Journal of Southern Medical University ›› 2025, Vol. 45 ›› Issue (7): 1372-1379.doi: 10.12122/j.issn.1673-4254.2025.07.03
Previous Articles Next Articles
Zhengyuan FAN1,2,3( ), Zihan SHEN3, Ya LI1,2,3, Tingting SHEN1,2,3, Gaofeng LI3, Suyun LI2,3(
), Zihan SHEN3, Ya LI1,2,3, Tingting SHEN1,2,3, Gaofeng LI3, Suyun LI2,3( )
)
Received:2025-03-21
Online:2025-07-20
Published:2025-07-17
Contact:
Suyun LI
E-mail:fanzhengyuan0225@163.com;lisuyun2000@126.com
Supported by:Zhengyuan FAN, Zihan SHEN, Ya LI, Tingting SHEN, Gaofeng LI, Suyun LI. Protective effect of Bufei Yishen Formula against cigarette smoke extract-induced human bronchial epithelial cell damage and its mechanism[J]. Journal of Southern Medical University, 2025, 45(7): 1372-1379.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2025.07.03
| Group | Intervention methods |
|---|---|
| Control | DMEM+10% blank serum |
| CSE | DMEM+10%CSE+10% blank serum |
| BYL | DMEM+10%CSE+5% medicated serum+5%blank serum |
| BYH | DMEM+10%CSE+10% medicated serum |
| PDTC | DMEM+10%CSE+10% blank serum+10 μmol/L PDTC |
| BYH+PDTC | DMEM+10%CSE+10% medicated serum+10 μmol/L PDTC |
| S-CMC | DMEM+10%CSE+10% blank serum+10 μmol/L S-CMC |
Tab.1 Treatment protocols in each group
| Group | Intervention methods |
|---|---|
| Control | DMEM+10% blank serum |
| CSE | DMEM+10%CSE+10% blank serum |
| BYL | DMEM+10%CSE+5% medicated serum+5%blank serum |
| BYH | DMEM+10%CSE+10% medicated serum |
| PDTC | DMEM+10%CSE+10% blank serum+10 μmol/L PDTC |
| BYH+PDTC | DMEM+10%CSE+10% medicated serum+10 μmol/L PDTC |
| S-CMC | DMEM+10%CSE+10% blank serum+10 μmol/L S-CMC |
| Gene | Primer sequence (5'-3') | Length(bp) |
|---|---|---|
