南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 362-373.doi: 10.12122/j.issn.1673-4254.2026.02.14
• • 上一篇
耿召碟1( ), 胡丽1, 代云莉1, 罗荣刚1, 徐韬1, 廖旭阳1, 袁质平2, 王建塔3, 肖瑛1(
), 胡丽1, 代云莉1, 罗荣刚1, 徐韬1, 廖旭阳1, 袁质平2, 王建塔3, 肖瑛1( )
)
收稿日期:2025-04-30
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
肖瑛
E-mail:3303068472@qq.com;yxhx20060725@126.com
作者简介:耿召碟,在读硕士研究生,E-mail: 3303068472@qq.com
基金资助:
Zhaodie GENG1( ), Li HU1, Yunli DAI1, Ronggang LUO1, Tao XU1, Xuyang LIAO1, Zhiping YUAN2, Jianta WANG3, Ying XIAO1(
), Li HU1, Yunli DAI1, Ronggang LUO1, Tao XU1, Xuyang LIAO1, Zhiping YUAN2, Jianta WANG3, Ying XIAO1( )
)
Received:2025-04-30
Online:2026-02-20
Published:2026-03-10
Contact:
Ying XIAO
E-mail:3303068472@qq.com;yxhx20060725@126.com
Supported by:摘要:
目的 探讨树豆酮酸A衍生物XJ-60是否可通过抑制SP1/TGF-β/Smad3信号通路发挥改善非酒精性脂肪肝肝纤维化的作用。 方法 体内实验:12只db/db小鼠采用完全随机设计分成非酒精性脂肪肝模型组(NAFLD组)和给药组(XJ-60组),6只/组;db/m小鼠为正常对照组(NC组),6只/组,XJ-60组给予XJ-60 50 mg/(kg/d)灌胃给药。HE、油红O染色和组织上清液丙氨酸氨基转移酶(ALT)、天门冬氨酸氨基转移酶(AST)判断NAFLD成模情况;免疫组织化学染色观察肝组织中特异性蛋白1(Sp1)、纤连蛋白(FN)的表达部位,Western blotting观察肝组织中Sp1、上皮细胞向间充质细胞转分化(EMT)、细胞外基质(ECM)相关指标的表达情况;ELISA检测肝组织中白介素-6(IL-6)的表达;Sp1与钙黏蛋白(E-ca)和Sp1与α-平滑肌肌动蛋白(α-SMA)进行相关性分析。体外实验:采用CCK8法筛选XJ-60治疗NAFLD的有效浓度;AML12细胞分为正常对照组(NC组),NAFLD模型组(PO组)采用游离脂肪酸刺激复制模型; 溶剂对照组(PO+DMSO组)在PO组的基础上加入与XJ-60相同剂量的DMSO作对照; 药物治疗组(PO+XJ-60组)加入游离脂肪酸后加入不同浓度梯度的XJ-60。油红O染色观察各组肝细胞脂滴沉积情况;Western blotting和Real-time qPCR检测各组SP1、转化生长因子-β(TGF-β)、Smad3、p-Smad3、EMT、ECM相关指标的表达情况;免疫荧光染色观察SP1、p-Smad3定位情况;并通过敲低SP1验证TGF-β/Smad3信号轴在NAFLD中的作用。 结果 体内实验结果表明XJ-60可降低db/db小鼠甘油三酯和总胆固醇(P<0.05);模型组HE、油红O染色以及ALT、AST提示存在肝细胞损伤且肝脏脂滴沉积模型复制成功(P<0.05);NAFLD组小鼠肝脏已出现炎性 细胞浸润和纤维化表型,XJ-60组有所改善;XJ-60组IL-6降低(P<0.05);FN、SP1分别在细胞质和细胞核中表达增多;相关性分析结果提示Sp1和E-ca呈负相关和α-SMA呈正相关(P<0.05)。体外实验中XJ-60治疗NAFLD最佳浓度为10 μmol/L(P<0.01),且PO组AML12细胞中的脂滴沉积增多,XJ-60组脂滴沉积减少(P<0.01);与PO组相比,XJ-60治疗后ECM和EMT相关指标mRNA水平和蛋白水平表达均降低(P<0.05),SP1、TGF-β、p-Smad3蛋白表达水平降低(P<0.05)。敲低SP1可显著降低TGF-β、p-Smad3和ECM的表达(P<0.05)。 结论 XJ-60在db/db小鼠NAFLD肝脏中发挥的抗纤维化作用及改善脂滴沉积的作用机制可能是通过抑制SP1的表达从而抑制TGF-β/Smad3信号通路致纤维化作用,减少下游细胞外基质的分泌和沉积。
耿召碟, 胡丽, 代云莉, 罗荣刚, 徐韬, 廖旭阳, 袁质平, 王建塔, 肖瑛. 树豆酮酸A衍生物XJ-60通过抑制SP1/TGF-β/Smad3信号轴改善非酒精性脂肪肝病小鼠的肝纤维化[J]. 南方医科大学学报, 2026, 46(2): 362-373.
Zhaodie GENG, Li HU, Yunli DAI, Ronggang LUO, Tao XU, Xuyang LIAO, Zhiping YUAN, Jianta WANG, Ying XIAO. Cajanonic acid A derivative XJ-60 improves liver fibrosis in mice with non-alcoholic fatty liver disease by inhibiting the SP1/TGF-β/Smad3 signaling axis[J]. Journal of Southern Medical University, 2026, 46(2): 362-373.
