南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (6): 1270-1279.doi: 10.12122/j.issn.1673-4254.2025.06.16
收稿日期:2024-11-27
出版日期:2025-06-20
发布日期:2025-06-27
通讯作者:
吴芃
E-mail:zhuyuexuan0630@ outlook.com;doctorwupeng @gmail.com
作者简介:祝越轩,在读硕士研究生,E-mail:zhuyuexuan0630@ outlook.com
基金资助:
Yuexuan ZHU( ), Zhangrui ZHU, Peng WU(
), Zhangrui ZHU, Peng WU( )
)
Received:2024-11-27
Online:2025-06-20
Published:2025-06-27
Contact:
Peng WU
E-mail:zhuyuexuan0630@ outlook.com;doctorwupeng @gmail.com
Supported by:摘要:
目的 探究多硫酸戊聚糖(PPS)通过调节肠道微生物群和胆汁酸代谢来缓解环磷酰胺(CYP)诱导的间质性膀胱炎/膀胱疼痛综合征(IC/BPS)小鼠模型的疗效及其潜在机制。 方法 通过随机化方法将6~8周龄雌性C57BL/6小鼠分为:对照组、PPS处理组(PPS组)、CYP诱导组(CYP组)和CYP+PPS联合处理组(C+P组),6只/组。PPS以25 mg/kg剂量连续3周灌胃处理,CYP以50 mg/kg剂量在第4周分3次腹腔注射以建立IC/BPS模型。采用16S rDNA测序及非靶向代谢组学分析肠道菌群与代谢产物变化;通过粪便微生物移植(FMT)实验(CYP-FMT与C+P-FMT受体组)验证菌群介导作用。体外以LPS诱导人膀胱上皮细胞(SV-HUC-1)炎症模型,分组探究脱氧胆酸(DCA)及TGR5抑制剂(SBI-115)对屏障功能的影响。 结果 PPS治疗提升了CYP诱导的IC/BPS小鼠模型的机械疼痛阈值,改善了尿动力学参数,并减轻了膀胱炎症及屏障损伤(P<0.05)。PPS通过增加肠道中嗜木聚糖真杆菌的丰度和促进DCA产生,调节了肠道菌群和胆汁酸代谢(P<0.05)。FMT实验证实了PPS的疗效依赖于肠道菌群。在细胞层面,DCA激活TGR5受体,减轻了LPS诱导的SV-HUC-1细胞炎症和屏障损伤(P<0.05)。 结论 PPS通过富集嗜木聚糖真杆菌菌群促进DCA生成,激活TGR5信号通路,从而减轻膀胱炎症并修复屏障功能。本研究首次揭示PPS通过菌群-胆汁酸-TGR5轴调控IC/BPS的新机制,为靶向肠道微生态治疗膀胱疾病提供理论依据。
祝越轩, 诸章睿, 吴芃. 多硫酸戊聚糖缓解环磷酰胺诱导的小鼠间质性膀胱炎/膀胱疼痛综合征的机制:基于调节肠道微生物群和胆汁酸代谢[J]. 南方医科大学学报, 2025, 45(6): 1270-1279.
Yuexuan ZHU, Zhangrui ZHU, Peng WU. Pentosan polysulfate alleviates cyclophosphamide-induced interstitial cystitis/bladder pain syndrome in mice by modulating gut microbiota and bile acid metabolism[J]. Journal of Southern Medical University, 2025, 45(6): 1270-1279.