| TLR4 | Forward: CTCCCTGGTGTTGGATTTTACG Reverse: CTCGTTTCTCACCCAGTCCTCA | 226 |
| I-κB | Forward: CCCAAGTACCCGGATACAGCA Reverse: GTCATCGTAGGGCAACTCATCTT | 133 |
| NF-κB | Forward: GTATTGCTGTGCCTTCCCGA Reverse: CAAGGCCTGGTTTGAGATCTG | 146 |
| AQP5 | Forward: AGCCCCTCTCACTGGGTCTT Reverse: CTCTCGATGATCTTCCCAGTCC | 165 |
| MUC5AC | Forward: GGTCTTCTACTTCCCTGGTCTGTG Reverse: GTCAGCTCAACAACTAGGCCATC | 159 |
| MUC5B | Forward: CAGGCTAGTCCTCAACTTCCTGT Reverse: GTGTTGTGGGCGTAGAACTCATT | 141 |
Tab.2 Primer sequence for RT-qPCR
| Gene | Primer sequence (5'-3') | Length(bp) |
|---|---|---|
| TLR4 | Forward: CTCCCTGGTGTTGGATTTTACG Reverse: CTCGTTTCTCACCCAGTCCTCA | 226 |
| I-κB | Forward: CCCAAGTACCCGGATACAGCA Reverse: GTCATCGTAGGGCAACTCATCTT | 133 |
| NF-κB | Forward: GTATTGCTGTGCCTTCCCGA Reverse: CAAGGCCTGGTTTGAGATCTG | 146 |
| AQP5 | Forward: AGCCCCTCTCACTGGGTCTT Reverse: CTCTCGATGATCTTCCCAGTCC | 165 |
| MUC5AC | Forward: GGTCTTCTACTTCCCTGGTCTGTG Reverse: GTCAGCTCAACAACTAGGCCATC | 159 |
| MUC5B | Forward: CAGGCTAGTCCTCAACTTCCTGT Reverse: GTGTTGTGGGCGTAGAACTCATT | 141 |
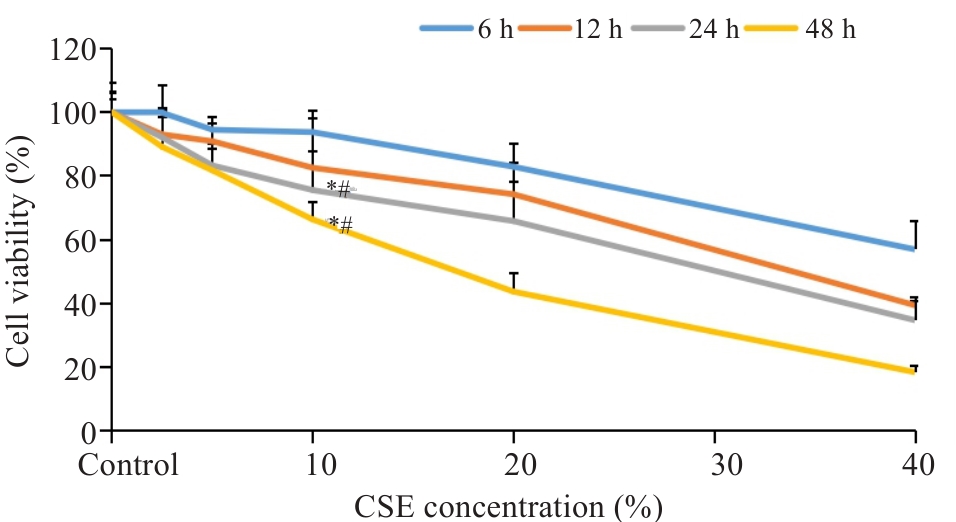
Fig.1 Survival rate of BEAS-2B cells treated with different concentrations of cigarette smoke extract (CSE). *P<0.05 vs Control; #P<0.05 vs 10% CSE for 6 h.
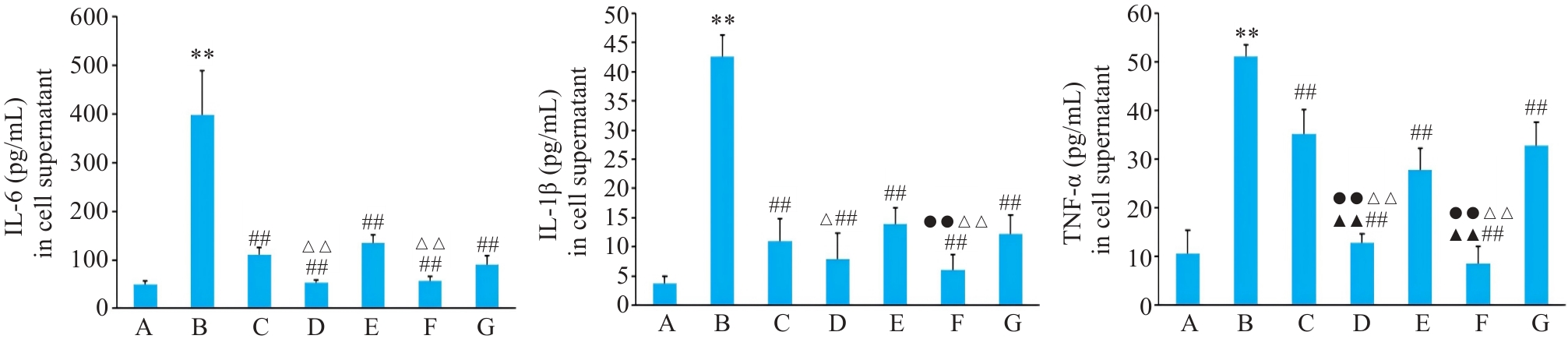
Fig.3 Expressions of IL-6, IL-1β and TNF-α in BEAS-2B cells with different treatments. A: Control group. B: CSE treatment group. C: Low-dose BYF-medicated serum group. D: High-dose BYF-medicated serum group. E: PDTC treatment group. F: High-dose BYF-medicated serum and PDTC treatment group. G: S-CMC treatment group. **P<0.01 vs A; ##P<0.01 vs B; ▲▲P<0.01 vs C; △P<0.05, △△P<0.01 vs E; ●●P<0.01 vs G.