| Primer | Sequence(5´→3´) | Optimal Annealing Te-mperature (℃) |
|---|---|---|
| SP1 | F: TGCCAAATCTACTCCAGCA | 53.6 |
| R: AGCAAGGTGATGTTCCCA | ||
| FN | F: ATGTGGACCCCTCCTGATAGT | 57.3 |
| R: GCCCAGTGATTTCAGCAAAGG | ||
| Col-IV | F: TTTGGCTCGCCACCATAGAG | 60.04 |
| R: GCAGAGGCGAGCATCATAGT | ||
| TGF-β | F: CTCCCGTGGCTTCTAGTGC | 57.2 |
| R: GCCTTAGTTTGGACAGGATCTG | ||
| E-ca | F:CAGGTCTCCTCATGGCTTTGC | 57.7 |
| R:CTTCCGAAAAGAAGGCTGTCC | ||
| α-SMA | F: CCCAGACATCAGGGAGTAATGG | 56.1 |
| R: TCTATCGGATACTTCAGCGTCA | ||
| GAPDH | F: GGTTGTCTCCTGCGACTTCA | 57 |
| R: TGGTCCAGGGTTTCTTACTCC |
表1 Real-time qPCR引物序列
Tab.1 Primer sequence for RT-qPCR
| Primer | Sequence(5´→3´) | Optimal Annealing Te-mperature (℃) |
|---|---|---|
| SP1 | F: TGCCAAATCTACTCCAGCA | 53.6 |
| R: AGCAAGGTGATGTTCCCA | ||
| FN | F: ATGTGGACCCCTCCTGATAGT | 57.3 |
| R: GCCCAGTGATTTCAGCAAAGG | ||
| Col-IV | F: TTTGGCTCGCCACCATAGAG | 60.04 |
| R: GCAGAGGCGAGCATCATAGT | ||
| TGF-β | F: CTCCCGTGGCTTCTAGTGC | 57.2 |
| R: GCCTTAGTTTGGACAGGATCTG | ||
| E-ca | F:CAGGTCTCCTCATGGCTTTGC | 57.7 |
| R:CTTCCGAAAAGAAGGCTGTCC | ||
| α-SMA | F: CCCAGACATCAGGGAGTAATGG | 56.1 |
| R: TCTATCGGATACTTCAGCGTCA | ||
| GAPDH | F: GGTTGTCTCCTGCGACTTCA | 57 |
| R: TGGTCCAGGGTTTCTTACTCC |
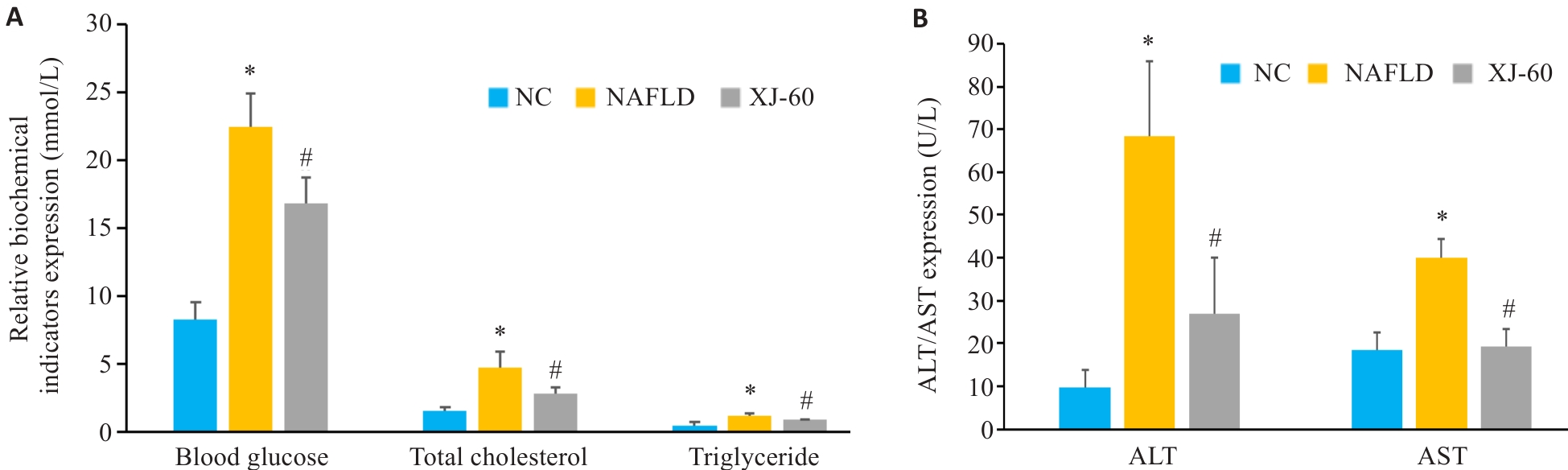
图1 各组小鼠的血糖、甘油三酯和总胆固醇水平及及肝组织上清液ALT及AST表达水平
Fig.1 Levels of blood glucose, triglycerides and total cholesterol and expression levels of ALT and AST in the liver tissue of the mice in each group (n=6, Mean±SD). A: Levels of blood glucose, triglyceride and total cholesterol in different groups. B: Expression levels of ALT and AST in the liver tissue in different groups. *P<0.05 vs NC; #P<0.05 vs NAFLD.
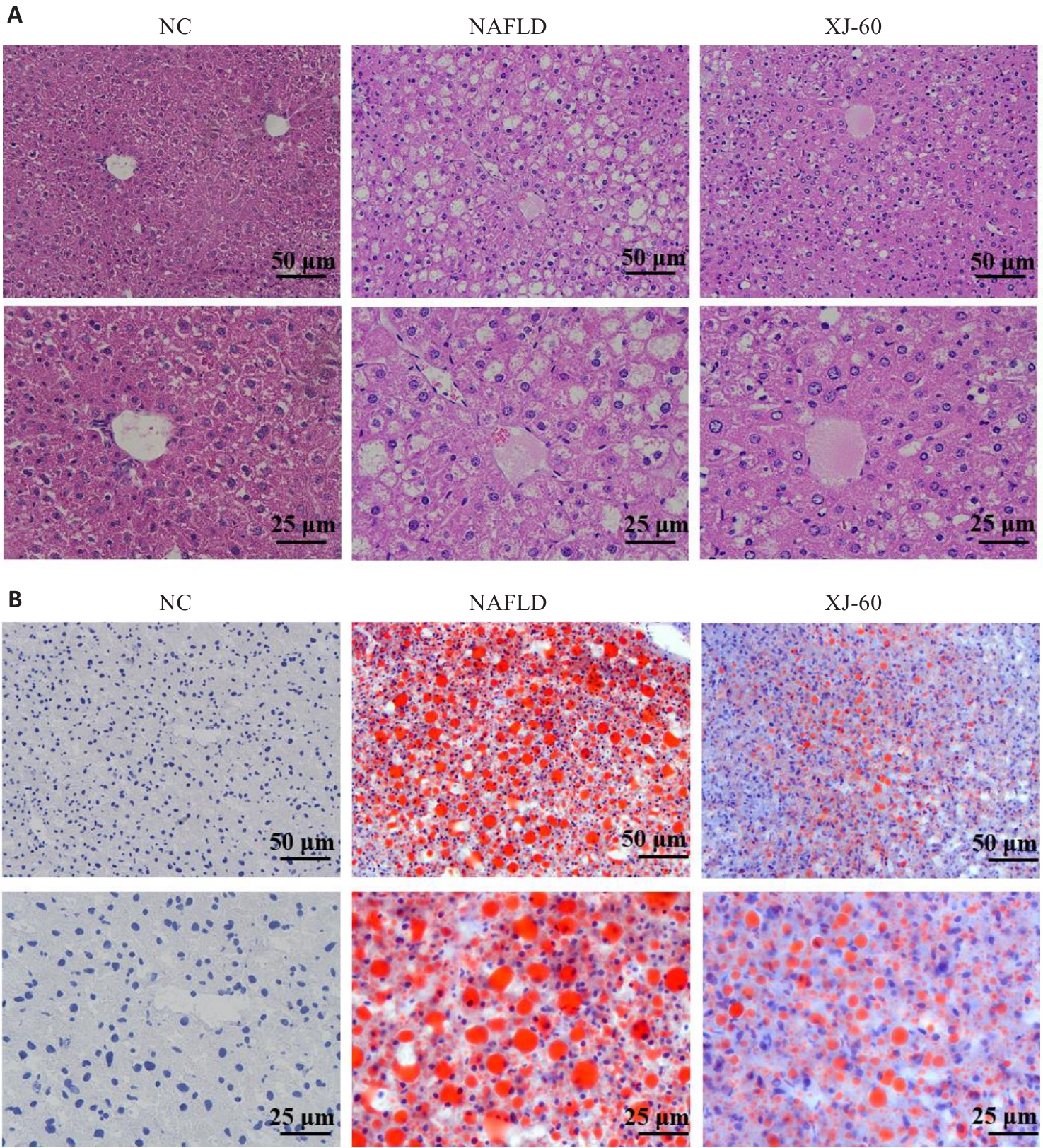
图2 各组肝组织中病理改变及脂肪堆积程度
Fig.2 Pathological changes and fat accumulation in the liver tissue of the mice in each group (n=6, Mean±SD). A: HE staining. B: Oil red O staining.
