南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 541-549.doi: 10.12122/j.issn.1673-4254.2026.03.08
• 临床研究 • 上一篇
收稿日期:2025-09-26
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
龚英,李海侠
E-mail:x011218jie@163.com;gongy3@mail2.sysu.edu.cn;yingchun1220@163.com
作者简介:肖 洁,在读硕士研究生,E-mail: x011218jie@163.com
基金资助:
Jie XIAO( ), Lingyue JIN, Lina DUAN, Ying GONG(
), Lingyue JIN, Lina DUAN, Ying GONG( ), Haixia LI(
), Haixia LI( )
)
Received:2025-09-26
Online:2026-03-20
Published:2026-03-26
Contact:
Ying GONG, Haixia LI
E-mail:x011218jie@163.com;gongy3@mail2.sysu.edu.cn;yingchun1220@163.com
Supported by:摘要:
目的 探讨唾液链球菌来源胞外囊泡(SS-EVs)对自然杀伤(NK)细胞活化功能的调控作用及其在系统性红斑狼疮(SLE)疾病进展中的潜在机制。 方法 收集35例SLE患者及38例健康对照者的临床样本,检测其外周血T细胞、B细胞及NK细胞的数量与比例。提取患者粪便DNA,通过PCR扩增出唾液链球菌DNA片段,并通过琼脂糖凝胶电泳评估其丰度水平。采用超速离心技术分离SS-EVs,并通过透射电镜、纳米颗粒追踪分析和Western blotting对其进行表征。将SS-EVs与NK细胞共培养,通过定量聚合酶链式反应(qPCR)及流式细胞术体外检测NK细胞的活化状态及其细胞毒功能;将24只MRL/lpr小鼠随机分成3组,8只/组,每次分别灌胃100 μL PBS、100 μL含20 μg的E.coli-EVs和SS-EVs,通过HE染色、免疫荧光及ELISA评估肾脏病理改变、免疫复合物沉积及外周炎症因子水平,从而评估SS-EVs对狼疮性肾炎的影响。 结果 SLE患者外周血中NK细胞数量与比例下降(P<0.0001),粪便中SS丰度升高,且与SLEDAI评分呈正相关(R2=0.8369)。体外研究显示,SS-EVs上调NK细胞表面激活性受体的表达(P<0.01),增强其对靶细胞的杀伤效能;体内实验发现,SS-EVs可加剧小鼠肾组织炎症反应,促进C3和IgG免疫复合物沉积,升高血清中IL-6、TNF-α、IL-8及CCL20等炎症因子水平(P<0.05)。 结论 SLE患者的肠道中唾液链球菌富集,并通过释放SS-EVs促进NK细胞毒性功能的增强,诱导肾脏炎症反应及免疫复合物沉积,从而加重SLE的疾病进展。SS及其胞外囊泡可能为SLE的防治提供全新的靶向策略。
肖洁, 金凌岳, 段丽娜, 龚英, 李海侠. 唾液链球菌胞外囊泡通过活化自然杀伤细胞加剧系统性红斑狼疮肾炎病理进展[J]. 南方医科大学学报, 2026, 46(3): 541-549.
Jie XIAO, Lingyue JIN, Lina DUAN, Ying GONG, Haixia LI. Streptococcus salivarius-derived extracellular vesicles exacerbate progression of lupus nephritis by activating natural killer cells[J]. Journal of Southern Medical University, 2026, 46(3): 541-549.
| Primer name | Sequence (5'-3') |
|---|---|
| GAPDH -FOR | CTGTTCGACAGTCAGCCGCATC |
| GAPDH -REV | GCGCCCAATACGACCAAATCCG |
| TNF-FOR | GAGGCCAAGCCCTGGTATG |
| TNF-REV | CGGGCCGATTGATCTCAGC |
| GZMB-FOR | CCCTGGGAAAACACTCACACA |