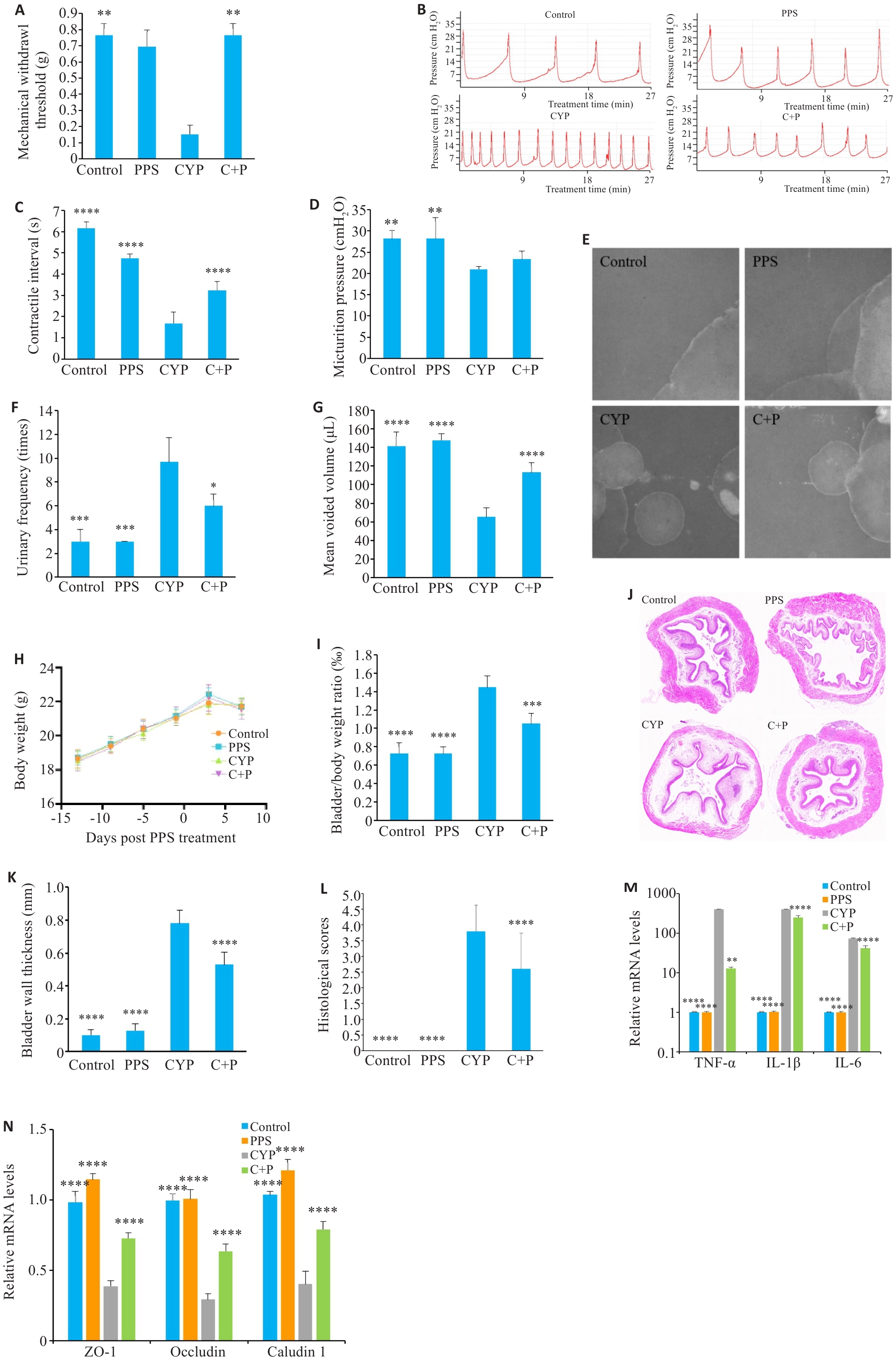
图1 PPS对CYP诱导的IC/BPS小鼠模型的影响
Fig.1 Effect of pentosan polysulfate (PPS) on cyclophosphamide (CYP)‑ induced interstitial cystitis/bladder pain syndrome (IC/BPS) in mice. A: Comparison of mechanical pain thresholds of the mice across the groups. B-D: Urodynamic test results and their parameters in different groups. E-G: Voiding cystometry analysis of changes in voiding behavior of the mice. H: Body weight change curve of the mice. I: Bladder to body weight ratio of the mice. J-L: HE staining of the bladder tissues in different groups (Original magnification: ×10). M: PPS reduces elevated mRNA levels of inflammatory factors induced by CYP. N: PPS lowers mRNA levels of tight junction proteins in CYP-treated mice. Data are presented as Mean±SE. Statistical significance was determined by one-way ANOVA or unpaired Student's t-test. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 vs CYP. C+P: CYP+PPS.