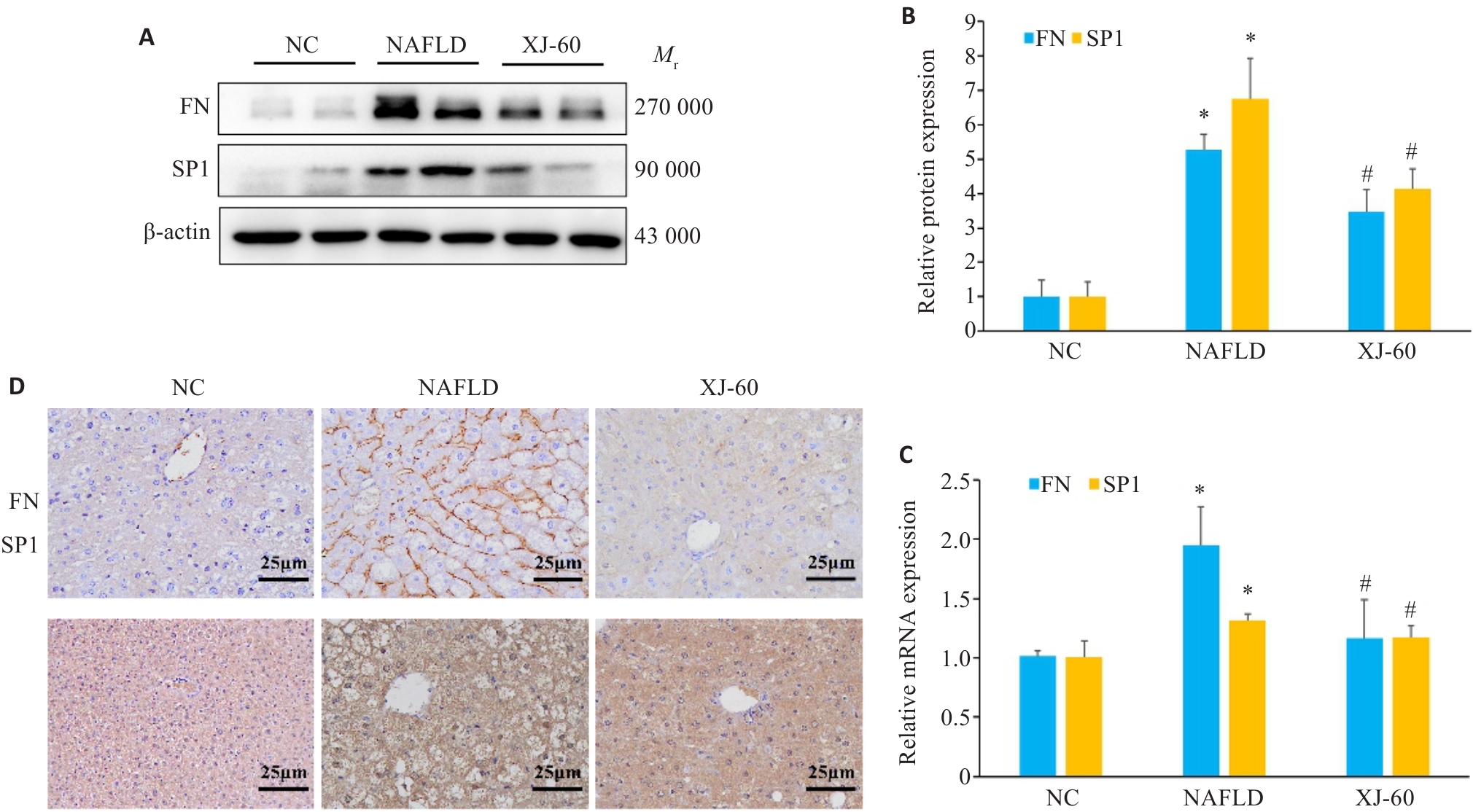
图4 Western blotting、Real-time qPCR、免疫组化染色在各组肝组织中的表达和分布
Fig.4 Protein levels of Sp1 and FN in the liver tissues of the mice in each group determined by Western blotting (n=6, Mean±SD). A, B: Western blotting for detecting protein expression levels of FN and SP1. C: RT-qPCR for detecting expressions of Sp1 and FN mRNA. D: Immunohistochemical staining for Sp1 and FN in the liver tissues of the mice in each group. *P<0.05 vs NC; #P<0.05 vs NAFLD.
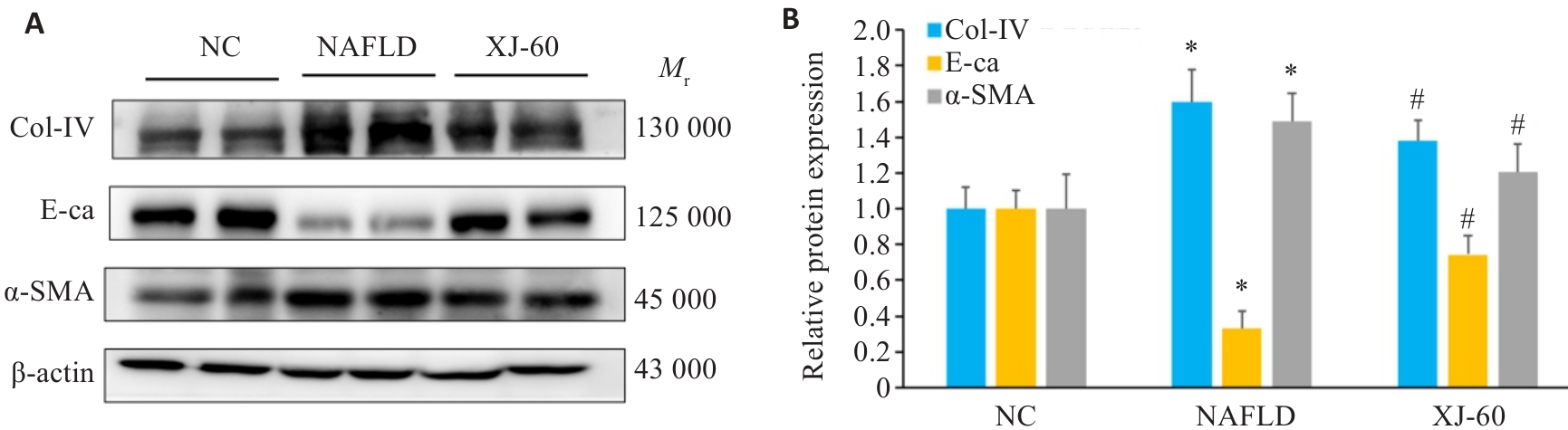
图5 各组肝组织中Col-Ⅳ、E-ca和α-SMA蛋白表达水平
Fig.5 Expression levels of Col-IV, E-ca and α-SMA in the liver tissues of the mice in each group detected by Western blotting (n=6, Mean±SD). *P<0.05 vs NC; #P<0.05 vs NAFLD.
