, Logue SE. Unfolding the interactions between endoplasmic reticulum stress and oxidative stress[J]. Antioxidants, 2023, 12(5): 981. doi:10.3390/antiox12050981 |
| 36 | Rahman I, Morrison D, Donaldson K, et al. Systemic oxidative stress in asthma, COPD, and smokers[J]. Am J Respir Crit Care Med, 1996, 154(4 Pt 1): 1055-60. doi:10.1164/ajrccm.154.4.8887607 |
| 37 | Mizushima N, Levine B. Autophagy in human diseases[J]. N Engl J Med, 2020, 383(16): 1564-76. doi:10.1056/nejmra2022774 |
| 38 | Jiang L, Diaz PT, Best TM, et al. Molecular characterization of redox mechanisms in allergic asthma[J]. Ann Allergy Asthma Immunol, 2014, 113(2): 137-42. doi:10.1016/j.anai.2014.05.030 |
| 39 | Frisardi V, Matrone C, Street ME. Metabolic syndrome and autophagy: focus on HMGB1 protein[J]. Front Cell Dev Biol, 2021, 9: 654913. doi:10.3389/fcell.2021.654913 |
| 40 | Tang DL, Kang R, Zeh HJ, et al. The multifunctional protein HMGB1: 50 years of discovery[J]. Nat Rev Immunol, 2023, 23(12): 824-41. doi:10.1038/s41577-023-00894-6 |
| 41 | Xu SL, Liu WH, Zhang LC, et al. High mobility group box 1 levels as potential predictors of asthma severity[J]. Chin Med J, 2023, 136(13): 1606-8. doi:10.1097/cm9.0000000000002491 |
| 42 | Rong YL, Fan J, Ji CY, et al. USP11 regulates autophagy-dependent ferroptosis after spinal cord ischemia-reperfusion injury by deubiquitinating Beclin 1[J]. Cell Death Differ, 2022, 29(6): 1164-75. doi:10.1038/s41418-021-00907-8 |
| 43 | Wu WL, Gao JL, Chen D, et al. Epithelial microRNA-30a-3p targets RUNX2/HMGB1 axis to suppress airway eosinophilic inflammation in asthma[J]. Respir Res, 2022, 23(1): 17. doi:10.1186/s12931-022-01933-x |
| 44 | Zeng XN, Liu FW, Liu KY, et al. HMGB1 could restrict 1, 3-β-glucan induced mice lung inflammation by affecting Beclin1 and Bcl2 interaction and promoting the autophagy of epithelial cells[J]. Ecotoxicol Environ Saf, 2021, 222: 112460. doi:10.1016/j.ecoenv.2021.112460 |
| 45 | Zhu Q, Song JX, Chen JY, et al. Corynoxine B targets at HMGB1/2 to enhance autophagy for α-synuclein clearance in fly and rodent models of Parkinson's disease[J]. Acta Pharm Sin B, 2023, 13(6): 2701-14. doi:10.1016/j.apsb.2023.03.011 |