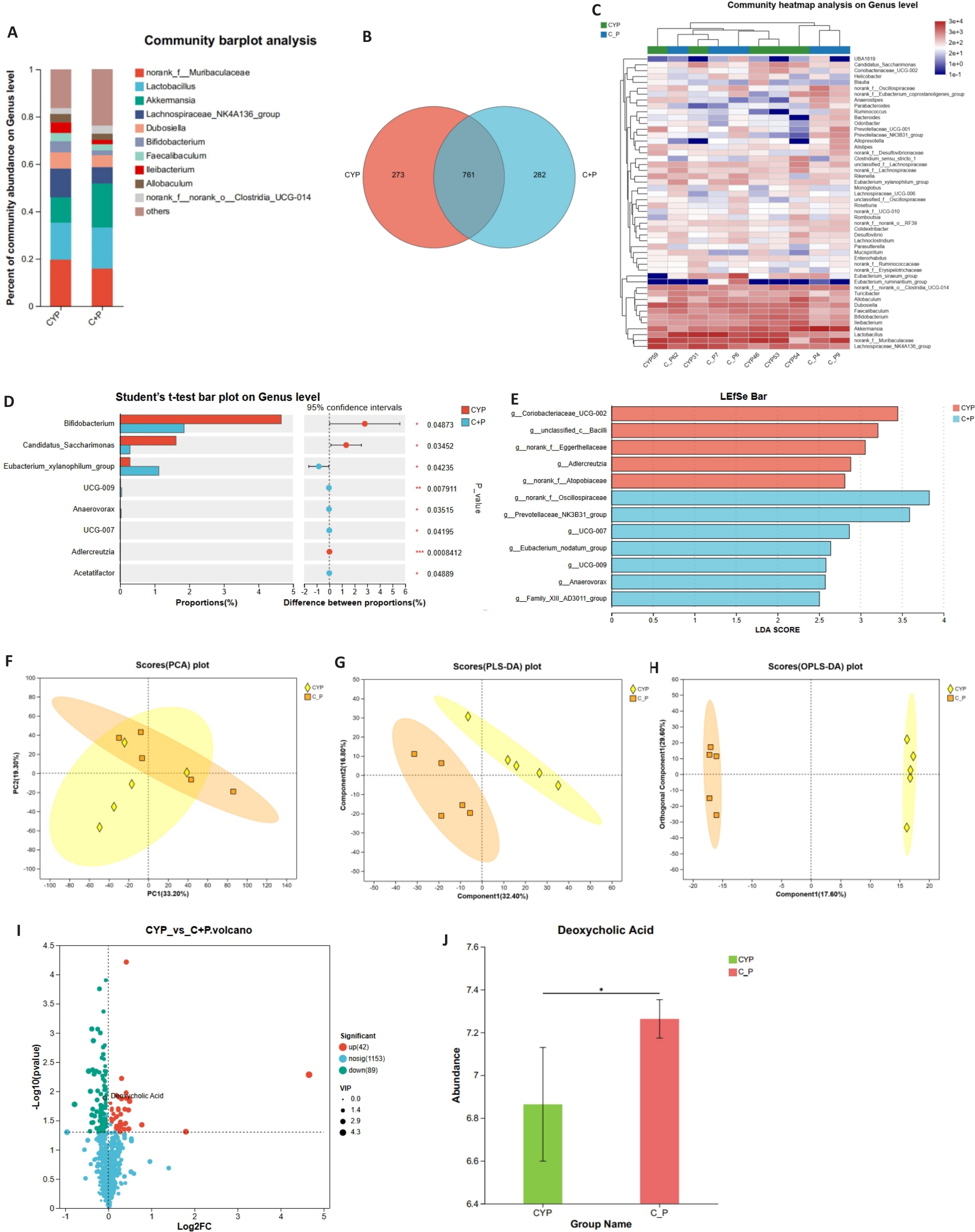
图2 PPS对CYP诱导的IC/BPS小鼠肠道菌群及其代谢物组成的影响
Fig.2 Impact of PPS on gut microbiota and its metabolites in mice with CYP-induced IC/BPS. A: Comparison of gut microbiota composition at the genus level between CYP group and C+P group (n=5). B: Venn diagram showing the shared and unique species of gut microbiota between CYP group and C+P group (n=5). C: Heatmap displaying differences in gut microbiota composition at the genus level (n=5). D: Statistical test for differences in gut microbiota composition at the genus level (n=5). E: LEfSe analysis confirms significant differences in gut microbial community composition between CYP group and C+P group. F-H: Principal component analysis (PCA), partial least squares discriminant analysis (PLS-DA), and orthogonal partial least squares discriminant analysis (OPLS-DA) of non-targeted metabolomics data. I: Volcano plot showing differential metabolites between CYP group and C+P group. J: Comparison of DCA concentration in cecal contents between the two groups. *P<0.05.