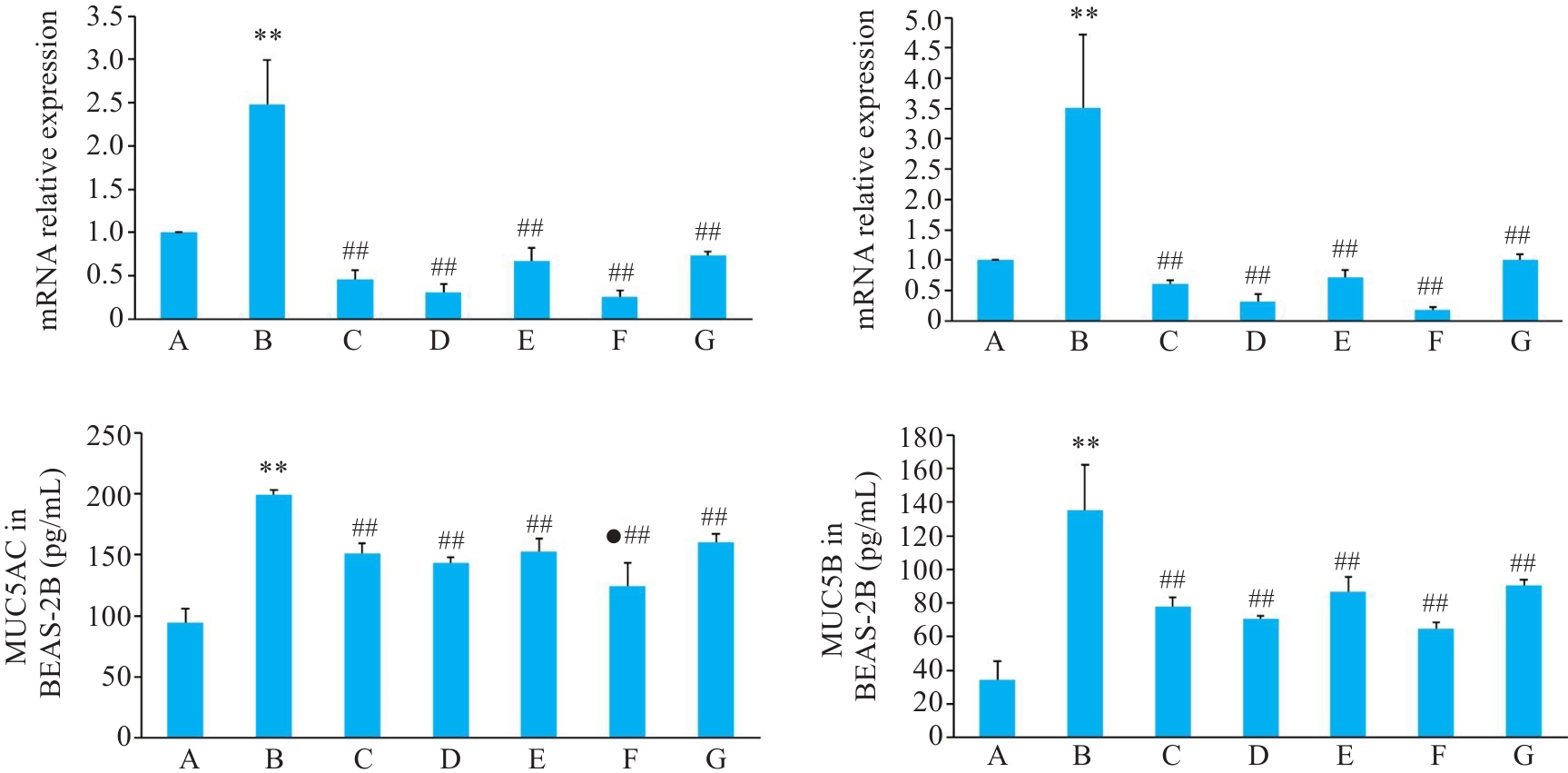
Fig.4 Relative mRNA and protein expressions of MUC5AC and MUC5B in BEAS-2B cells with different treatments. A: Control group. B: CSE treatment group. C: Low-dose BYF-medicated serum group. D: High-dose BYF-medicated serum group. E: PDTC treatment group. F: High-dose BYF-medicated serum and PDTC treatment group. G: S-CMC treatment group. **P<0.01 vs A; ##P<0.01 vs B; ●P<0.05 vs G.