| GZMB-REV | GCACAACTCAATGGTACTGTCG |
| IL2-FOR | AACTCCTGTCTTGCATTGCAC |
| IL2-REV | GCTCCAGTTGTAGCTGTGTTT |
| Perforin-FOR | GACTGCCTGACTGTCGAGG |
| Perforin-REV | TCCCGGTAGGTTTGGTGGAA |
| NKG2D-FOR | CCTTGACCGAAAGTTACTGTGG |
| NKG2D-REV | GGCTGGCATTTTGAGACATACAA |
| NKG2A-FOR | AGCTCCATTTTAGCAACTGAACA |
| NKG2A-REV | CAACTATCGTTACCACAGAGGC |
表1 引物序列
Tab.1 Primer sequences for qPCR
| Primer name | Sequence (5'-3') |
|---|---|
| GAPDH -FOR | CTGTTCGACAGTCAGCCGCATC |
| GAPDH -REV | GCGCCCAATACGACCAAATCCG |
| TNF-FOR | GAGGCCAAGCCCTGGTATG |
| TNF-REV | CGGGCCGATTGATCTCAGC |
| GZMB-FOR | CCCTGGGAAAACACTCACACA |
| GZMB-REV | GCACAACTCAATGGTACTGTCG |
| IL2-FOR | AACTCCTGTCTTGCATTGCAC |
| IL2-REV | GCTCCAGTTGTAGCTGTGTTT |
| Perforin-FOR | GACTGCCTGACTGTCGAGG |
| Perforin-REV | TCCCGGTAGGTTTGGTGGAA |
| NKG2D-FOR | CCTTGACCGAAAGTTACTGTGG |
| NKG2D-REV | GGCTGGCATTTTGAGACATACAA |
| NKG2A-FOR | AGCTCCATTTTAGCAACTGAACA |
| NKG2A-REV | CAACTATCGTTACCACAGAGGC |
| General information | SLE group (n=35) | HC group (n=38) | P | |
|---|---|---|---|---|
| Gender [n (%)] | Female | 30 (85.71%) | 31 (81.58%) | 0.756 |
| Male | 5 (14.29%) | 7 (18.42%) | ||
| Age (year) | 36.57±12.50 | 39.34±11.84 | 0.334 | |
| WBC (109/L) | 6.07±2.81 | 5.66±1.17 | 0.422 | |
| LYM (109/L) | 1.53±0.76 | 2.20±0.47 | <0.001 | |
| NEU (109/L) | 4.12±2.09 | 3.12±0.93 | 0.043 | |
| RBC (1012/L) | 4.12±0.67 | 4.89±0.32 | <0.001 | |
| HGB (g/L) | 113.48±14.73 | 135.67±17.63 | <0.001 | |
| HCT (L/L) | 0.37±0.38 | 0.42±0.43 | <0.001 | |
| PLT (109/L) | 243.42±82.22 | 255.64±59.45 | 0.512 | |
| SLEDAI | 6.74±3.35 | - | - | |
| WBC: White blood cell; LYM: Lymphocyte; NEU: Neutrophil; RBC: Red blood cell; HGB: Hemoglobin; HCT: Hematocrit; PLT: Platelet. Data are presented as Mean±SD. P values were calculated by Mann-Whitney U test or t test. | ||||
表2 SLE组与HC组的临床特征
Tab.2 Clinical characteristics of the SLE patients and healthy control subjects (Mean±SD)