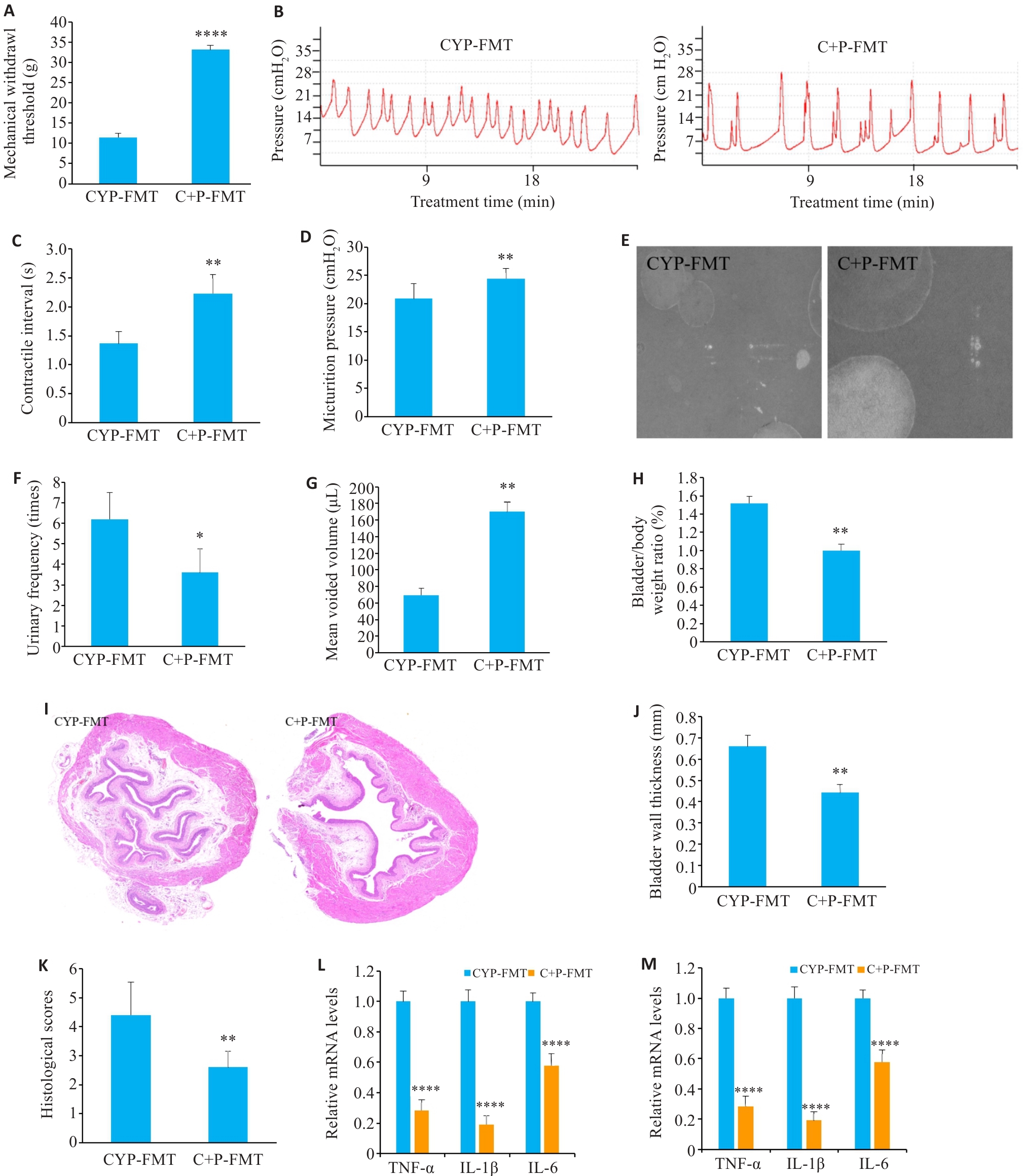
图3 PPS干预后小鼠的粪便FMT对CYP诱导的IC/BPS的影响
Fig.3 Effect of fecal microbiota transplantation (FMT) from PPS-treated mice on CYP-induced IC/BPS. A: Comparison of mechanical pain thresholds among the groups. B-D: Results of urodynamic testing and their parameters. E-G: Voiding cystometry analysis. H: Bladder to body weight ratio. I-K: HE staining of the bladder tissue in the groups (×10). L: C+P-FMT reduces CYP-induced elevation of mRNA levels of inflammatory factors. M: C+P-FMT restores CYP-induced decrease of mRNA levels of tight junction proteins. Data are presented as Mean±SE. Statistical significance was determined by one-way ANOVA or unpaired Student's t-test. *P<0.05, **P<0.01, ****P<0.0001 vs CYP+FMT.