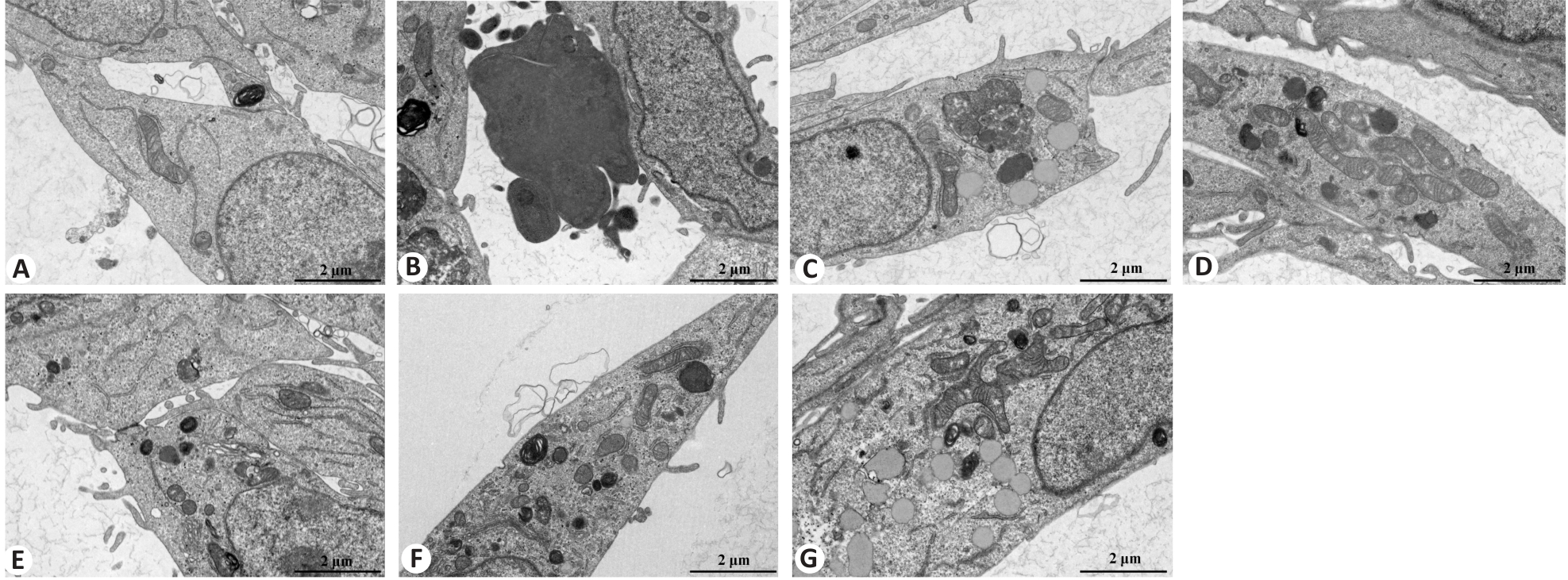
Fig.5 Transmission electron microscopy for examining ultrastructural changes in BEAS-2B cells following different treatments (Scale bar=2 μm). A: Control group. B: CSE treatment group. C: Low-dose BYF-medicated serum group. D: High-dose BYF-medicated serum group. E: PDTC treatment group. F: High-dose BYF-medicated serum and PDTC treatment group. G: S-CMC treatment group.
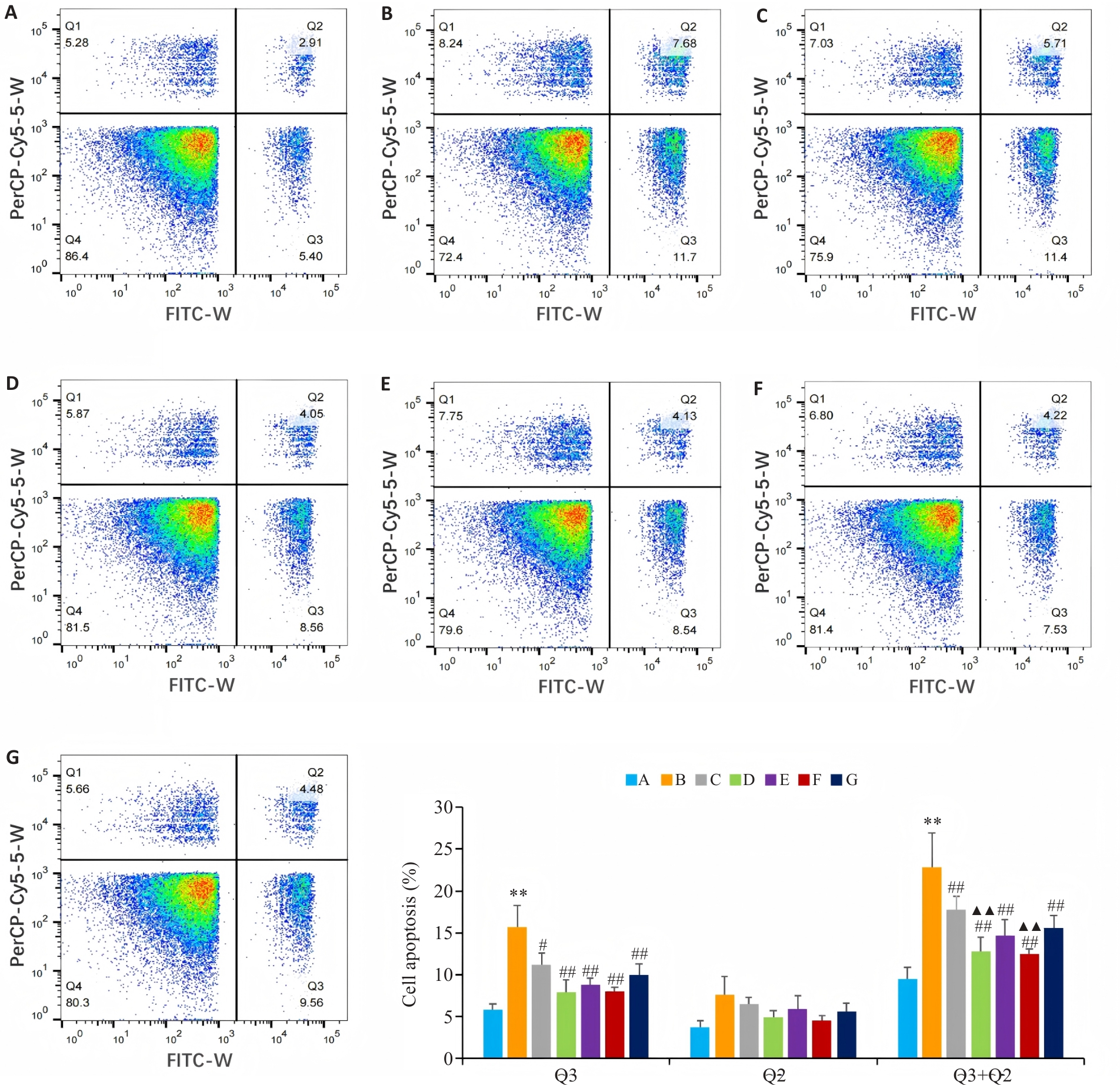
Fig. 6 Flow cytometry of apoptosis rates of BEAS-2B cells in different groups. A: Control group. B: CSE treatment group. C: Low-dose BYF-medicated serum group. D: High-dose BYF-medicated serum group. E: PDTC group. F: High-dose BYF-medicated serum+PDTC group. G: S-CMC group. **P<0.01 vs A; #P<0.05, ##P<0.01 vs B; ▲▲P<0.01 vs C.
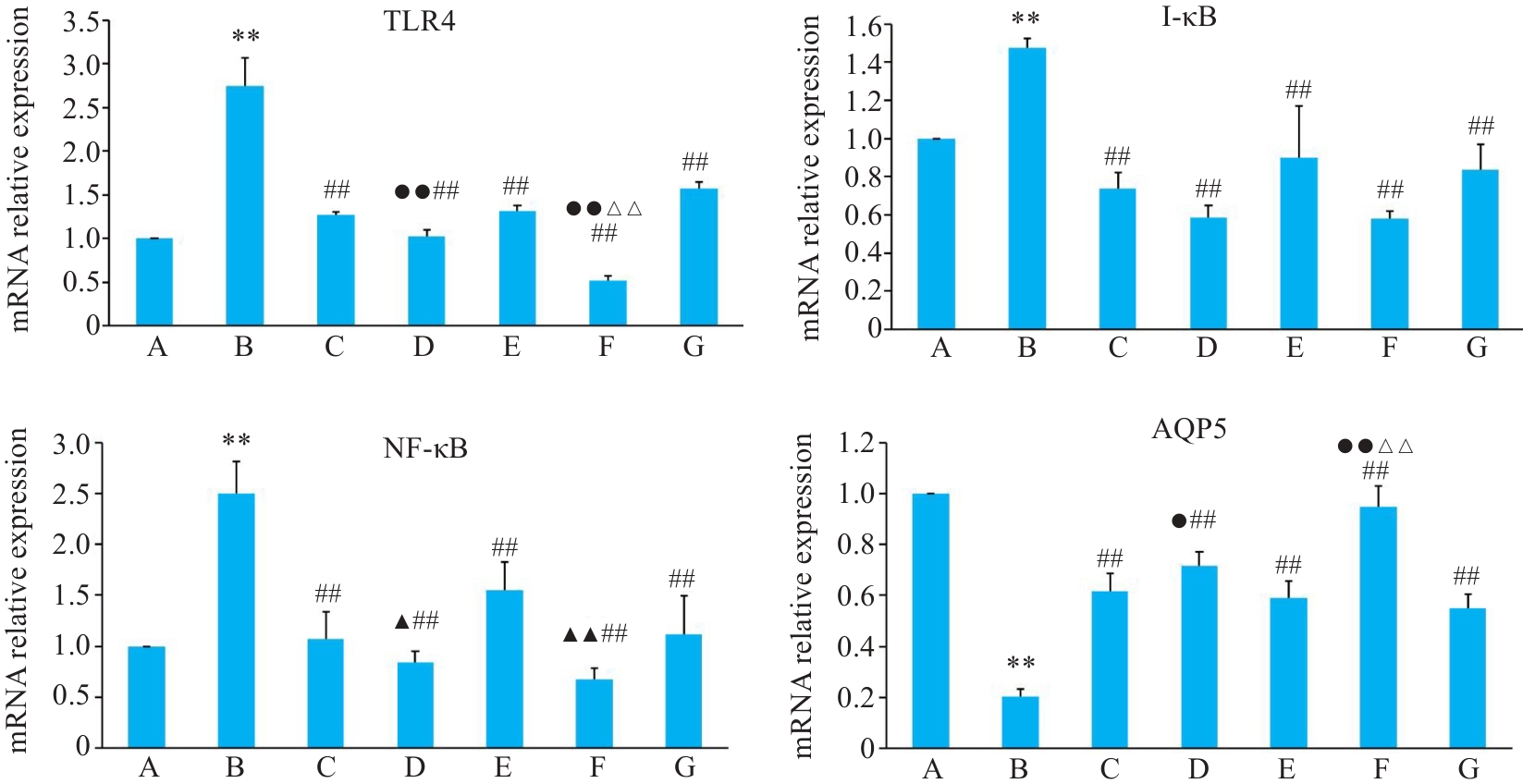
Fig.7 Relative mRNA expressions of TLR4, I-κB, NF-κB and AQP5 in BEAS-2B cells in different groups. A: Control group. B: CSE treatment group. C: Low-dose BYF-medicated serum group. D: High-dose BYF-medicated serum group. E: PDTC group. F: High-dose BYF-medicated serum+PDTC group. G: S-CMC group. **P<0.01 vs A; ##P<0.01 vs B; ▲P<0.05, ▲▲P<0.01 vs C; △△P<0.01 vs E; ●P<0.05, ●●P<0.01 vs G.
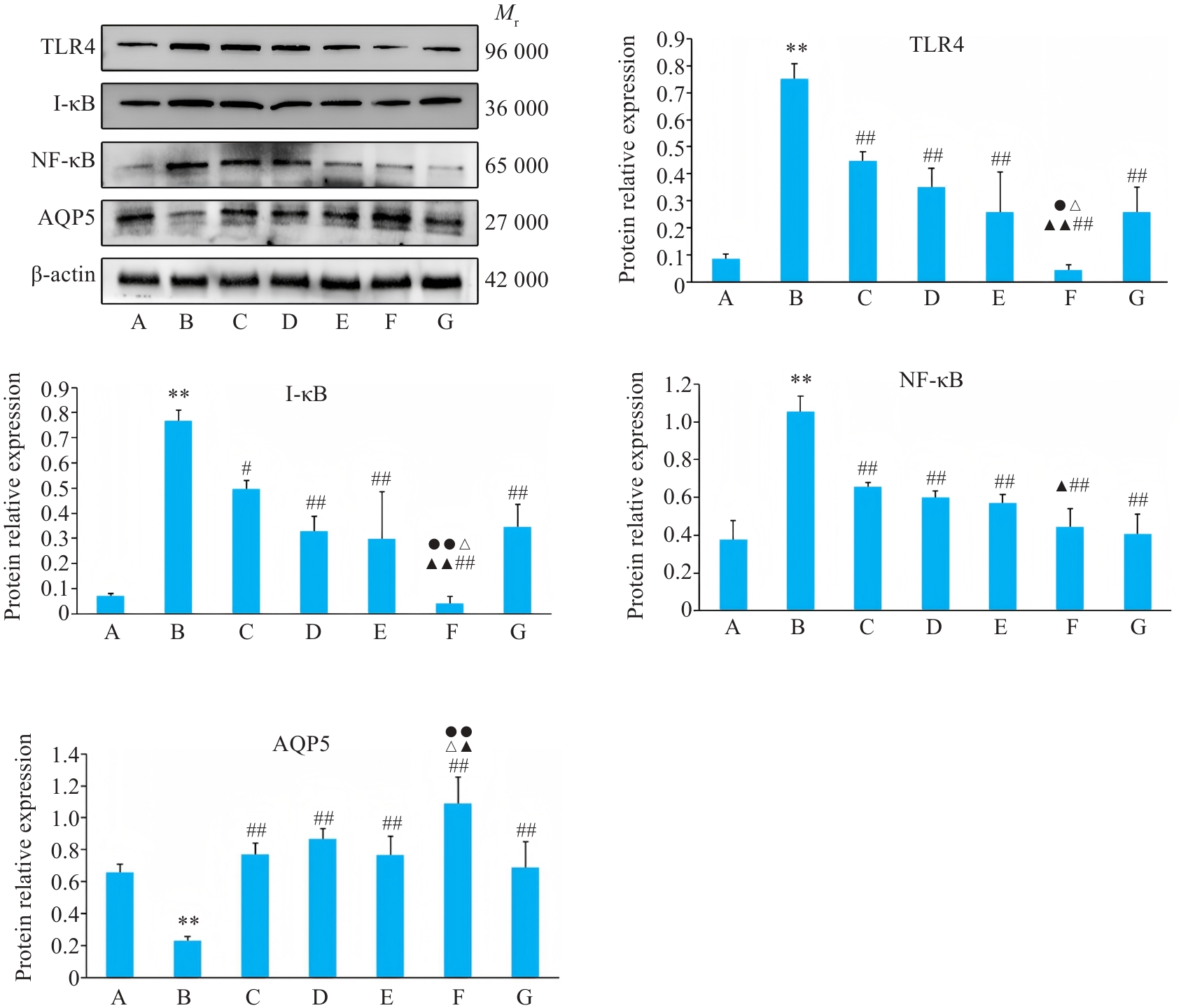
Fig.8 Relative protein expressions of TLR4, I-κB, NF-κB and AQP5 of BEAS-2B cells in different groups. A: Control group. B: CSE treatment group. C: Low-dose BYF-medicated serum group. D: High-dose BYF-medicated serum group. E: PDTC group. F: High-dose BYF-medicated serum+PDTC group. G: S-CMC group. **P<0.01 vs A; #P<0.05, ##P<0.01 vs B; ▲P<0.05, ▲▲P<0.01 vs C; △P<0.05, △△P<0.01 vs E; ●P<0.05, ●●P<0.01 vs G.