| General information | SLE group (n=35) | HC group (n=38) | P | |
|---|---|---|---|---|
| Gender [n (%)] | Female | 30 (85.71%) | 31 (81.58%) | 0.756 |
| Male | 5 (14.29%) | 7 (18.42%) | ||
| Age (year) | 36.57±12.50 | 39.34±11.84 | 0.334 | |
| WBC (109/L) | 6.07±2.81 | 5.66±1.17 | 0.422 | |
| LYM (109/L) | 1.53±0.76 | 2.20±0.47 | <0.001 | |
| NEU (109/L) | 4.12±2.09 | 3.12±0.93 | 0.043 | |
| RBC (1012/L) | 4.12±0.67 | 4.89±0.32 | <0.001 | |
| HGB (g/L) | 113.48±14.73 | 135.67±17.63 | <0.001 | |
| HCT (L/L) | 0.37±0.38 | 0.42±0.43 | <0.001 | |
| PLT (109/L) | 243.42±82.22 | 255.64±59.45 | 0.512 | |
| SLEDAI | 6.74±3.35 | - | - | |
| WBC: White blood cell; LYM: Lymphocyte; NEU: Neutrophil; RBC: Red blood cell; HGB: Hemoglobin; HCT: Hematocrit; PLT: Platelet. Data are presented as Mean±SD. P values were calculated by Mann-Whitney U test or t test. | ||||
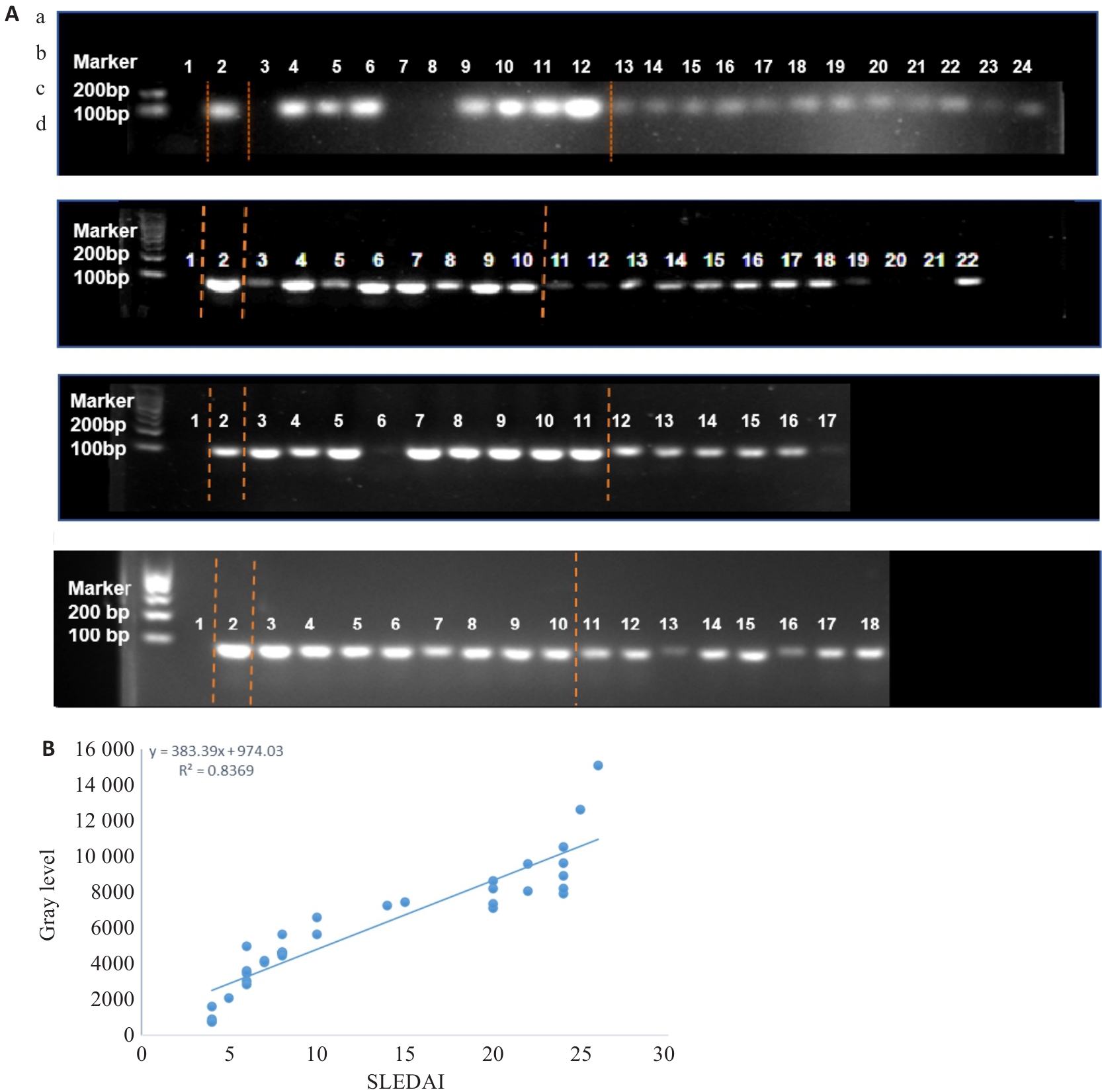
图1 粪便SS核酸琼脂糖凝胶电泳结果
Fig.1 Agarose gel electrophoresis of fecal Streptococcus salivarius (SS) nucleic acids. A: Results of agarose gel electrophoresis. Lane 1: Deionized water (negative control); Lane 2: SS standard strain (positive control). a: Lanes 3-12 are SLE group, and lanes 13-24 are HC group; b: Lanes 3-10 are SLE group, and lanes 11-22 are HC group; c: Lanes 3-11 are SLE group, and lanes 12-17 are HC group; d: Lanes 3-10 are SLE group, and lanes 11-18 are HC group. B: Correlation analysis between the grayscale value of fecal SS nucleic acids from SLE patients and the SLEDAI score.
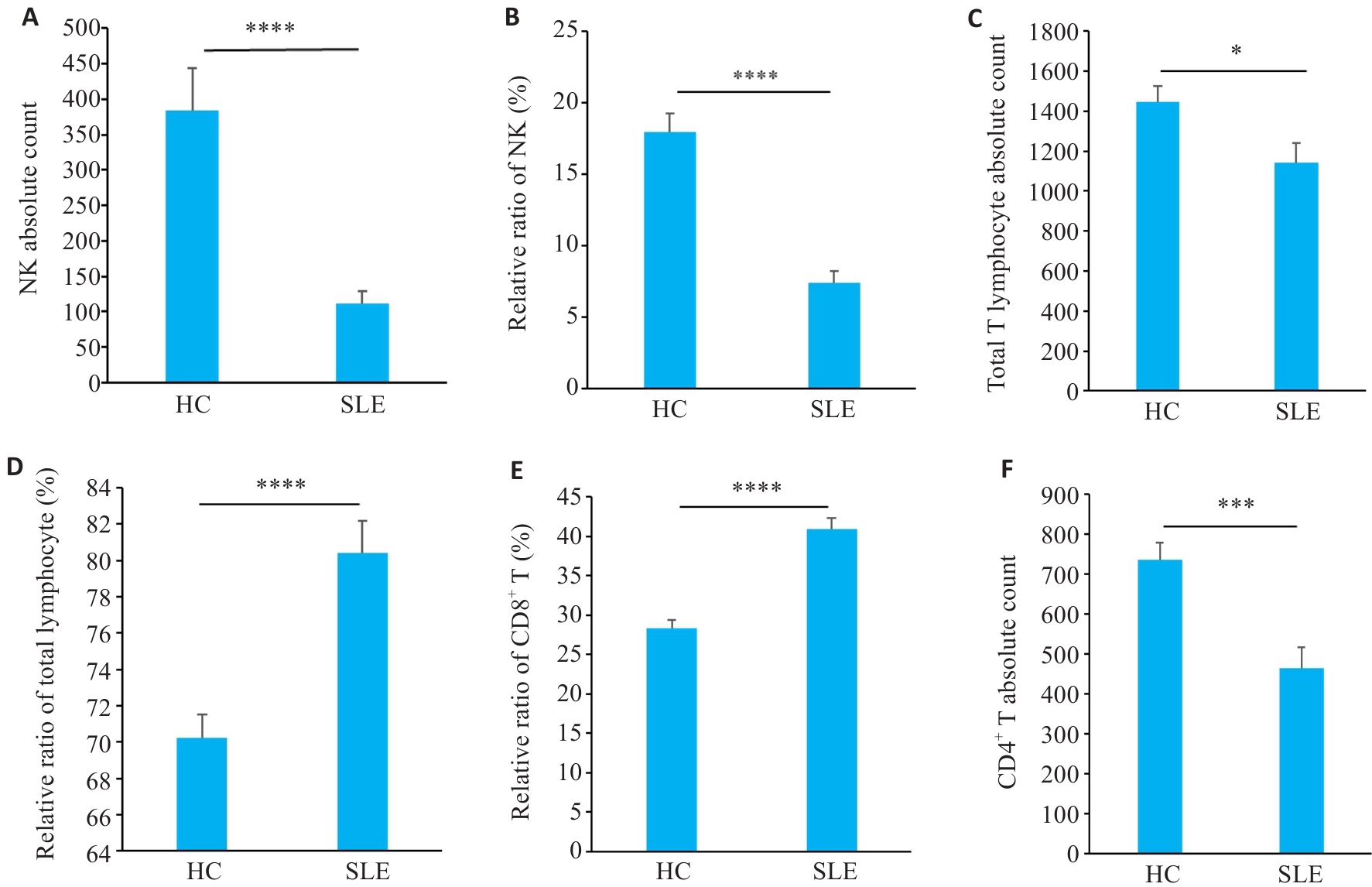
图2 SLE患者外周血免疫细胞亚群分布情况
Fig.2 Distribution of peripheral blood immune cell subsets in SLE patients. A, B: NK cell proportions and absolute counts in SLE patients and HC groups. C, D: Total T cell absolute counts and subset proportions. E: CD8+ T cell subset proportions. F: CD4+ T cell absolute counts (n=35 in SLE group, n=38 in HC group). *P<0.05, ***P<0.001, ****P<0.0001.