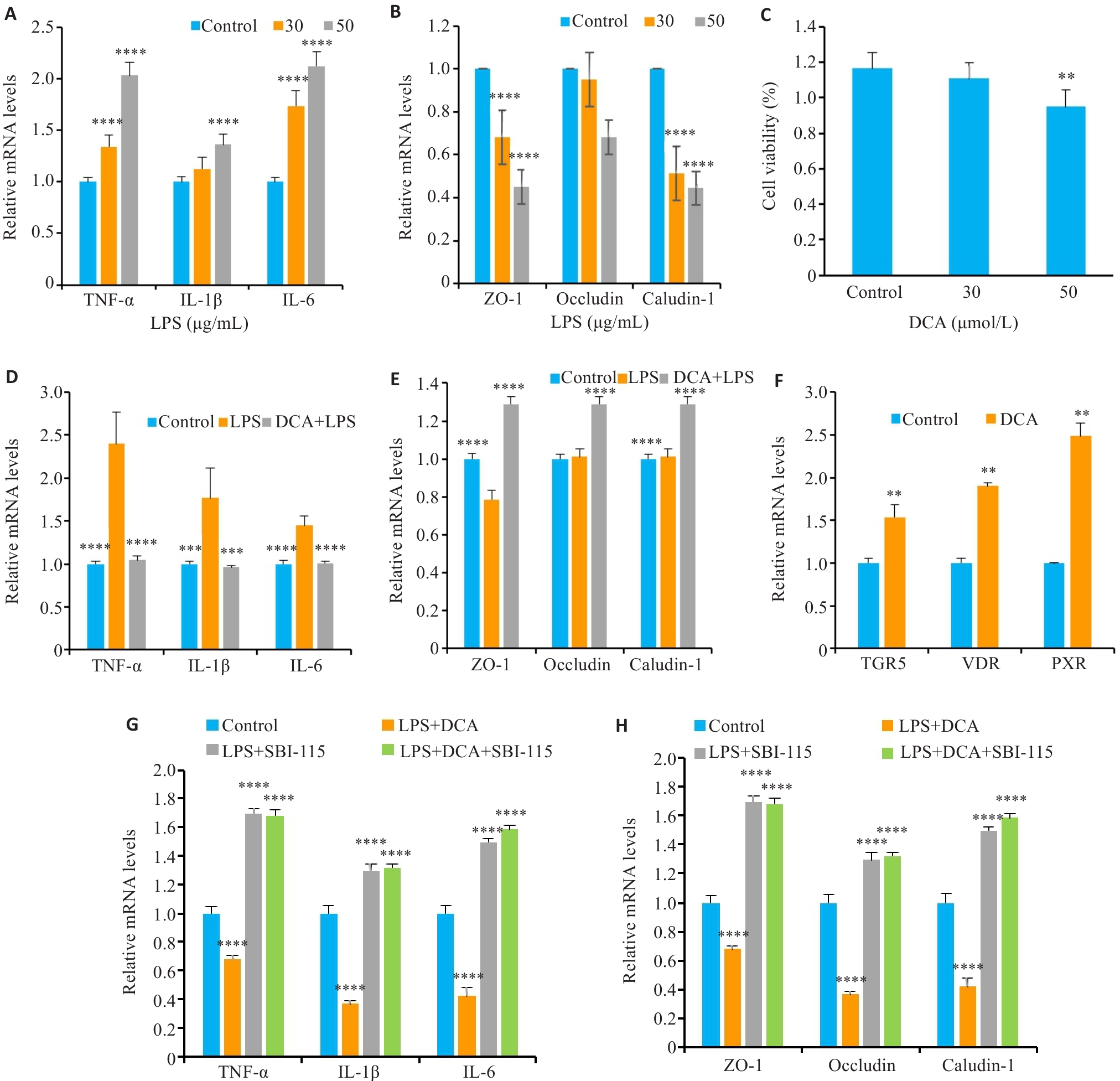
图4 PPS代谢相关产物DCA通过激活TGR5缓解膀胱上皮炎症损伤
Fig.4 PPS metabolism-related product DCA alleviates bladder epithelial inflammatory injury by activating TGR5. A: Expression levels of inflammatory factors in SV-HUC-1 cells treated with different concentrations of LPS. B: Expression levels of tight junction proteins in SV-HUC-1 cells treated with different concentrations of LPS. C: Viability of SV-HUC-1 cells treated with different concentrations of DCA. D: DCA reduces LPS-induced inflammatory factor expression levels. E: DCA restores LPS-induced reduction of expressions of tight junction proteins in SV-HUC-1 cells. F: DCA stimulates bile acid receptor mRNA expression in SV-HUC-1 cells. G: SBI-115 inhibits the effect of DCA on inflammatory factor expressions in LPS-induced SV-HUC-1 cells. H: SBI-115 inhibits the effect of DCA on tight junction protein expressions in LPS-induced SV-HUC-1 cells. Data are presented as Mean±SE. Statistical significance was determined by one-way ANOVA or unpaired Student's t-test. **P<0.01, ***P<0.001, ****P<0.0001 vs Control (A-C and F-H) or LPS (D-E).