| 46 | Meng QY, Pu LY, Qi MR, et al. Laminar shear stress inhibits inflammation by activating autophagy in human aortic endothelial cells through HMGB1 nuclear translocation[J]. Commun Biol, 2022, 5(1): 425. doi:10.1038/s42003-022-03392-y |
| [1] | 王坤, 方昊翔, 曹晓梅, 朱子衡. miR-139-5p调控Notch/RBP-J/Hes1轴促进支气管哮喘骨髓间充质干细胞“归巢”[J]. 南方医科大学学报, 2024, 44(12): 2283-2290. |
| [2] | 匡红, 蔡文涵, 刘一鸣, 温佳新, 田硕, 薛志强. SLC2A1抑制肺腺癌铁死亡并促进肺腺癌细胞的增殖和侵袭[J]. 南方医科大学学报, 2024, 44(12): 2404-2411. |
| [3] | 杨勤军, 王卉, 徐淑钰, 杨程, 丁焕章, 吴迪, 朱洁, 童佳兵, 李泽庚. 参芪调肾方减轻慢阻肺肺肾气虚证大鼠气道炎症的机制:基于铁死亡途径[J]. 南方医科大学学报, 2024, 44(10): 1937-1946. |
| [4] | 徐 晨, 李春颖, 王 胜. 益肺健脾方降低香烟烟雾诱导的人支气管上皮细胞的炎性损伤和黏液高分泌:基于抑制TLR4/NF-κB信号通路[J]. 南方医科大学学报, 2023, 43(4): 507-515. |
| [5] | 刘秀秀, 许 乐, 吴敬医, 张一帆, 吴 超, 张 霞. 下调SIK2可减轻大鼠心肌缺血再灌注损伤:基于mTOR-ULK1信号通路的下调与细胞自噬的减少[J]. 南方医科大学学报, 2022, 42(7): 1082-1088. |
| [6] | 孙广瀚, 许 霞, 万 磊, 南淑玲, 王玉凤, 赵 黎, 程 卉, 王 坤, 刘 莹, 方妍妍, 孙 朗, 朱 俊. 程氏蠲痹汤通过抑制PI3K/Akt/mTOR信号轴促进类风湿关节炎滑膜成纤维细胞的自噬[J]. 南方医科大学学报, 2022, 42(11): 1726-1731. |
| [7] | 叶艳梅, 胡大鹏, 黄淑榆, 赵文驱, 梁健鹏, 黄俊文, 韩惠珊, 杨淑銮, 蔡绍曦, 赵海金. 过敏史减弱呼出气一氧化氮对慢性咳嗽患者痰嗜酸性粒细胞的预测作用[J]. 南方医科大学学报, 2022, 42(10): 1470-1475. |
| [8] | 肖博雅, 董理鑫, 高 慧, 杨凯朝, 王亚飞, 李晓凝, 邱海霞, 王爱国, 张 舜. 褪黑素可改善PBDE-47所致PC12细胞的异常自噬与凋亡[J]. 南方医科大学学报, 2021, 41(9): 1409-1414. |
| [9] | 谭漫琳, 简文星, 梁秋菊, 李树钧, 崔海燕. 不同评价系统对慢阻肺患者病情及治疗疗效的评估价值[J]. 南方医科大学学报, 2021, 41(7): 1119-1124. |
| [10] | 董泉明, 宋天然, 姜晨宇, 姚 钦, 陈 芳. FEV1多元线性回归模型在肺功能测试中的应用[J]. 南方医科大学学报, 2020, 40(12): 1799-1803. |
| [11] | 钟凯华, 陈 东, 吴志明, 王晓红, 潘 斌, 陈南辉, 钟伟枫. siRNA干扰BIRC6对肾癌786-O细胞凋亡和自噬的影响[J]. 南方医科大学学报, 2020, 40(11): 1651-1655. |
| [12] | 张明洋, 王 静, 李敏超. 薄荷醇增强白介素-13诱导的人支气管上皮细胞粘液蛋白5AC的合成与分泌[J]. 南方医科大学学报, 2020, 40(10): 1432-1438. |
| [13] | 陈海博,李敏超. 薄荷醇通过mTOR活化促进人支气管上皮细胞气道炎症相关因子的表达[J]. 南方医科大学学报, 2019, 39(11): 1344-1349. |
| [14] | 李芳,黄果,彭平,刘瑶,李双辉,刘罗根,章运生. 自噬相关基因3过表达促进乳腺癌细胞自噬并抑制盐霉素诱导的细胞凋亡[J]. 南方医科大学学报, 2019, 39(02): 162-. |
| [15] | 郭维丽,李敏超. 冷刺激敏感哮喘患者的临床及炎症表型特点[J]. 南方医科大学学报, 2019, 39(02): 181-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||