| Protein/Marker | Sp1 |
|---|---|
| Sp1 | 1.000 |
| E-ca | -0.767** |
| α-SMA | 0.777** |
表2 Spearman秩相关性分析
Tab.2 Spearman rank correlation analysis
| Protein/Marker | Sp1 |
|---|---|
| Sp1 | 1.000 |
| E-ca | -0.767** |
| α-SMA | 0.777** |
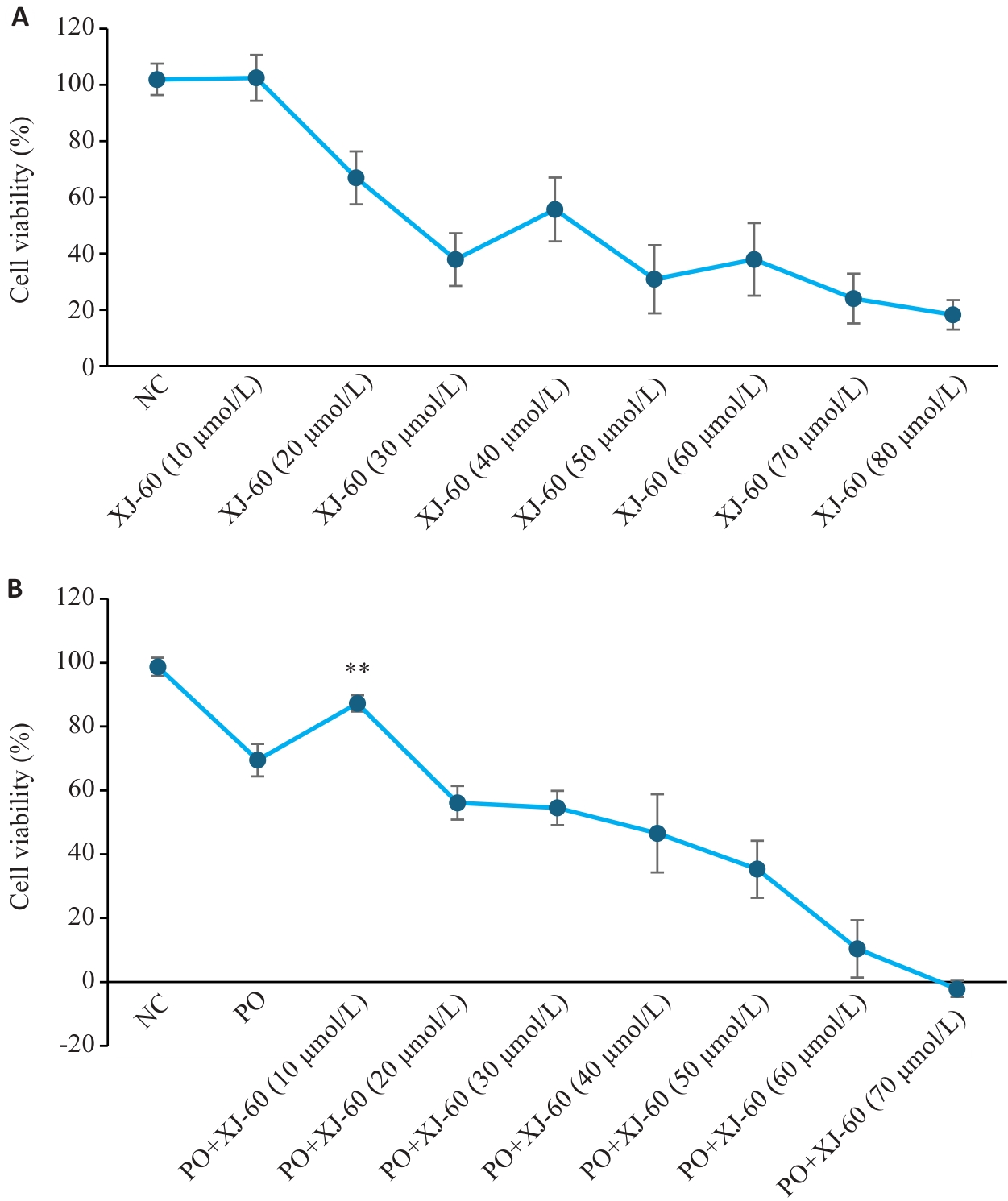
图6 CCK8实验中各分组细胞存活率对比图
Fig.6 Comparison of cell survival rate of each group in CCK8 experiment (n=3, Mean±SD). A: Screening the safe concentration range of XJ-60 acting on AML12 cells without FFA intervention, P>0.05 vs NC. B: The optimal therapeutic concentration of XJ-60 acting on AML12 cells under FFA intervention. **P<0.01 vs PO.
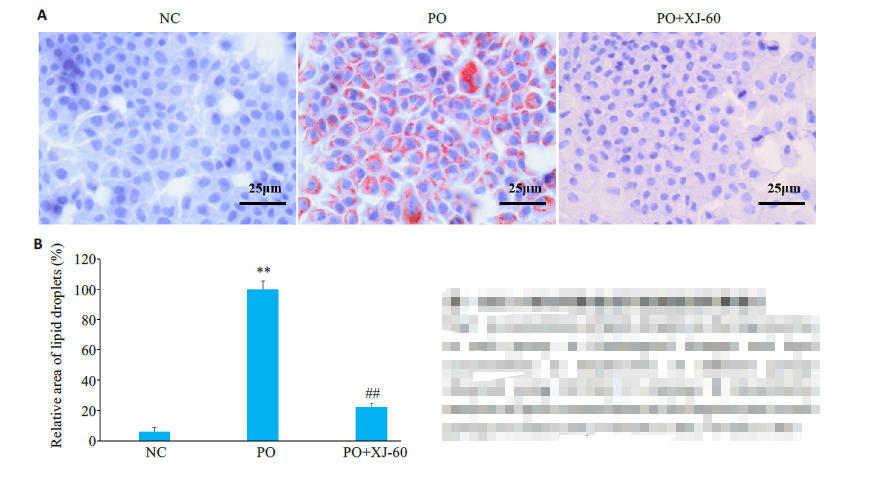
图7 油红O染色观察各组肝细胞脂滴积累情况
Fig.7 Accumulation of lipid droplets in the hepatocytesin each group. A: Pathological changes and lipidaccumulation in hepatocytes of different groupsobserved using Oil Red O staining. B: Quantitativeanalysis of lipid accumulation in the hepatocytes in eachgroup (n=3, Mean±SD), **P<0.01 vs NC; ##P<0.01 vs PO.