| 1 | Li J, Yi X, Ai JZ. Broaden horizons: the advancement of interstitial cystitis/bladder pain syndrome[J]. Int J Mol Sci, 2022, 23(23): 14594. doi:10.3390/ijms232314594 |
| 2 | Quentin Clemens J, Erickson DR, Varela NP, et al. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome[J]. J Urol, 2022, 208(1): 34-42. doi:10.1097/ju.0000000000002756 |
| 3 | Mohammad A, Laboulaye MA, Shenhar C, et al. Mechanisms of oxidative stress in interstitial cystitis/bladder pain syndrome[J]. Nat Rev Urol, 2024, 21(7): 433-49. doi:10.1038/s41585-023-00850-y |
| 4 | Adak A, Khan MR. An insight into gut microbiota and its functionalities[J]. Cell Mol Life Sci, 2019, 76(3): 473-93. doi:10.1007/s00018-018-2943-4 |
| 5 | El-Salhy M, Hatlebakk JG, Hausken T. Diet in irritable bowel syndrome (IBS): interaction with gut microbiota and gut hormones[J]. Nutrients, 2019, 11(8): 1824. doi:10.3390/nu11081824 |
| 6 | Coelho A, Oliveira R, Antunes-Lopes T, et al. Partners in crime: NGF and BDNF in visceral dysfunction[J]. Curr Neuropharmacol, 2019, 17(11): 1021-38. doi:10.2174/1570159x17666190617095844 |
| 7 | Fu CW, Zhang YW, Liang LH, et al. The microbiota in patients with interstitial cystitis/bladder pain syndrome: a systematic review[J]. BJU Int, 2024, 134(6): 869-80. doi:10.1111/bju.16439 |
| 8 | Lindeke-Myers A, Hanif AM, Jain N. Pentosan polysulfate maculopathy[J]. Surv Ophthalmol, 2022, 67(1): 83-96. doi:10.1016/j.survophthal.2021.05.005 |
| 9 | Anderson VR, Perry CM. Pentosan polysulfate: a review of its use in the relief of bladder pain or discomfort in interstitial cystitis[J]. Drugs, 2006, 66(6): 821-35. doi:10.2165/00003495-200666060-00006 |
| 10 | Smith MM, Melrose J. Xylan prebiotics and the gut microbiome promote health and wellbeing: potential novel roles for pentosan polysulfate[J]. Pharmaceuticals, 2022, 15(9): 1151. doi:10.3390/ph15091151 |
| 11 | Augé C, Gamé X, Vergnolle N, et al. Characterization and validation of a chronic model of cyclophosphamide-induced interstitial cystitis/bladder pain syndrome in rats[J]. Front Pharmacol, 2020, 11: 1305. doi:10.3389/fphar.2020.01305 |
| 12 | Wu J, Guan TJ, Zheng SR, et al. Inhibition of inflammation by pentosan polysulfate impedes the development and progression of severe diabetic nephropathy in aging C57B6 mice[J]. Lab Invest, 2011, 91(10): 1459-71. doi:10.1038/labinvest.2011.93 |
| 13 | Mroz MS, Lajczak NK, Goggins BJ, et al. The bile acids, deoxycholic acid and ursodeoxycholic acid, regulate colonic epithelial wound healing[J]. Am J Physiol Gastrointest Liver Physiol, 2018, 314(3): G378-87. doi:10.1152/ajpgi.00435.2016 |
| 14 | Chambers LM, Esakov Rhoades EL, Bharti R, et al. Disruption of the gut microbiota confers cisplatin resistance in epithelial ovarian cancer[J]. Cancer Res, 2022, 82(24): 4654-69. doi:10.1158/0008-5472.can-22-0455 |