| [1] | Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease(2024 report)[EB/OL].[2023-11-15]. . doi:10.1111/j.1440-1843.2005.00692.x |
| [2] | Wang C, Xu JY, Yang L, et al. Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study): a national cross-sectional study[J]. Lancet, 2018, 391(10131): 1706-17. doi:10.1016/s0140-6736(18)30841-9 |
| [3] | Chen SM, Kuhn M, Prettner K, et al. The global economic burden of chronic obstructive pulmonary disease for 204 countries and territories in 2020-50: a health-augmented macroeconomic mo-delling study[J]. Lancet Glob Health, 2023, 11(8): e1183-93. doi:10.1016/s2214-109x(23)00217-6 |
| [4] | Shin DU, Eom JE, Song HJ, et al. Camellia sinensis L. alleviates pulmonary inflammation induced by porcine pancreas elastase and cigarette smoke extract[J]. Antioxidants (Basel), 2022, 11(9): 1683. doi:10.3390/antiox11091683 |
| [5] | 中华医学会, 中华医学会杂志社, 中华医学会全科医学分会, 等. 中国慢性阻塞性肺疾病基层诊疗与管理指南(2024年)[J]. 中华全科医师杂志, 2024, 23(6): 578-602. doi:10.3760/cma.j.cn114798-20240326-00177 |
| [6] | Li SY, Li JS, Wang MH, et al. Effects of comprehensive therapy based on traditional Chinese medicine patterns in stable chronic obstructive pulmonary disease: a four-center, open-label, randomized, controlled study[J]. BMC Complement Altern Med, 2012, 12: 197. doi:10.1186/1472-6882-12-197 |
| [7] | Wang MH, Li JS, Li SY, et al. Effects of comprehensive therapy based on traditional Chinese medicine patterns on older patients with chronic obstructive pulmonary disease: a subgroup analysis from a four-center, randomized, controlled study[J]. Front Med, 2014, 8(3): 368-75. doi:10.1007/s11684-014-0360-0 |
| [8] | Asada M, Yoshida M, Hatachi Y, et al. L-carbocisteine inhibits respiratory syncytial virus infection in human tracheal epithelial cells[J]. Respir Physiol Neurobiol, 2012, 180(1): 112-8. doi:10.1016/j.resp.2011.10.017 |
| [9] | 梅晓峰, 任周新, 余海滨. 补肺益肾方通过调控Notch信号通路改善香烟烟雾提取物诱导气道上皮细胞黏液高分泌[J]. 中华危重病急救医学, 2022, 34(7): 704-9. doi:10.13422/j.cnki.syfjx.20220397 |
| [10] | Jia LD, Liu XF, Liu XG, et al. Bufei Yishen formula protects the airway epithelial barrier and ameliorates COPD by enhancing autophagy through the Sirt1/AMPK/Foxo3 signaling pathway[J]. Chin Med, 2024, 19(1): 32. doi:10.1186/s13020-024-00905-1 |
| [11] | 李建生. 正虚积损为慢性阻塞性肺疾病的主要病机[J]. 中华中医药杂志, 2011, 26(8): 710-3. |
| [12] | Li JS, Xie Y, Zhao P, et al. A Chinese herbal formula ameliorates COPD by inhibiting the inflammatory response via downregulation of p65, JNK, and p38[J]. Phytomedicine, 2021, 83: 153475. doi:10.1016/j.phymed.2021.153475 |
| [13] | Li JS, Ma JD, Tian YG, et al. Effective-component compatibility of Bufei Yishen formula II inhibits mucus hypersecretion of chronic obstructive pulmonary disease rats by regulating EGFR/PI3K/mTOR signaling[J]. J Ethnopharmacol, 2020, 257: 112796. doi:10.1016/j.jep.2020.112796 |
| [14] | 李高峰, 刘淑娟, 李 亚, 等. 调补肺肾三法通过抑制ERK1/2信号通路改善COPD大鼠气道黏液高分泌[J]. 中国实验动物学报, 2024, 32(4): 411-22. |