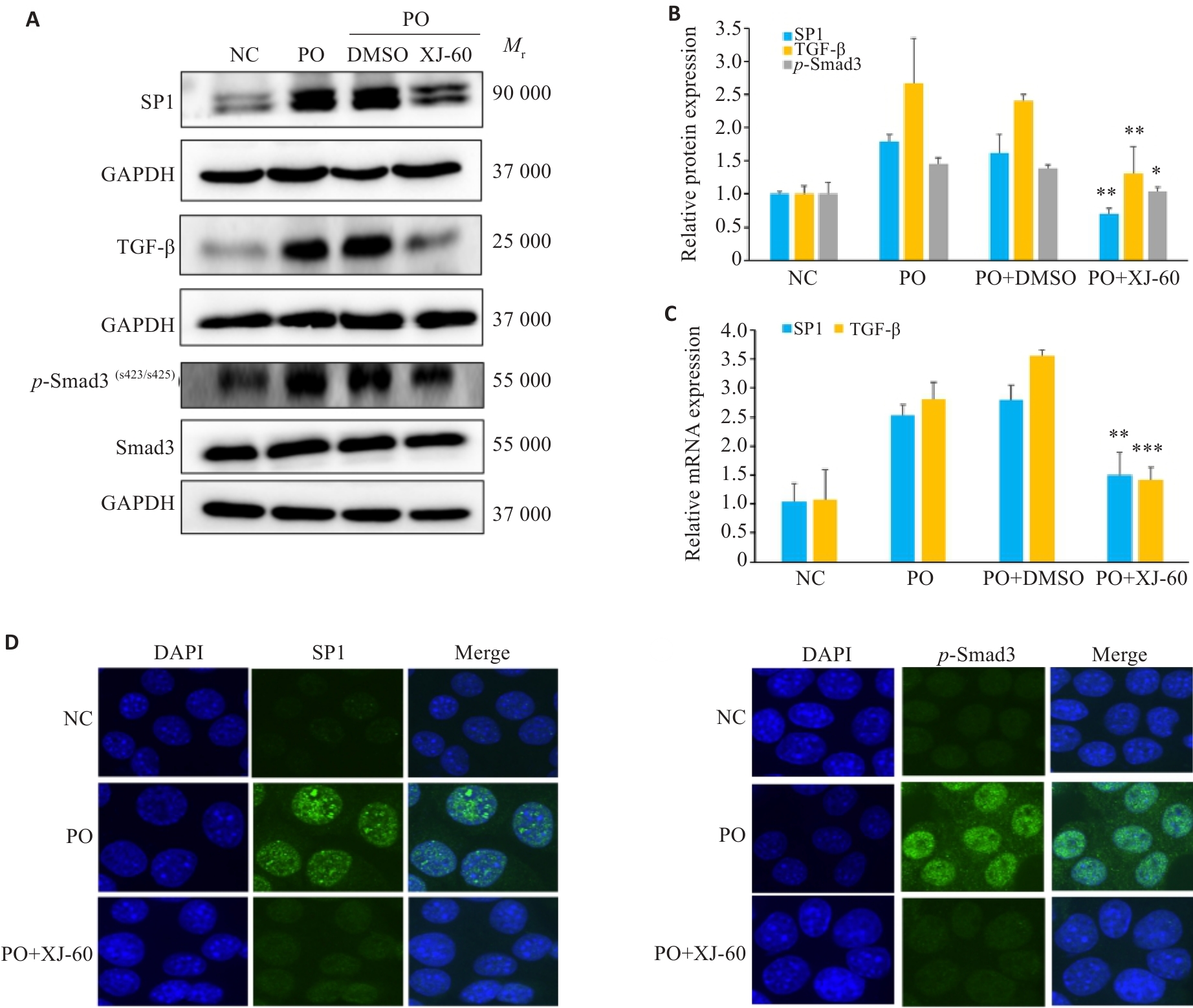
图8 XJ-60对AML12细胞中SP1/TGF-β/Smad3信号通路的影响
Fig.8 Effect of XJ-60 on expressions of SP1/TGF-β/Smad3 signaling pathway in FFA-induced AML12 cells (n=3, Mean±SD). A, B: Protein expression levels of SP1, TGF-β and p-Smad3 in AML12 cells in each group (*P<0.05, **P<0.01 vs PO+DMSO). C: mRNA expression levels of SP1, TGF-β and p-Smad3 in hepatocytes of each group (**P<0.01,***P<0.001 vs PO+DMSO). D: SP1 and p-Smad3 immunofluorescence staining of AML12 cells in each group (×1000).
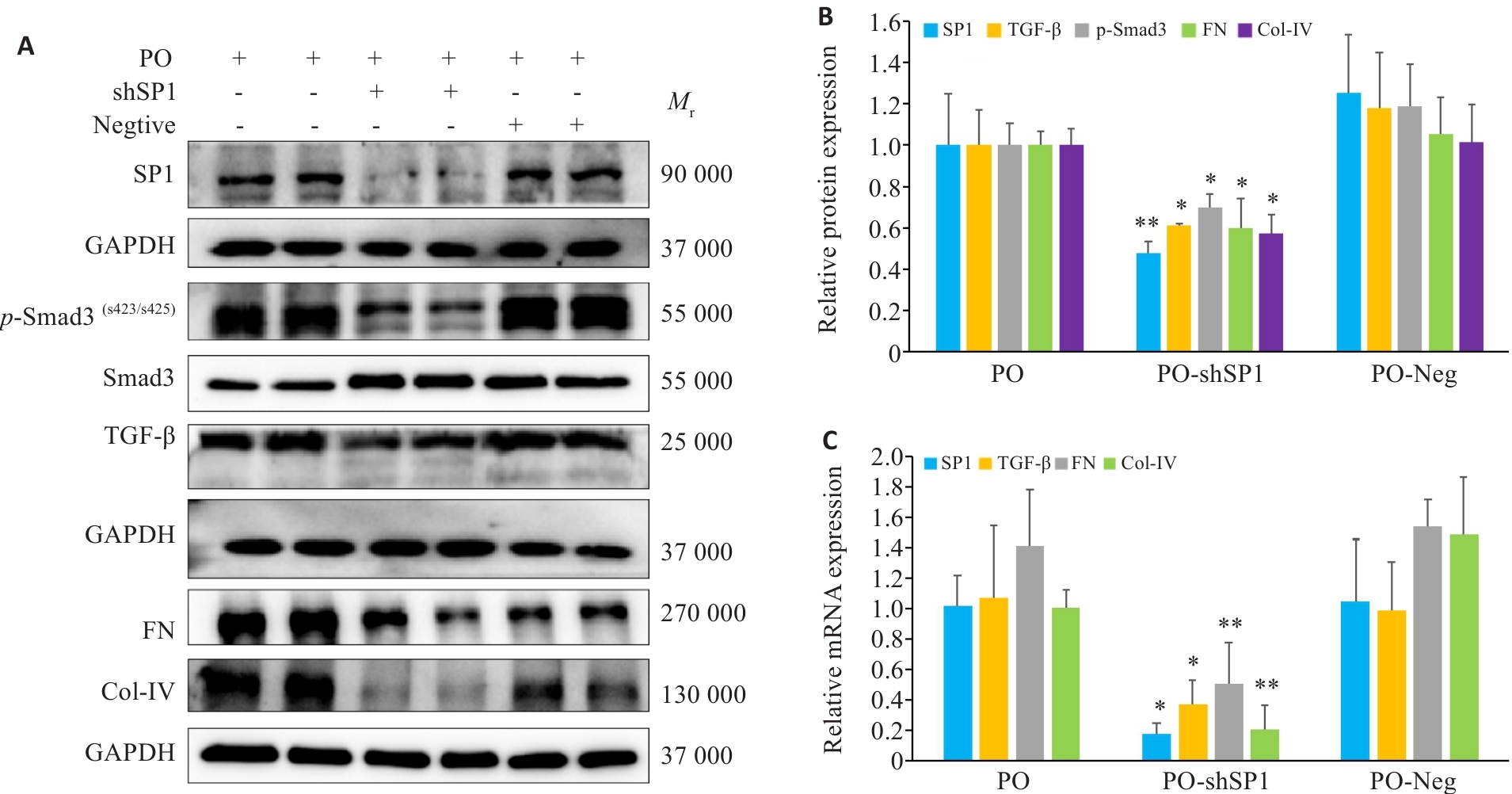
图9 敲低SP1后各指标的表达变化
Fig.9 Changes in expressions of the protein markers in AML12 cells after SP1 knockdown (n=3, Mean±SD). A, B: Protein expression levels of FN, Col-IV, SP1, TGF‑β and p-Smad3 in AML12 cells (*P<0.05, **P<0.01 vs PO+Neg). C: mRNA expression levels of FN, Col-IV, SP1, TGF-β and p-Smad3 in AML12 cells in each group (*P<0.05, **P<0.01 vs PO+Neg).