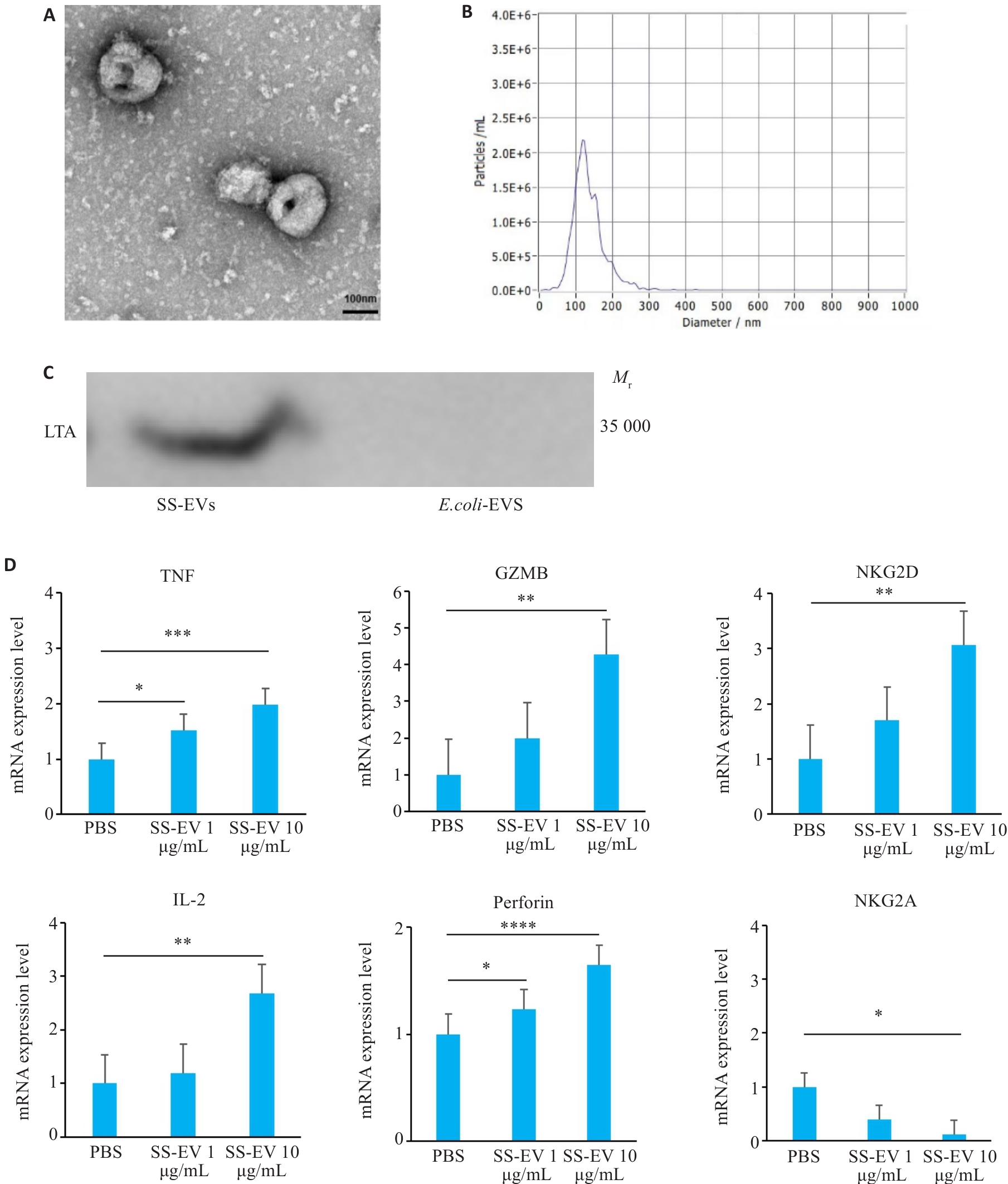
图3 SS-EVs的物理特性及NK细胞受体调控作用
Fig.3 Physical properties of Streptococcus salivarius-derived extracellular vesicles (SS-EVs)and their regulatory effect on NK cell receptors. A: Transmission electron microscopy (TEM) of SS-EVs (Scale bar=100 nm). B: NTA analysis showing size distribution of SS-EVs (100-200 nm). C: Western blotting of LTA protein expression in SS-EVs. D: qPCR analysis of mRNA expression changes of the activating receptors (TNF, GZMB, NKG2D, IL-2, and Perforin) and inhibitory receptors (NKG2A) in NK cells co-cultured with SS-EVs (n=3). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
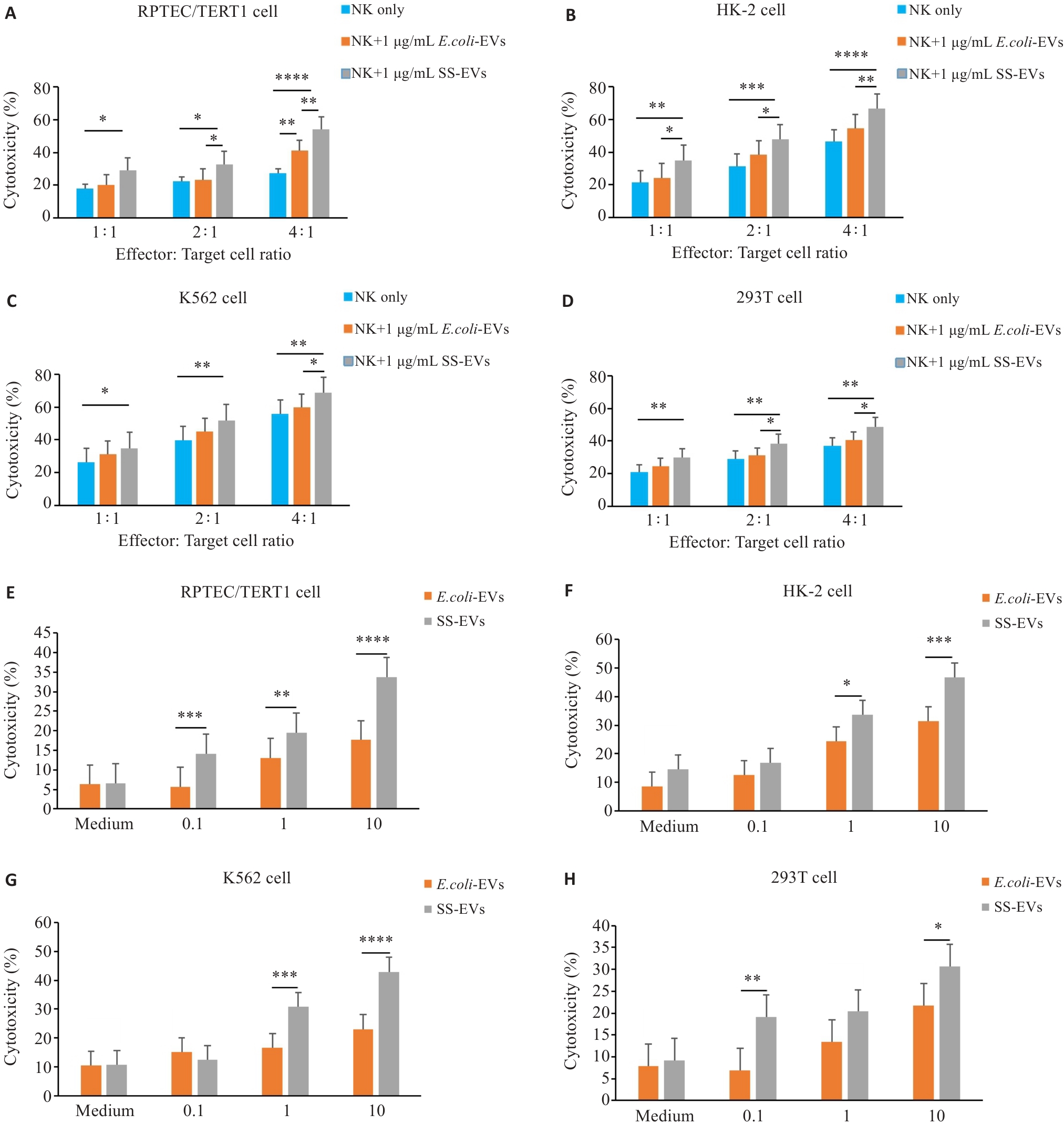
图4 SS-EVs增强NK细胞对靶细胞的毒性作用
Fig.4 SS-EVs enhance cytotoxic effect of NK cells on target cells. A-D: Relationship between NK cell to target cell ratio (1:1 to 4:1) and NK cell cytotoxicity at a fixed SS-EV concentration (1 μg/mL) (n=3). E-H: Flow cytometry analysis of the effects of varying SS-EV concentrations (0.1-10 μg/mL) on NK cell cytotoxicity against 4 target cells (n=3). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
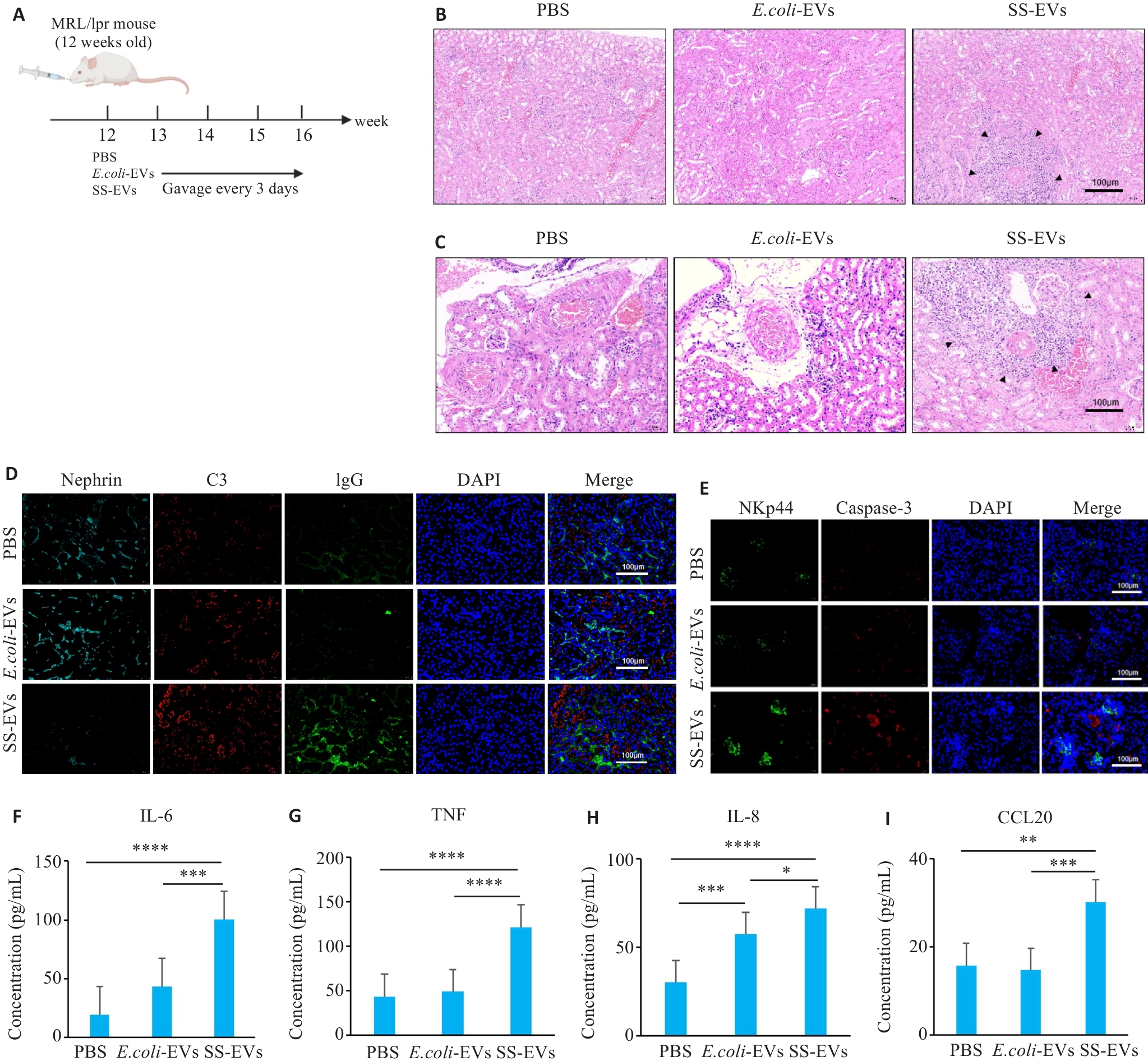
图5 SS-EVs灌胃加重MRL/lpr小鼠肾脏病理损伤
Fig.5 SS-EVs gavage aggravates kidney pathological damage in MRL/lpr mice. A: Schematic diagram of animal grouping and gavage. B: Significant inflammatory cell infiltration in the kidneys of mice gavaged with SS-EVs. C: Significant perivascular inflammatory cell infiltration in the kidneys of mice gavaged with SS-EVs. D: SS-EVs damage the renal medulla and promote immune complex deposition. E:SS-EVs induce NK cell infiltration in the kidneys and renal apoptosis. F-I: ELISA for IL-6, TNF, IL-8, and CCL20 levels in mouse serum (n=6). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
| [1] | Siegel CH, Sammaritano LR. Systemic lupus erythematosus: a review[J]. JAMA, 2024, 331(17): 1480-91. doi:10.1001/jama.2024.2315 |
| [2] | Cojocaru M, Cojocaru IM, Silosi I, et al. Manifestations of systemic lupus erythematosus[J]. Maedica (Bucur), 2011, 6(4): 330-6. |
| [3] | Weckerle CE, Niewold TB. The unexplained female predominance of systemic lupus erythematosus: clues from genetic and cytokine studies[J]. Clin Rev Allergy Immunol, 2011, 40(1): 42-9. doi:10.1007/s12016-009-8192-4 |