| 15 | Zhou ZP, Qiu YF, Li K, et al. Unraveling the impact of Lactobacillus spp. and other urinary microorganisms on the efficacy of mirabegron in female patients with overactive bladder[J]. Front Cell Infect Microbiol, 2022, 12: 1030315. doi:10.3389/fcimb.2022.1030315 |
| 16 | Bon K, Lichtensteiger CA, Wilson SG, et al. Characterization of cyclophosphamide cystitis, a model of visceral and referred pain, in the mouse: species and strain differences[J]. J Urol, 2003, 170(3): 1008-12. doi:10.1097/01.ju.0000079766.49550.94 |
| 17 | Wegner KA, Abler LL, Oakes SR, et al. Void spot assay procedural optimization and software for rapid and objective quantification of rodent voiding function, including overlapping urine spots[J]. Am J Physiol Renal Physiol, 2018, 315(4): F1067-80. doi:10.1152/ajprenal.00245.2018 |
| 18 | Bjorling DE, Wang ZY, Vezina CM, et al. Evaluation of voiding assays in mice: impact of genetic strains and sex[J]. Am J Physiol Renal Physiol, 2015, 308(12): F1369-78. doi:10.1152/ajprenal.00072.2015 |
| 19 | Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C(T)) Method[J]. Methods, 2001, 25(4): 402-8. doi:10.1006/meth.2001.1262 |
| 20 | Zhang ML, Chekan JR, Dodd D, et al. Xylan utilization in human gut commensal bacteria is orchestrated by unique modular organization of polysaccharide-degrading enzymes[J]. Proc Natl Acad Sci USA, 2014, 111(35): E3708-17. doi:10.1073/pnas.1406156111 |
| 21 | Ridlon JM, Ikegawa S, Alves JMP, et al. Clostridium scindens: a human gut microbe with a high potential to convert glucocorticoids into androgens[J]. J Lipid Res, 2013, 54(9): 2437-49. doi:10.1194/jlr.M038869 |
| 22 | Chelakkot C, Choi Y, Kim DK, et al. Akkermansia muciniphila-derived extracellular vesicles influence gut permeability through the regulation of tight junctions[J]. Exp Mol Med, 2018, 50(2): e450. doi:10.1038/emm.2017.282 |
| 23 | Blacher E, Levy M, Tatirovsky E, et al. Microbiome-modulated metabolites at the interface of host immunity[J]. J Immunol, 2017, 198(2): 572-80. doi:10.4049/jimmunol.1601247 |
| 24 | Kawamata Y, Fujii R, Hosoya M, et al. A G protein-coupled receptor responsive to bile acids[J]. J Biol Chem, 2003, 278(11): 9435-40. doi:10.1074/jbc.m209706200 |
| 25 | Masyuk TV, Masyuk AI, Lorenzo Pisarello M, et al. TGR5 contributes to hepatic cystogenesis in rodents with polycystic liver diseases through cyclic adenosine monophosphate/Gαs signaling[J]. Hepatology, 2017, 66(4): 1197-218. doi:10.1002/hep.29284 |
| 26 | Zhao CJ, Wu KY, Hao HY, et al. Gut microbiota-mediated secondary bile acid alleviates Staphylococcus aureus-induced mastitis through the TGR5-cAMP-PKA-NF-κB/NLRP3 pathways in mice[J]. NPJ Biofilms Microbiomes, 2023, 9(1): 8. doi:10.1038/s41522-023-00374-8 |