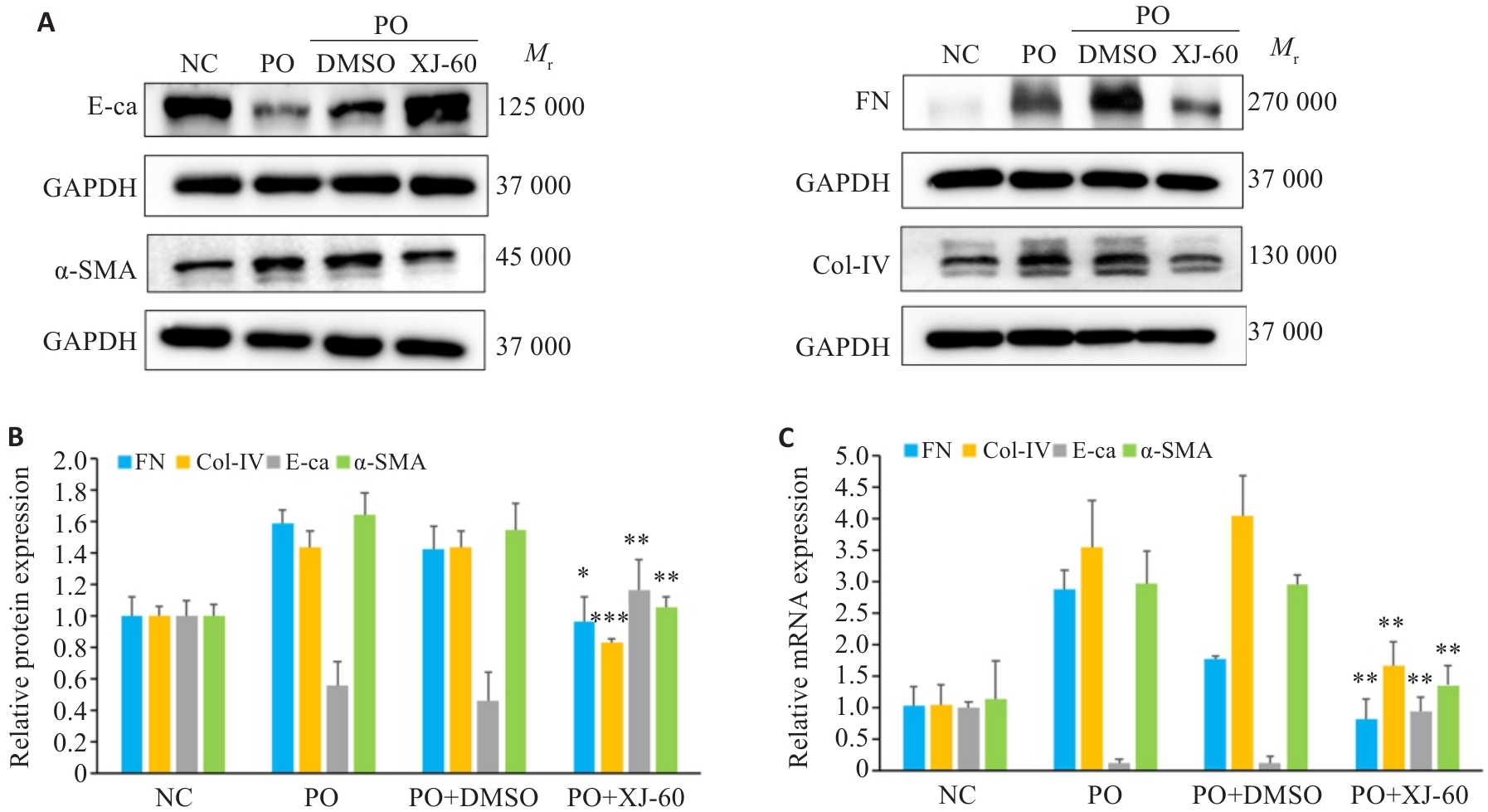
图10 XJ-60对EMT及ECM的改善情况
Fig.10 Effect of XJ-60 treatment on expressions of EMT and ECM markers in AML12 cells (n=3, Mean±SD). A, B: Protein expression levels of FN, Col-IV, E-ca and α-SMA in AML12 cells in each group (*P<0.05, **P<0.01, ***P<0.001 vs PO+DMSO). C: mRNA expression levels of FN, Col-IV, E-ca and α-SMA in hepatocytes of each group (**P<0.01 vs PO+DMSO).
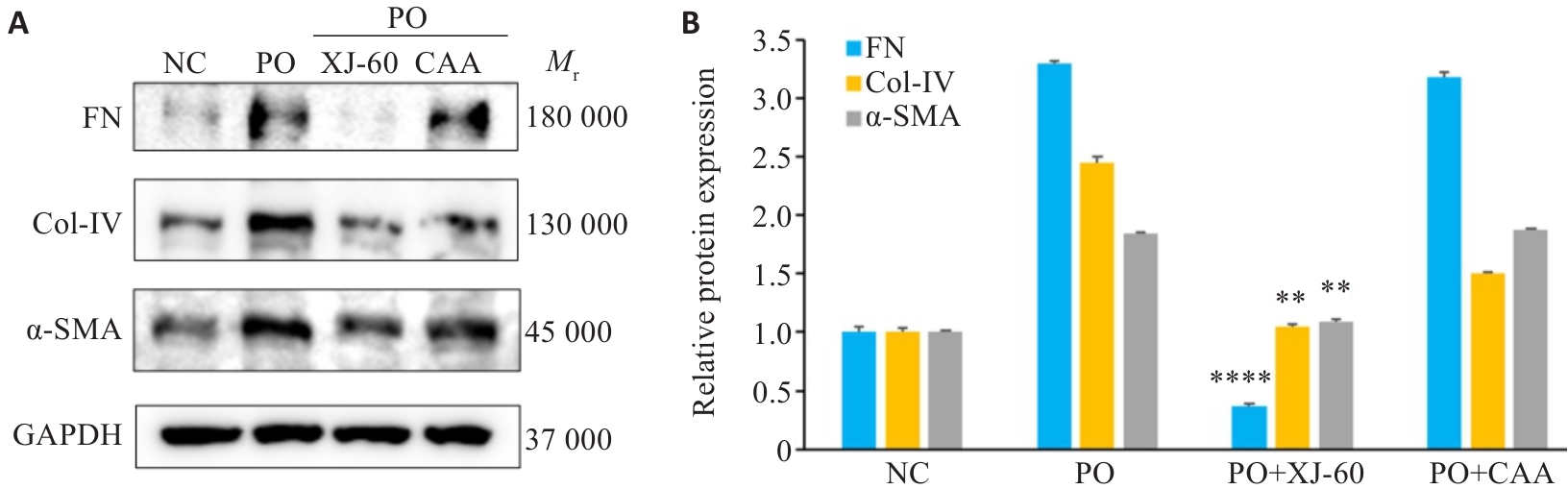
图11 CAA与XJ-60对AML12细胞纤维化的改善情况
Fig.11 Effects of CAA and XJ-60 for improving fibrosis in AML12 cells (n=3, Mean±SD). A: Protein bands of FN, Col-IV and α-SMA in AML12 cells in each group. B: Relative protein expression levels. **P<0.01, ****P<0.0001 vs PO+CAA.