| [4] | McMurray RW, May W. Sex hormones and systemic lupus erythematosus: review and meta-analysis[J]. Arthritis Rheum, 2003, 48(8): 2100-10. doi:10.1002/art.11105 |
| [5] | Rosario C, Seguro L, Vasconcelos C, et al. Is there a cure for systemic lupus erythematosus[J]? Lupus, 2013, 22(5): 417-21. doi:10.1177/0961203313479839 |
| [6] | Yu HT, Nagafuchi Y, Fujio K. Clinical and immunological biomarkers for systemic lupus erythematosus[J]. Biomolecules, 2021, 11(7): 928. doi:10.3390/biom11070928 |
| [7] | Suárez-Fueyo A, Bradley SJ, Tsokos GC. T cells in systemic lupus erythematosus[J]. Curr Opin Immunol, 2016, 43: 32-8. doi:10.1016/j.coi.2016.09.001 |
| [8] | Karrar S, Cunninghame Graham DS. Abnormal B cell development in systemic lupus erythematosus: what the genetics tell us[J]. Arthritis Rheumatol, 2018, 70(4): 496-507. doi:10.1002/art.40396 |
| [9] | Perera Arachchige AS. Human NK cells: From development to effector functions[J]. Innate Immun, 2021, 27(3): 212-29. doi:10.1177/17534259211001512 |
| [10] | Mace EM. Human natural killer cells: Form, function, and development[J]. J Allergy Clin Immunol, 2023, 151(2): 371-85. doi:10.1016/j.jaci.2022.09.022 |
| [11] | Jamal E, Azmy E, Ayed M, et al. Clinical impact of percentage of natural killer cells and natural killer-like T cell population in acute myeloid leukemia[J]. J Hematol, 2020, 9(3): 62-70. doi:10.14740/jh655 |
| [12] | Ramírez-Labrada A, Pesini C, Santiago L, et al. All about (NK cell-mediated) death in two acts and an unexpected encore: initiation, execution and activation of adaptive immunity[J]. Front Immunol, 2022, 13: 896228. doi:10.3389/fimmu.2022.896228 |
| [13] | Paul S, Lal G. The molecular mechanism of natural killer cells function and its importance in cancer immunotherapy[J]. Front Immunol, 2017, 8: 1124. doi:10.3389/fimmu.2017.01124 |
| [14] | Wang W, Erbe AK, Hank JA, et al. NK cell-mediated antibody-dependent cellular cytotoxicity in cancer immunotherapy[J]. Front Immunol, 2015, 6: 368. doi:10.3389/fimmu.2015.00368 |
| [15] | Hill GR, Betts BC, Tkachev V, et al. Current concepts and advances in graft-versus-host disease immunology[J]. Annu Rev Immunol, 2021, 39: 19-49. doi:10.1146/annurev-immunol-102119-073227 |