| 27 | Chiang JYL, Ferrell JM. Bile acid receptors FXR and TGR5 signaling in fatty liver diseases and therapy[J]. Am J Physiol Gastrointest Liver Physiol, 2020, 318(3): G554-73. doi:10.1152/ajpgi.00223.2019 |
| 28 | Yang F, Mao CY, Guo LL, et al. Structural basis of GPBAR activation and bile acid recognition[J]. Nature, 2020, 587(7834): 499-504. doi:10.1038/s41586-020-2569-1 |
| 29 | Plovier H, Everard A, Druart C, et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice[J]. Nat Med, 2017, 23(1): 107-13. doi:10.1038/nm.4236 |
| 30 | Russell DW. The enzymes, regulation, and genetics of bile acid synthesis[J]. Annu Rev Biochem, 2003, 72: 137-74. doi:10.1146/annurev.biochem.72.121801.161712 |
| 31 | Dai C, Huang YH, Jiang M. Fecal microbiota transplantation for irritable bowel syndrome: Current evidence and perspectives[J]. World J Gastroenterol, 2024, 30(16): 2179-83. doi:10.3748/wjg.v30.i16.2179 |
| 32 | Zhao SN, Gong ZZ, Zhou JF, et al. Deoxycholic acid triggers NLRP3 inflammasome activation and aggravates DSS-induced colitis in mice[J]. Front Immunol, 2016, 7: 536. doi:10.3389/fimmu.2016.00536 |
| [1] | 黄凯悦, 齐景馨, 罗文谦, 林怡萱, 陈梅妹, 甘慧娟. 温胆汤通过调控肠道菌群-胆汁酸轴改善代谢综合征痰证大鼠的代谢表型[J]. 南方医科大学学报, 2025, 45(6): 1174-1184. |
| [2] | 王旋成, 朱一帆, 周海琳, 黄宗声, 陈鸿炜, 张嘉豪, 杨珊伊, 陈广辉, 张淇淞. 血清非靶向代谢组学联合靶向胆汁酸代谢组学筛查结直肠癌的潜在生物标志物[J]. 南方医科大学学报, 2023, 43(3): 443-453. |
| [3] | 贺娇娇,吕麟亚,彭俊伟,李长春,孔祥如,章 均,彭 亮. 生酮饮食抑制裸鼠皮下移植人神经母细胞瘤的生长[J]. 南方医科大学学报, 2020, 40(08): 1155-1164. |
| [4] | 陈春秀, 周 永, 黄荣凤, 王妙然, 李 月, 李继斌. 胆汁酸通过p-STAT3-SOCS3途径调节下丘脑神经元食欲及抑制信号肽表达[J]. 南方医科大学学报, 2020, 40(07): 1001-1007. |
| [5] | 徐艳军,易甜甜,徐肖肖,裴夫瑜,何岳林,吴学东. 环磷酰胺对铁过载小鼠骨髓造血干细胞的影响[J]. 南方医科大学学报, 2020, 40(01): 110-117. |
| [6] | 罗东,汪威,陈俊林,刘宝茹,陈锦云,王嫣,陈文直. 低强度脉冲超声对阿霉素联合环磷酰胺化疗后大鼠造血功能的影响[J]. 南方医科大学学报, 2019, 39(07): 836-. |
| [7] | 罗东,罗月苹,刘宝茹,梁丹丹,蒋璟玮,汪威,陈俊林,王嫣,陈文直. 新西兰兔再生障碍性贫血模型的建立[J]. 南方医科大学学报, 2017, 37(12): 1660-. |
| [8] | 王斐,赵丛,田映红,尹艳茹. 胆汁酸影响兔肺泡灌洗液的表面张力[J]. 南方医科大学学报, 2014, 34(10): 1465-. |
| [9] | 辛小敏,钟慕晓,张珊珊,彭 瑶,朱 薇,张亚历. 胆汁酸核受体激动剂对脂联素及其受体的影响[J]. 南方医科大学学报, 2014, 34(01): 109-. |
| [10] | 王斐,赵丛,田映红,尹艳茹. 高胆汁酸血症影响兔呼吸功能的实验研究[J]. 南方医科大学学报, 2013, 33(08): 1181-. |
| [11] | 冯晨,唐锁勤,王建文,刘英,杨光. Topotecan plus cyclophosphamide as maintenance chemotherapy for children with high-risk neuroblastoma in complete remission: short-term curative effects and toxicity[J]. 南方医科大学学报, 2013, 33(08): 1107-. |
| [12] | 彭萍,吴璇,关婷,陈为,张艳玲,张伟,杨传红,叶长烂,王善林. 1-磷酸-神经鞘氨醇干预化疗对荷瘤小鼠卵巢功能和抑瘤效果的影响[J]. 南方医科大学学报, 2012, 32(03): 383-. |
| [13] | 梁利霞,晁岚,邓晓惠,于红玲,刘文军,王旭平. 小鼠骨髓移植修复卵巢功能损伤[J]. 南方医科大学学报, 2011, 31(09): 1534-. |
| [14] | 刘青云; 刘春城; 胡良燕;. 不同化疗方案对手术后卵巢上皮性癌疗效的对比研究[J]. 南方医科大学学报, 2006, 26(06): 863-864. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||