| [1] | Powell EE, Wong VW, Rinella M. Non-alcoholic fatty liver disease[J]. Lancet, 2021, 397(10290): 2212-24. doi:10.1016/s0140-6736(20)32511-3 |
| [2] | Brunt EM, Kleiner DE, Carpenter DH, et al. NAFLD: reporting histologic findings in clinical practice[J]. Hepatology, 2021, 73(5): 2028-38. doi:10.1002/hep.31599 |
| [3] | Rodríguez-Lara A, Rueda-Robles A, Sáez-Lara MJ, et al. From non-alcoholic fatty liver disease to liver cancer: microbiota and inflammation as key players[J]. Pathogens, 2023, 12(7): 940-55. doi:10.3390/pathogens12070940 |
| [4] | Zhou Y, Zhang L, Ma Y, et al. Secretome of senescent hepatic stellate cells favors malignant transformation from nonalcoholic steatohepatitis-fibrotic progression to hepatocellular carcinoma[J]. Theranostics, 2023, 13(13): 4430-48. doi:10.7150/thno.85369 |
| [5] | Martínez-Espinosa I, Serrato JA, Cabello-Gutiérrez C, et al. Mechanisms of microRNA regulation of the epithelial-mesenchymal transition (EMT) in lung cancer[J]. Life: Basel, 2024, 14(11): 1431. doi:10.3390/life14111431 |
| [6] | Nair B, Nath LR. Inevitable role of TGF‑β1 in progression of nonalcoholic fatty liver disease[J]. J Recept Signal Transduct Res, 2020, 40(3): 195-200. doi:10.1080/10799893.2020.1726952 |
| [7] | Chen C, Chen J, Wang Y, et al. Ganoderma lucidum polysaccharide inhibits HSC activation and liver fibrosis via targeting inflammation, apoptosis, cell cycle, and ECM-receptor interaction mediated by TGF-β/Smad signaling[J]. Phytomedicine, 2023, 110: 154626. doi:10.1016/j.phymed.2022.154626 |
| [8] | Xin X, Cheng X, Zeng F, et al. The role of TGF-β/SMAD signaling in hepatocellular carcinoma: from mechanism to therapy and prognosis[J]. Int J Biol Sci, 2024, 20(4): 1436-51. doi:10.7150/ijbs.89568 |
| [9] | Sun Y, Chen X, Chen L, et al. MFAP2 promotes HSCs activation through FBN1/TGF-β/Smad3 pathway[J]. J Cell Mol Med, 2023, 27(21): 3235-46. doi:10.1111/jcmm.17884 |
| [10] | Ding J, Fayyaz AI, Ding Y, et al. Role of specificity protein 1 (SP1) in cardiovascular diseases: pathological mechanisms and therapeutic potentials[J]. Biomolecules, 2024, 14(7): 807-21. doi:10.3390/biom14070807 |
| [11] | Zhou W, Fang J, Jia Q, et al. Transcription factor specificity protein (SP) family in renal physiology and diseases[J]. PeerJ, 2025, 13: e18820. doi:10.7717/peerj.18820 |
| [12] | Shen ZJ, Zhang YZ, Bu GK, et al. Renal denervation improves mitochondrial oxidative stress and cardiac hypertrophy through inactivating SP1/BACH1-PACS2 signaling[J]. Int Immunophar-macol, 2024, 141: 112778. doi:10.1016/j.intimp.2024.112778 |
| [13] | Liu X, Gong Q, Yang L, et al. microRNA-199a-5p regulates epithelial-to-mesenchymal transition in diabetic cataract by targeting SP1 gene[J]. Mol Med, 2020, 26(1): 122-222. doi:10.1186/s10020-020-00250-7 |
| [14] | Firneisz G. Non-alcoholic fatty liver disease and type 2 diabetes mellitus: the liver disease of our age[J]? World J Gastroenterol,2014, 20(27):9072-89. |
| [15] | Zeng J, Fan JG, Francque SM. Therapeutic management of metabolic dysfunction associated steatotic liver disease[J]. United European Gastroenterol J, 2024, 12(2): 177-86. doi:10.1002/ueg2.12525 |
| [16] | Mantovani A, Byrne CD, Targher G. Efficacy of peroxisome proliferator-activated receptor agonists, glucagon-like peptide-1 receptor agonists, or sodium-glucose cotransporter-2 inhibitors for treatment of non-alcoholic fatty liver disease: a systematic review[J]. Lancet Gastroenterol Hepatol, 2022, 7(4): 367-78. doi:10.1016/s2468-1253(21)00261-2 |
| [17] | Liu J, Wang LN. Peroxisome proliferator-activated receptor gamma agonists for preventing recurrent stroke and other vascular events in people with stroke or transient ischaemic attack[J]. Cochrane Database Syst Rev, 2023, 1: CD010693. doi:10.1002/14651858.cd010693.pub6 |
| [18] | Yang R, Wang L, Xie J, et al. Treatment of type 2 diabetes mellitus via reversing insulin resistance and regulating lipid homeostasis in vitro and in vivo using cajanonic acid A[J]. Int J Mol Med, 2018, 42(5): 2329-42. |
| [19] | Loomba R, Friedman SL, Shulman GI. Mechanisms and disease consequences of nonalcoholic fatty liver disease[J]. Cell, 2021, 184(10): 2537-64. doi:10.1016/j.cell.2021.04.015 |
| [20] | Nevens F, Andreone P, Mazzella G, et al. A placebo-controlled trial of obeticholic acid in primary biliary cholangitis[J]. N Engl J Med, 2016, 375(7): 631-43. doi:10.1056/nejmc1611913 |
| [21] | Kamata S, Honda A, Ishii I. Current clinical trial status and future prospects of PPAR-targeted drugs for treating nonalcoholic fatty liver disease[J]. Biomolecules, 2023, 13(8): 1264-85. doi:10.3390/biom13081264 |
| [22] | 王 璐. 树豆酮酸A调节血糖血脂的作用和机理研究[D]. 广州中医药大学, 2012. |
| [23] | 沈小玲, 王 璐, 胡英杰, 等.树豆酮酸A在制备糖尿病伴随症及高脂血症药物中的应用: CN201210119665.5[P].CN 102670576A[2025-12-11]. |
| [24] | 谢 洁. 树豆酮酸A改善胰岛素抵抗的作用研究[D].广州中医药大学, 2014. |
| [25] | 王一凡, 吕厚星, 李志阳, et al. 树豆酮酸A可减轻db/db小鼠非酒精性脂肪肝损伤 [J]. 中国生物化学与分子生物学报, 2024, 40(3): 373-82. |