| [16] | Xiao J, Luo YH, Duan LN, et al. Exploring differential gene expression and biomarker potential in systemic lupus erythematosus: a retrospective study[J]. PeerJ, 2025, 13: e19891. doi:10.7717/peerj.19891 |
| [17] | Duarte-García A, Hocaoglu M, Valenzuela-Almada M, et al. Rising incidence and prevalence of systemic lupus erythematosus: a population-based study over four decades[J]. Ann Rheum Dis, 2022, 81(9): 1260-6. doi:10.1136/annrheumdis-2022-222276 |
| [18] | Fu YF, Lyu J, Wang SS. The role of intestinal microbes on intestinal barrier function and host immunity from a metabolite perspective[J]. Front Immunol, 2023, 14: 1277102. doi:10.3389/fimmu.2023.1277102 |
| [19] | Bechler KK, Stolyar L, Steinberg E, et al. Predicting patients who are likely to develop Lupus Nephritis of those newly diagnosed with Systemic Lupus Erythematosus[J]. AMIA Annu Symp Proc, 2023, 2022: 221-30. |
| [20] | Singh BK, Singh S. Systemic lupus erythematosus and infections[J]. Reumatismo, 2020, 72(3): 154-69. doi:10.4081/reumatismo.2020.1303 |
| [21] | Jin LY, Xiao J, Luo YH, et al. Exploring gut microbiota in systemic lupus erythematosus: insights and biomarker discovery potential[J]. Clin Rev Allergy Immunol, 2025, 68(1): 42. doi:10.1007/s12016-025-09051-4 |
| [22] | Yan R, Jiang HY, Gu SL, et al. Fecal metabolites were altered, identified as biomarkers and correlated with disease activity in patients with systemic lupus erythematosus in a GC-MS-based metabolomics study[J]. Front Immunol, 2020, 11: 2138. doi:10.3389/fimmu.2020.02138 |
| [23] | Kaci G, Goudercourt D, Dennin V, et al. Anti-inflammatory properties of Streptococcus salivarius, a commensal bacterium of the oral cavity and digestive tract[J]. Appl Environ Microbiol, 2014, 80(3): 928-34. doi:10.1128/aem.03133-13 |
| [24] | Wescombe PA, Hale JDF, Heng NCK, et al. Developing oral probiotics from Streptococcus salivarius [J]. Future Microbiol, 2012, 7(12): 1355-71. doi:10.2217/fmb.12.113 |
| [25] | García-Ríos P, Pecci-Lloret MP, Oñate-Sánchez RE. Oral manifestations of systemic lupus erythematosus: a systematic review[J]. Int J Environ Res Public Health, 2022, 19(19): 11910. doi:10.3390/ijerph191911910 |