| [15] | Fang X, Wang ZH, Qi CZ, et al. The changes of MRP2 expression in three kinds of pulmonary inflammation models: the downregulation occurred in cigarette smoke extract (CSE) stimulation group and CSE plus LPS stimulation group, unchanged in LPS stimulation group[J]. Toxicol Mech Methods, 2021, 31(6): 413-24. doi:10.1080/15376516.2021.1903638 |
| [16] | Yamaguchi NH. Smoking, immunity, and DNA damage[J]. Transl Lung Cancer Res, 2019, 8(): S3-6. doi:10.21037/tlcr.2019.03.02 |
| [17] | Heinzelmann K, Fysikopoulos A, Jaquin TJ, et al. Pulmonary-delivered Anticalin Jagged-1 antagonists reduce experimental airway mucus hyperproduction and obstruction[J]. Am J Physiol Lung Cell Mol Physiol, 2025, 328(1): L75-92. doi:10.1152/ajplung.00059.2024 |
| [18] | Carpenter J, Wang Y, Gupta R, et al. Assembly and organization of the N-terminal region of mucin MUC5AC: indications for structural and functional distinction from MUC5B[J]. Proc Natl Acad Sci USA, 2021, 118(39): e2104490118. doi:10.1073/pnas.2104490118 |
| [19] | Yadav E, Yadav N, Hus A, et al. Aquaporins in lung health and disease: Emerging roles, regulation, and clinical implications[J]. Respir Med, 2020, 174: 106193. doi:10.1016/j.rmed.2020.106193 |
| [20] | Hill DB, Button B, Rubinstein M, et al. Physiology and pathophysiology of human airway mucus[J]. Physiol Rev, 2022, 102(4): 1757-836. doi:10.1152/physrev.00004.2021 |
| [21] | Fokkens WJ, Backer V, Lund VJ, et al. Pocket guide: biologics in upper and lower airways in adults[J]. Rhinology, 2025, 63(2): 242-4. |
| [22] | Bae CH, Na HG, Choi YS, et al. Clusterin induces MUC5AC expression via activation of NF-κB in human airway epithelial cells[J]. Clin Exp Otorhinolaryngol, 2018, 11(2): 124-32. doi:10.21053/ceo.2017.00493 |
| [23] | Fu HT, Zhang Y, Zhang P, et al. Tumor necrosis factor-α promotes airway mucus hypersecretion by repressing miR-146a-5p and miR-134-5p levels in human airway epithelial cells[J]. Transl Cancer Res, 2021, 10(9): 4047-56. doi:10.21037/tcr-20-3375 |
| [24] | Liu HL, Zhou L, Wang XF, et al. Dexamethasone upregulates macro-phage PIEZO1 via SGK1, suppressing inflammation and increasing ROS and apoptosis[J]. Biochem Pharmacol, 2024, 222: 116050. doi:10.1016/j.bcp.2024.116050 |
| [25] | Zhang MY, Lu Y, Liu LL, et al. Role and mechanism of miR-181a-5p in mice with chronic obstructive pulmonary disease by regulating HMGB1 and the NF‑κB pathway[J]. Cells Tissues Organs, 2023, 212(3): 245-57. doi:10.1159/000522155 |
| [1] | QIN Na, HUANG Lin, DONG Rui, LI Fen, TANG Xuheng, ZENG Zhenhua, WANG Xingmin, YANG Hong. Polydatin improves intestinal barrier injury after traumatic brain injury in rats by reducing oxidative stress and inflammatory response via activating SIRT1-mediated deacetylation of SOD2 and HMGB1 [J]. Journal of Southern Medical University, 2022, 42(1): 93-100. |
| [2] | . Curcumin suppresses cigarette smoke extract-induced oxidative stress through PPARγ/ NF-κB pathway in human bronchial epithelial cells in vitro [J]. Journal of Southern Medical University, 2018, 38(10): 1209-. |
| [3] |
.
Role of phospholipid transfer protein in cigarette smoke-induced apoptosis of RLE-6TN cells [J]. Journal of Southern Medical University, 2015, 35(07): 941-. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||