| [26] | Yao Y, Luo ZP, Li HW, et al. P38γ modulates the lipid metabolism in non-alcoholic fatty liver disease by regulating theJAK-STATsignaling pathway[J]. FASEB J, 2023, 37(1): e22716. doi:10.1096/fj.202200939rr |
| [27] | Cai W, Wu S, Ming X, et al. IL6 Derived from Macrophages under Intermittent Hypoxia Exacerbates NAFLD by Promoting Ferroptosis via MARCH3-Led Ubiquitylation of GPX4 [J]. Advanced science (Weinheim, Baden-Wurttemberg, Germany), 2024, 11(41): e2402241. doi:10.1002/advs.202402241 |
| [28] | Zhang HC, Zhang GP, Zhang JY, et al. Transcription factor SP1 and oncoprotein PPP1R13L regulate nicotine-induced epithelial-mesenchymal transition in lung adenocarcinoma via a feedback loop[J]. Biochem Pharmacol, 2022, 206: 115344. doi:10.1016/j.bcp.2022.115344 |
| [29] | Xie J, Lin H, Jin F, et al. Jia Wei Qingxin Lotus Seed Drink ameliorates epithelial mesenchymal transition injury in diabetic kidney disease via inhibition of JMJD1C/SP1/ZEB1 signaling pathway[J]. Phytomedicine, 2024, 135: 156142. doi:10.1016/j.phymed.2024.156142 |
| [30] | Song LY, Huang YL, Liu L, et al. Meteorin-like alleviates hepatic steatosis by regulating hepatic triglyceride secretion and fatty acid oxidation[J]. Cell Rep, 2025, 44(2): 115246. doi:10.1016/j.celrep.2025.115246 |
| [31] | Li Y, Chen R, Wu J, et al. Salvianolic acid B protects against pulmonary fibrosis by attenuating stimulating protein 1-mediated macrophage and alveolar type 2 cell senescence[J]. Phytother Res, 2024, 38(2): 620-35. doi:10.1002/ptr.8070 |
| [32] | Zhu QQ, Yang XY, Zhang XJ, et al. EGCG targeting Notch to attenuate renal fibrosis via inhibition of TGFβ/Smad3 signaling pathway activation in streptozotocin-induced diabetic mice[J]. Food Funct, 2020, 11(11): 9686-95. doi:10.1039/d0fo01542c |
| [33] | Ren Q, Wang B, Guo F, et al. Natural flavonoid pectolinarigenin alleviated hyperuricemic nephropathy via suppressing TGFβ/SMAD3 and JAK2/STAT3 signaling pathways[J]. Front Pharmacol, 2021, 12: 792139. doi:10.3389/fphar.2021.792139 |
| [34] | Ding L, Li Y, Yang Y, et al. Wenfei buqi Tongluo formula against bleomycin-induced pulmonary fibrosis by inhibiting TGF-β/Smad3 pathway[J]. Front Pharmacol, 2021, 12: 762998. doi:10.3389/fphar.2021.762998 |
| [35] | Ding A, Bian YY, Zhang ZH. SP1/TGF‑β1/SMAD2 pathway is involved in angiogenesis during osteogenesis[J]. Mol Med Rep, 2020, 21(3): 1581-9. |
| [36] | Yang ZS, Zhang H, Yin MH, et al. TGF‑β1/Smad3 upregulates UCA1 to promote liver fibrosis through DKK1 and miR18a[J]. J Mol Med, 2022, 100(10): 1465-78. doi:10.1007/s00109-022-02248-6 |
| [37] | Wang J, Fan Z, Li J, et al. Transcription factor specificity protein 1-mediated Serine/threonine kinase 39 upregulation promotes the proliferation, migration, invasion and epithelial-mesenchymal transition of hepatocellular carcinoma cells by activating the transforming growth factor‑β1/Smad2/3 pathway[J]. Bioengineered, 2021, 12(1): 3566-77. doi:10.1080/21655979.2021.1947939 |
| [38] | Li YJ, Fan WG, Link F, et al. Transforming growth factor β latency: a mechanism of cytokine storage and signalling regulation in liver homeostasis and disease[J]. JHEP Rep, 2022, 4(2): 100397. doi:10.1016/j.jhepr.2021.100397 |
| [1] | 于滢, 涂丽, 刘洋, 宋雪翼, 邵倩倩, 唐小龙. TGF-β通过miR-23a-3p/IRF1轴下调主要组织相容性复合体I类表达促进肝癌免疫逃逸[J]. 南方医科大学学报, 2025, 45(7): 1397-1408. |
| [2] | 石情, 冉苏叶, 宋铃榆, 杨红, 王文娟, 刘晗琳, 刘琦. NLRP6过表达通过AMPK/CPT1A/PGC1A通路促进肝细胞脂肪氧化分解改善非酒精性脂肪肝[J]. 南方医科大学学报, 2025, 45(1): 118-125. |
| [3] | 朱梦云, 王剑锋. 康柏西普可逆转TGF-β2诱导的晶状体上皮细胞发生上皮间质转化:基于调节TGF-β/Smad信号通路[J]. 南方医科大学学报, 2024, 44(8): 1459-1466. |
| [4] | 陈光亚, 向星亮, 曾兆祥, 黄荣增, 金姝娜, 肖明中, 宋成武. 地五养肝方对非酒精性脂肪肝小鼠循环外泌体溶血甘油磷脂的调控作用[J]. 南方医科大学学报, 2024, 44(7): 1382-1388. |
| [5] | 孔 祥, 张 腾, 张 妍, 高灵犀, 汪 文, 汪梦燕, 王国栋, 吕 坤. 过表达lncRNA HEM2M改善非酒精性脂肪肝病小鼠的肝脏损伤[J]. 南方医科大学学报, 2024, 44(1): 1-8. |
| [6] | 徐龙飞, 韩 晶, 杨 喆, 杨燕平, 陈金慧, 吴西军, 王 琪, 洪 艳. LRG1抑制小鼠肝巨噬细胞活化从而改善代谢相关脂肪性肝病:基于增强TGF-β1信号通路[J]. 南方医科大学学报, 2023, 43(7): 1164-1171. |
| [7] | 熊紫玉, 周乐鹏, 陈静芬, 李 萌, 谢日华. 产后抑郁与初乳中转化生长因子-β水平的关系:90例巢氏队列研究[J]. 南方医科大学学报, 2022, 42(9): 1426-1430. |
| [8] | 朱雅莉, 章述军, 阳 成, 薛 薇, 张 佳, 李佳俊, 赵金秋, 徐 静, 黄文祥. 非酒精性脂肪性肝病患者的肝组织差异蛋白的定量分析:基于iTRAQ技术[J]. 南方医科大学学报, 2021, 41(9): 1381-1387. |
| [9] | 陈 旭, 申 颖, 赵海霞, 郭文奇. 吡非尼酮通过下调TGF-β/Smad通路中TGF-β3的表达抑制兔Tenons囊成纤维细胞增殖[J]. 南方医科大学学报, 2021, 41(11): 1692-1699. |
| [10] | 卢家美,张晶晶,吕 毅,王 博,贾丽娜,师建华. 二甲双胍抑制大鼠胆管成纤维细胞胶原生成的分子信号机制[J]. 南方医科大学学报, 2020, 40(05): 640-646. |
| [11] | 王 珊,张 健,张卫欢,汪海涛,侯婧悦,张瑞秀,刘红芬,吴寿岭. 体质量指数联合腰围对 2型糖尿病患者新发非酒精性脂肪肝的预测价值[J]. 南方医科大学学报, 2019, 39(11): 1293-1297. |
| [12] | 白志勋,陆静,杨亦彬. TGF-β1/ILK/FSP1信号通路在环孢素诱导肾小管上皮细胞转分化中的作用[J]. 南方医科大学学报, 2019, 39(07): 804-. |
| [13] | 杨妙婷,陈芝娟,肖淳欣,唐外姣,周本杰. 护肝清脂片药物血清对非酒精性脂肪肝细胞模型内质网应激的影响[J]. 南方医科大学学报, 2018, 38(11): 1277-. |
| [14] | 李仕良,王洁,黄鹏,古贤君,黄美英,黄非凡. TGF-β基因启动子-509C/T位点多态性与桂西壮族IgA肾病核心家系的关联性及其铁皮石斛的治疗效果[J]. 南方医科大学学报, 2018, 38(08): 986-. |
| [15] | 姚笑睿,夏凡,唐外姣,周本杰. 护肝清脂片对非酒精性脂肪肝大鼠肝脏中AMPK通路激活及NF-κB- p65 蛋白的影响[J]. 南方医科大学学报, 2017, 37(01): 56-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||