| [26] | Duan LN, Lin WY, Zhang Y, et al. Exosomes in autoimmune diseases: a review of mechanisms and diagnostic applications[J]. Clin Rev Allergy Immunol, 2025, 68(1): 5. doi:10.1007/s12016-024-09013-2 |
| [27] | Muñoz-Echeverri LM, Benavides-López S, Geiger O, et al. Bacterial extracellular vesicles: biotechnological perspective for enhanced productivity[J]. World J Microbiol Biotechnol, 2024, 40(6): 174. doi:10.1007/s11274-024-03963-7 |
| [28] | Ou ZH, Bo ST, Huang XY, et al. Single-particle analysis of circulating bacterial extracellular vesicles reveals their biogenesis, changes in blood and links to intestinal barrier[J]. J Extracell Vesicles, 2023, 12(12): e12395. doi:10.1002/jev2.12395 |
| [29] | Nie XK, Li QQ, Chen XY, et al. Bacterial extracellular vesicles: Vital contributors to physiology from bacteria to host[J]. Microbiol Res, 2024, 284: 127733. doi:10.1016/j.micres.2024.127733 |
| [30] | Gao Y, Zhang YK, Liu XG. Rheumatoid arthritis: pathogenesis and therapeutic advances[J]. MedComm (2020), 2024, 5(3): e509. doi:10.1002/mco2.509 |
| [31] | Mueller AL, Payandeh Z, Mohammadkhani N, et al. Recent advances in understanding the pathogenesis of rheumatoid arthritis: new treatment strategies[J]. Cells, 2021, 10(11): 3017. doi:10.3390/cells10113017 |
| [32] | Hong MK, Li Z, Liu HH, et al. Fusobacterium nucleatum aggravates rheumatoid arthritis through FadA-containing outer membrane vesicles[J]. Cell Host Microbe, 2023, 31(5): 798-810.e7. doi:10.1016/j.chom.2023.03.018 |
| [33] | Salgueiro VC, Passemar C, Vázquez-Iniesta L, et al. Extracellular vesicles in mycobacteria: new findings in biogenesis, host-pathogen interactions, and diagnostics[J]. mBio, 2024, 15(5): e0255223. doi:10.1128/mbio.02552-23 |
| [34] | Kucuksezer UC, Aktas Cetin E, Esen F, et al. The role of natural killer cells in autoimmune diseases[J]. Front Immunol, 2021, 12: 622306. doi:10.3389/fimmu.2021.622306 |
| [35] | Novelli L, Barbati C, Capuano C, et al. KLRG1 is reduced on NK cells in SLE patients, inversely correlates with disease activity and is modulated by hydroxychloroquine in vitro [J]. Lupus, 2023, 32(4): 549-59. doi:10.1177/09612033231160979 |
| [36] | Seow WK, Lam JHC, Tsang AKL, et al. Oral Streptococcus species in pre-term and full-term children-a longitudinal study[J]. Int J Paediatr Dent, 2009, 19(6): 406-11. doi:10.1111/j.1365-263x.2009.01003.x |
| [37] | Iraci N, Leonardi T, Gessler F, et al. Focus on extracellular vesicles: physiological role and signalling properties of extracellular membrane vesicles[J]. Int J Mol Sci, 2016, 17(2): 171. doi:10.3390/ijms17020171 |
| [38] | Rafieezadeh D, Rafieezadeh A. Extracellular vesicles and their therapeutic applications: a review article (part1)[J]. Int J Physiol Pathophysiol Pharmacol, 2024, 16(1): 1-9. doi:10.62347/qpag5693 |
| [1] | 杜庆锋, 杨超, 夏雪晴, 王婷. 细胞外囊泡在神经系统疾病中的治疗潜力[J]. 南方医科大学学报, 2025, 45(9): 2046-2054. |
| [2] | 汤忠富, 黄传兵, 李明, 程丽丽, 陈君洁, 尚双双, 刘思娣. 芪黄健脾滋肾颗粒通过抑制MyD88/NF-κB通路减轻MRL/lpr小鼠肾损害[J]. 南方医科大学学报, 2025, 45(8): 1625-1632. |
| [3] | 程丽丽, 汤忠富, 李明, 陈君洁, 尚双双, 刘思娣, 黄传兵. 芪黄健脾滋肾颗粒通过AIM2/Blimp-1/Bcl-6轴抑制B细胞分化改善MRL/lpr小鼠肾损害[J]. 南方医科大学学报, 2025, 45(11): 2297-2308. |
| [4] | 多文娟, 王怡祥, 王家兴, 许鑫龙, 李林献, 杨栋臣, 申启利, 杨立春, 刘晓静, 金启旺, 褚亮, 杨小迪. 重组日本血吸虫半胱氨酸蛋白酶抑制剂可通过调节炎症微环境对“二次打击”脓毒症小鼠发挥治疗作用[J]. 南方医科大学学报, 2025, 45(1): 110-117. |
| [5] | 陈君洁, 黄传兵, 李 明. 健脾滋肾方抑制系统性红斑狼疮患者的足细胞自噬:基于网络药理学和临床研究[J]. 南方医科大学学报, 2024, 44(3): 465-473. |
| [6] | 范毅平, 罗梦琳, 黄东宗, 刘 琳, 傅 博, 王潇宇, 关淼升, 李鸿波. 缓释地塞米松改性丝胶蛋白水凝胶支架促进大鼠下颌骨缺损修复:基于调节巨噬细胞M2极化[J]. 南方医科大学学报, 2024, 44(3): 533-540. |
| [7] | 苏晓玲, 廖道庸, 李超, 陈丽, 王婧芸, 甘甜, 罗浩荡, 伍宁, 何军. 唾液链球菌K12对肺炎支原体感染小鼠的预防作用[J]. 南方医科大学学报, 2024, 44(12): 2300-2307. |
| [8] | 李云飞, 庞利君, 束龙武, 李明, 黄传兵. 芪黄健脾滋肾颗粒可改善小鼠系统性红斑狼疮血小板减少:基于Ca2+/CaMKK2/AMPK/mTOR信号通路介导的自噬[J]. 南方医科大学学报, 2024, 44(12): 2327-2334. |
| [9] | 关深元, 沈智勇, 林名岛, 邓海军, 方 媛. 泛癌组织STIP1的表达与肿瘤免疫浸润及预后相关:基于生物信息学方法[J]. 南方医科大学学报, 2023, 43(7): 1179-1193. |
| [10] | 管鹏飞, 崔瑞文, 王其友, 孙永建. 负载骨髓干细胞来源外泌体的3D水凝胶通过调节免疫促进损伤软骨的修复[J]. 南方医科大学学报, 2022, 42(4): 528-537. |
| [11] | 王德奖, 李 挺, 徐颖怡, 杨雪雯, 何铭垣, 张智勇, 吴 炜, 燕 翼. 富血小板血浆可减轻大鼠急性心肌缺血-再灌注损伤[J]. 南方医科大学学报, 2021, 41(5): 775-782. |
| [12] | 李 静, 郑 晶, 王敏达, 张 艳, 江益凡, 张小凤, 郭 普. 抑制CD96可促进NK细胞分泌IFN-γ并减轻小鼠肺部的衣原体感染[J]. 南方医科大学学报, 2020, 40(07): 930-935. |
| [13] | 王凯喆,魏余辉,张 萍,王建华,胡 钧,王丽华,李 宾. 细胞外囊泡成像方法最新研究进展[J]. 南方医科大学学报, 2020, 40(02): 279-286. |
| [14] | 黄雨茜,张 浩,张 双,孙 剑. 维生素D受体与MCP-1在系统性红斑狼疮患者中的表达及意义[J]. 南方医科大学学报, 2020, 40(01): 99-103. |
| [15] | 吴凡,陈韵颖,肖花,邹子良,宁靖,陈海珊,邹和群. 尿液细胞外囊泡中的podocalyxin在糖尿病肾病诊断中的价值[J]. 南方医科大学学报, 2018, 38(09): 1126-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||